Acyl-coenzyme A thioesterase 4 is an enzyme that in humans is encoded by the ACOT4 gene. [5] [6] [7]
Acyl-coenzyme A thioesterase 4 is an enzyme that in humans is encoded by the ACOT4 gene. [5] [6] [7]
The protein encoded by the ACOT4 gene is part of a family of Acyl-CoA thioesterases, which catalyze the hydrolysis of various Coenzyme A esters of various molecules to the free acid plus CoA. These enzymes have also been referred to in the literature as acyl-CoA hydrolases, acyl-CoA thioester hydrolases, and palmitoyl-CoA hydrolases. The reaction carried out by these enzymes is as follows:
CoA ester + H2O → free acid + coenzyme A
These enzymes use the same substrates as long-chain acyl-CoA synthetases, but have a unique purpose in that they generate the free acid and CoA, as opposed to long-chain acyl-CoA synthetases, which ligate fatty acids to CoA, to produce the CoA ester. [8] The role of the ACOT- family of enzymes is not well understood; however, it has been suggested that they play a crucial role in regulating the intracellular levels of CoA esters, Coenzyme A, and free fatty acids. Recent studies have shown that Acyl-CoA esters have many more functions than simply an energy source. These functions include allosteric regulation of enzymes such as acetyl-CoA carboxylase, [9] hexokinase IV, [10] and the citrate condensing enzyme. Long-chain acyl-CoAs also regulate opening of ATP-sensitive potassium channels and activation of Calcium ATPases, thereby regulating insulin secretion. [11] A number of other cellular events are also mediated via acyl-CoAs, for example signal transduction through protein kinase C, inhibition of retinoic acid-induced apoptosis, and involvement in budding and fusion of the endomembrane system. [12] [13] [14] Acyl-CoAs also mediate protein targeting to various membranes and regulation of G Protein α subunits, because they are substrates for protein acylation. [15] In the mitochondria, acyl-CoA esters are involved in the acylation of mitochondrial NAD+ dependent dehydrogenases; because these enzymes are responsible for amino acid catabolism, this acylation renders the whole process inactive. This mechanism may provide metabolic crosstalk and act to regulate the NADH/NAD+ ratio in order to maintain optimal mitochondrial beta oxidation of fatty acids. [16] The role of CoA esters in lipid metabolism and numerous other intracellular processes are well defined, and thus it is hypothesized that ACOT- enzymes play a role in modulating the processes these metabolites are involved in. [17]
Enoyl-CoA-(∆) isomerase (EC 5.3.3.8, also known as dodecenoyl-CoA- isomerase, 3,2-trans-enoyl-CoA isomerase, ∆3 ,∆2 -enoyl-CoA isomerase, or acetylene-allene isomerase, is an enzyme that catalyzes the conversion of cis- or trans-double bonds of coenzyme A bound fatty acids at gamma-carbon to trans double bonds at beta-carbon as below:
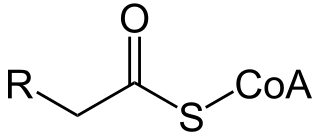
Acyl-CoA is a group of coenzymes that metabolize fatty acids. Acyl-CoA's are susceptible to beta oxidation, forming, ultimately, acetyl-CoA. The acetyl-CoA enters the citric acid cycle, eventually forming several equivalents of ATP. In this way, fats are converted to ATP, the universal biochemical energy carrier.
The long chain fatty acyl-CoA ligase is an enzyme of the ligase family that activates the oxidation of complex fatty acids. Long chain fatty acyl-CoA synthetase catalyzes the formation of fatty acyl-CoA by a two-step process proceeding through an adenylated intermediate. The enzyme catalyzes the following reaction,
Serine hydrolases are one of the largest known enzyme classes comprising approximately ~200 enzymes or 1% of the genes in the human proteome. A defining characteristic of these enzymes is the presence of a particular serine at the active site, which is used for the hydrolysis of substrates. The hydrolysis of the ester or peptide bond proceeds in two steps. First, the acyl part of the substrate is transferred to the serine, making a new ester or amide bond and releasing the other part of the substrate is released. Later, in a slower step, the bond between the serine and the acyl group is hydrolyzed by water or hydroxide ion, regenerating free enzyme. Unlike other, non-catalytic, serines, the reactive serine of these hydrolases is typically activated by a proton relay involving a catalytic triad consisting of the serine, an acidic residue and a basic residue, although variations on this mechanism exist.
The enzyme choloyl-CoA hydrolase (EC 3.1.2.27) catalyzes the reaction
Palmitoyl-CoA hydrolase (EC 3.1.2.2) is an enzyme in the family of hydrolases that specifically acts on thioester bonds. It catalyzes the hydrolysis of long chain fatty acyl thioesters of acyl carrier protein or coenzyme A to form free fatty acid and the corresponding thiol:

Palmitoyl protein hydrolase/thioesterases is an enzyme (EC 3.1.2.22) that removes thioester-linked fatty acyl groups such as palmitate from modified cysteine residues in proteins or peptides during lysosomal degradation. It catalyzes the reaction

Long-chain-fatty-acid—CoA ligase 1 is an enzyme that in humans is encoded by the ACSL1 gene.

Acyl-coenzyme A thioesterase 8 is an enzyme that in humans is encoded by the ACOT8 gene.

Long-chain-fatty-acid—CoA ligase 5 is an enzyme that in humans is encoded by the ACSL5 gene.

Long-chain-fatty-acid—CoA ligase 3 is an enzyme that in humans is encoded by the ACSL3 gene.

Acyl-CoA thioesterase 2, also known as ACOT2, is an enzyme which in humans is encoded by the ACOT2 gene.

Very long-chain acyl-CoA synthetase is an enzyme that in humans is encoded by the SLC27A2 gene.

Cytosolic acyl coenzyme A thioester hydrolase is an enzyme that in humans is encoded by the ACOT7 gene.

Acyl-coenzyme A thioesterase 11 also known as StAR-related lipid transfer protein 14 (STARD14) is an enzyme that in humans is encoded by the ACOT11 gene. This gene encodes a protein with acyl-CoA thioesterase activity towards medium (C12) and long-chain (C18) fatty acyl-CoA substrates which relies on its StAR-related lipid transfer domain. Expression of a similar murine protein in brown adipose tissue is induced by cold exposure and repressed by warmth. Expression of the mouse protein has been associated with obesity, with higher expression found in obesity-resistant mice compared with obesity-prone mice. Alternative splicing results in two transcript variants encoding different isoforms.

Bile acyl-CoA synthetase is an enzyme that in humans is encoded by the SLC27A5 gene.

Acyl-CoA thioesterase 6 is a protein that in humans is encoded by the ACOT6 gene. The protein, also known as C14orf42, is an enzyme with thioesterase activity.

Acyl-CoA thioesterase 9 is a protein that is encoded by the human ACOT9 gene. It is a member of the acyl-CoA thioesterase superfamily, which is a group of enzymes that hydrolyze Coenzyme A esters. There is no known function, however it has been shown to act as a long-chain thioesterase at low concentrations, and a short-chain thioesterase at high concentrations.

Acyl-CoA thioesterase 13 is a protein that in humans is encoded by the ACOT13 gene. This gene encodes a member of the thioesterase superfamily. In humans, the protein co-localizes with microtubules and is essential for sustained cell proliferation.

Acyl-CoA thioesterase 1 is a protein that in humans is encoded by the ACOT1 gene.