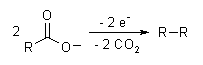Reactions
Organic oxidations take place at the anode. Compounds are reduced at the cathode. Radical intermediates are often invoked. The initial reaction takes place at the surface of the electrode and then the intermediates diffuse into the solution where they participate in secondary reactions.
The yield of an electrosynthesis is expressed both in terms of the chemical yield and current efficiency. Current efficiency is the ratio of Coulombs consumed in forming the products to the total number of Coulombs passed through the cell. Side reactions decrease the current efficiency.
The potential drop between the electrodes determines the rate constant of the reaction. Electrosynthesis is carried out with either constant potential or constant current. The reason one chooses one over the other is due to a trade-off of ease of experimental conditions versus current efficiency. Constant potential uses current more efficiently because the current in the cell decreases with time due to the depletion of the substrate around the working electrode (stirring is usually necessary to decrease the diffusion layer around the electrode). This is not the case under constant current conditions, however. Instead, as the substrate's concentration decreases the potential across the cell increases in order to maintain the fixed reaction rate. This consumes current in side reactions produced outside the target voltage.
Anodic oxidations
C-C coupling
Oxidation of a carbanion can lead to a coupling reaction for instance in the electrosynthesis of the tetramethyl ester of ethanetetracarboxylic acid from the corresponding malonate ester [9]
A well-known electrosynthesis is the Kolbe electrolysis, in which two carboxylic acids decarboxylate, and the remaining radicals couple:
A variation is called the non-Kolbe reaction when a heteroatom (nitrogen or oxygen) is present at the α-position. The intermediate oxonium ion is trapped by a nucleophile, usually solvent.
Amines to nitrile
Anodic electrosynthesis oxidize primary aliphatic amine to nitrile. [10] [11]
α-amino acids form nitriles and carbon dioxide via oxidative decarboxylation at AgO anodes (the latter is formed in-situ by oxidation of Ag2O): [7] [12] [ verification needed ]
Cyanoacetic acid from cathodic reduction of carbon dioxide and anodic oxidation of acetonitrile. [13]
Amides
Amides can be oxidized to N-acyliminium ions, which can be captured by various nucleophiles, for example:
This reaction type is called a Shono oxidation. An example is the α-methoxylation of N-carbomethoxypyrrolidine [14]
Alcohols to carboxylic acids
Propiolic acid is prepared commercially by oxidizing propargyl alcohol at a lead dioxide electrode. Similarly, 1,4-butynediol is oxidized to acetylenedicarboxylic acid. [15]
Cathodic reductions
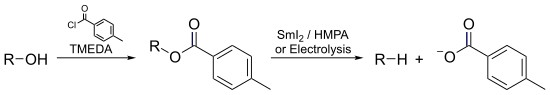
- In the Markó–Lam deoxygenation, an alcohol could be almost instantaneously deoxygenated by electroreducing its toluate ester.
- In concept, adiponitrile is prepared from dimerizing acrylonitrile: [16]
- 2 CH2=CHCN + 2 e− + 2 H+ → NC(CH2)4CN
- In practice,the cathodic hydrodimerization of activated olefins is applied industrially in the synthesis of adiponitrile from two equivalents of acrylonitrile
:This article needs additional citations for verification .(February 2022) -

- The cathodic reduction of arene compounds to the 1,4-dihydro derivatives is similar to a Birch reduction. Examples from industry are the reduction of phthalic acid:
and the reduction of 2-methoxynaphthalene:
- The Tafel rearrangement, named for Julius Tafel, was at one time an important method for the synthesis of certain hydrocarbons from alkylated ethyl acetoacetate, a reaction accompanied by the rearrangement reaction of the alkyl group: [17] [18]
- The cathodic reduction of a nitrile to a primary amine in a divided cell; the cathodic reduction of benzyl cyanide to phenethylamine is shown: [19]
- Cathodic reduction of a nitroalkene can give the oxime in good yield. At higher negative reduction potentials, the nitroalkene can be reduced further, giving the primary amine but with lower yield. [20]
- Azobenzene is prepared in industrial electrosynthesis using nitrobenzene. [16]
- An electrochemical carboxylation of a para-isobutyl benzyl chloride to Ibuprofen is promoted under supercritical carbon dioxide. [21]
- Cathodic reduction of a carboxylic acid (oxalic acid) to an aldehyde (glyoxylic acid, shows as the rare aldehyde form) in a divided cell: [22] [23]
- Originally phenylpropanoic acid could be prepared from reduction of cinnamic acid by electrolysis. [24]
- An electrocatalysis by a copper complex helps reduce carbon dioxide to oxalic acid; this conversion uses carbon dioxide as a feedstock to generate oxalic acid. [25]
- It has been reported that formate can be formed by the electrochemical reduction of CO2 (in the form of bicarbonate) at a lead cathode at pH 8.6: [26]
- HCO−3 + H2O + 2e− → HCO−2 + 2OH−
or
- CO2 + H2O + 2e− → HCO−2 + OH−
If the feed is CO2 and oxygen is evolved at the anode, the total reaction is:
- CO2 + OH− → HCO−2 + 1/2 O2
Redox reactions
- Cathodic reduction of carbon dioxide and anodic oxidation of acetonitrile afford cyanoacetic acid. [13]
- An electrosynthesis employing alternating current prepares phenol at both the cathode and the anode. [27]
- A green electrochemical procedure for methylene blue, using only dimethyl-4-phenylenediamine and sulfide ions has been proposed. [28]
Electrofluorination
In organofluorine chemistry, many perfluorinated compounds are prepared by electrochemical synthesis, which is conducted in liquid HF at voltages near 5–6 V using Ni anodes. The method was invented in the 1930s. [29] Amines, alcohols, carboxylic acids, and sulfonic acids are converted to perfluorinated derivatives using this technology. A solution or suspension of the hydrocarbon in hydrogen fluoride is electrolyzed at 5–6 V to produce high yields of the perfluorinated product.