
Fanconi anaemia (FA) is a rare, AR, genetic disease resulting in impaired response to DNA damage in the FA/BRCA pathway. Although it is a very rare disorder, study of this and other bone marrow failure syndromes has improved scientific understanding of the mechanisms of normal bone marrow function and development of cancer. Among those affected, the majority develop cancer, most often acute myelogenous leukemia (AML), MDS, and liver tumors. 90% develop aplastic anemia by age 40. About 60–75% have congenital defects, commonly short stature, abnormalities of the skin, arms, head, eyes, kidneys, and ears, and developmental disabilities. Around 75% have some form of endocrine problem, with varying degrees of severity. 60% of FA is FANC-A, 16q24.3, which has later onset bone marrow failure.

The Medical Research Council (MRC) Laboratory of Molecular Biology (LMB) is a research institute in Cambridge, England, involved in the revolution in molecular biology which occurred in the 1950–60s. Since then it has remained a major medical research laboratory at the forefront of scientific discovery, dedicated to improving the understanding of key biological processes at atomic, molecular and cellular levels using multidisciplinary methods, with a focus on using this knowledge to address key issues in human health.

Fanconi anemia group C protein is a protein that in humans is encoded by the FANCC gene. This protein delays the onset of apoptosis and promotes homologous recombination repair of damaged DNA. Mutations in this gene result in Fanconi anemia, a human rare disorder characterized by cancer susceptibility and cellular sensitivity to DNA crosslinks and other damages.

Fanconi anaemia, complementation group A, also known as FAA, FACA and FANCA, is a protein which in humans is encoded by the FANCA gene. It belongs to the Fanconi anaemia complementation group (FANC) family of genes of which 12 complementation groups are currently recognized and is hypothesised to operate as a post-replication repair or a cell cycle checkpoint. FANCA proteins are involved in inter-strand DNA cross-link repair and in the maintenance of normal chromosome stability that regulates the differentiation of haematopoietic stem cells into mature blood cells.

Fanconi anemia group D2 protein is a protein that in humans is encoded by the FANCD2 gene. The Fanconi anemia complementation group (FANC) currently includes FANCA, FANCB, FANCC, FANCD1, FANCD2, FANCE, FANCF, FANCG, FANCI, FANCJ, FANCL, FANCM, FANCN and FANCO.

Fanconi anemia group G protein is a protein that in humans is encoded by the FANCG gene.
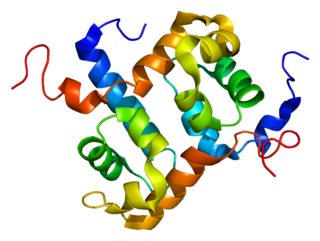
ERCC4 is a protein designated as DNA repair endonuclease XPF that in humans is encoded by the ERCC4 gene. Together with ERCC1, ERCC4 forms the ERCC1-XPF enzyme complex that participates in DNA repair and DNA recombination.

Fanconi anemia group F protein is a protein that in humans is encoded by the FANCF gene.
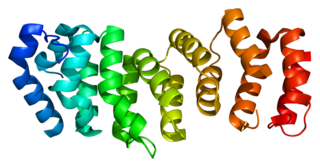
Fanconi anemia, complementation group E protein is a protein that in humans is encoded by the FANCE gene. The Fanconi anemia complementation group (FANC) currently includes FANCA, FANCB, FANCC, FANCD1, FANCD2, FANCE, FANCF, FANCG, and FANCL. Fanconi anemia is a genetically heterogeneous recessive disorder characterized by cytogenetic instability, hypersensitivity to DNA cross-linking agents, increased chromosomal breakage, and defective DNA repair. The members of the Fanconi anemia complementation group do not share sequence similarity; they are related by their assembly into a common nuclear protein complex. This gene encodes the protein for complementation groufcrp E.

E3 ubiquitin-protein ligase FANCL is an enzyme that in humans is encoded by the FANCL gene.

Fanconi anemia group B protein is a protein that in humans is encoded by the FANCB gene.

Fanconi anemia, complementation group I (FANCI) also known as KIAA1794, is a protein which in humans is encoded by the FANCI gene. Mutations in the FANCI gene are known to cause Fanconi anemia.

Partner and localizer of BRCA2, also known as PALB2 or FANCN, is a protein which in humans is encoded by the PALB2 gene.

SLX4 is a protein involved in DNA repair, where it has important roles in the final steps of homologous recombination. Mutations in the gene are associated with the disease Fanconi anemia.
Simon Joseph Boulton is a British scientist who has made important contributions to the understanding of DNA repair and the treatment of cancer resulting from DNA damage. He currently occupies the position of Senior Scientist and group leader of the DSB Repair Metabolism Laboratory at the Francis Crick Institute, London. He is also an honorary Professor at University College London.

FANCD2/FANCI-associated nuclease 1 (KIAA1018) is an enzyme that in humans is encoded by the FAN1 gene. It is a structure dependent endonuclease. It is thought to play an important role in the Fanconi Anemia (FA) pathway.

Andrew Neil James McKenzie is a molecular biologist and group leader in the Medical Research Council (MRC) Laboratory of Molecular Biology (LMB).

Lori Anne Passmore is a Canadian/British cryo electron microscopist and structural biologist who works at the Medical Research Council (MRC) Laboratory of Molecular Biology (LMB) at the University of Cambridge. She is known for her work on multiprotein complexes involved in gene expression and development of new supports for cryo-EM.

Helen Walden is an English structural biologist who received the Colworth medal from the Biochemical Society in 2015. She was awarded European Molecular Biology Organization (EMBO) membership in 2022. She is a Professor of Structural Biology at the University of Glasgow and has made significant contributions to the Ubiquitination field.
Agata Smogorzewska is a Polish-born scientist. She is an associate professor at Rockefeller University, heading the Laboratory of Genome Maintenance. Her work primarily focuses on DNA interstrand crosslink repair and the diseases resulting from deficiencies in this repair pathway, including Fanconi anemia and karyomegalic interstitial nephritis.
















