
Sialic acids are a class of alpha-keto acid sugars with a nine-carbon backbone. The term "sialic acid" was first introduced by Swedish biochemist Gunnar Blix in 1952. The most common member of this group is N-acetylneuraminic acid found in animals and some prokaryotes.

Neuraminic acid (5-amino-3,5-dideoxy-D-glycero-D-galacto-non-2-ulosonic acid) is an acidic (in particular ulosonic) amino sugar with a backbone formed by nine carbon atoms. Although 9-carbon sugars do not occur naturally, neuraminic acid may be regarded as a theoretical 9-carbon ketose in which the first link of the chain (the –CH2OH at position 1) is oxidised into a carboxyl group (–C(=O)OH), the hydroxyl group at position 3 is deoxidised (oxygen is removed from it), and the hydroxyl group at position 5 is substituted with an amino group (–NH2). Neuraminic acid may also be visualized as the product of an aldol-condensation of pyruvic acid and D-mannosamine (2-amino-2-deoxy-mannose).

N-Acetylmannosamine is a hexosamine monosaccharide. It is a neutral, stable naturally occurring compound. N-Acetylmannosamine is also known as N-Acetyl-D-mannosamine monohydrate,, N-Acetyl-D-mannosamine which can be abbreviated to ManNAc or, less commonly, NAM). ManNAc is the first committed biological precursor of N-acetylneuraminic acid. Sialic acids are the negatively charged, terminal monosaccharides of carbohydrate chains that are attached to glycoproteins and glycolipids (glycans).
In enzymology, a CMP-N-acetylneuraminate monooxygenase (EC 1.14.18.2) is an enzyme that catalyzes the chemical reaction
The enzyme 2-dehydro-3-deoxy-6-phosphogalactonate aldolase catalyzes the chemical reaction
The enzyme 2-dehydro-3-deoxy-D-pentonate aldolase catalyzes the chemical reaction
The enzyme 2-dehydro-3-deoxy-L-pentonate aldolase catalyzes the chemical reaction
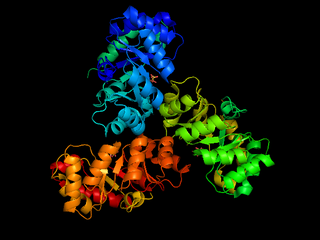
The enzyme 2-dehydro-3-deoxy-phosphogluconate aldolase, commonly known as KDPG aldolase, catalyzes the chemical reaction
The enzyme 4-hydroxy-2-oxoglutarate aldolase catalyzes the chemical reaction

Isocitrate lyase, or ICL, is an enzyme in the glyoxylate cycle that catalyzes the cleavage of isocitrate to succinate and glyoxylate. Together with malate synthase, it bypasses the two decarboxylation steps of the tricarboxylic acid cycle and is used by bacteria, fungi, and plants.

The enzyme methylisocitrate lyase catalyzes the chemical reaction
In enzymology, formate C-acetyltransferase is an enzyme. Pyruvate formate lyase is found in Escherichia coli and other organisms. It helps regulate anaerobic glucose metabolism. Using radical non-redox chemistry, it catalyzes the reversible conversion of pyruvate and coenzyme-A into formate and acetyl-CoA. The reaction occurs as follows:

In enzymology, a malate synthase (EC 2.3.3.9) is an enzyme that catalyzes the chemical reaction
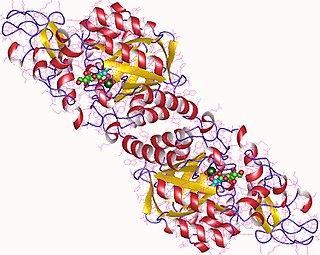
In enzymology, a N-acetylneuraminate synthase (EC 2.5.1.56) is an enzyme that catalyzes the chemical reaction
In enzymology, a N-acylneuraminate-9-phosphate synthase (EC 2.5.1.57) is an enzyme that catalyzes the chemical reaction
In enzymology, a beta-galactoside alpha-2,6-sialyltransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a lactosylceramide alpha-2,3-sialyltransferase is an enzyme that catalyzes the chemical reaction
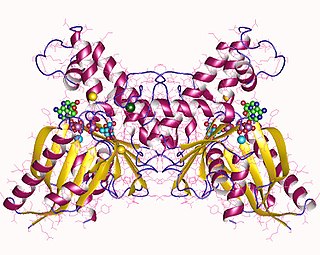
In enzymology, a N-acylmannosamine kinase is an enzyme that catalyzes the chemical reaction

In enzymology, a N-acylneuraminate cytidylyltransferase is an enzyme that catalyzes the chemical reaction
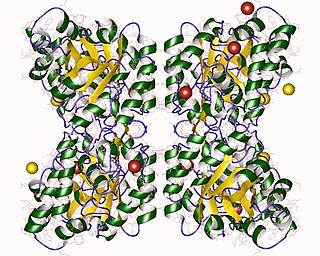
4-Hydroxy-tetrahydrodipicolinate synthase (EC 4.3.3.7, dihydrodipicolinate synthase, dihydropicolinate synthetase, dihydrodipicolinic acid synthase, L-aspartate-4-semialdehyde hydro-lyase (adding pyruvate and cyclizing), dapA (gene)) is an enzyme with the systematic name L-aspartate-4-semialdehyde hydro-lyase (adding pyruvate and cyclizing; (4S)-4-hydroxy-2,3,4,5-tetrahydro-(2S)-dipicolinate-forming). This enzyme catalyses the following chemical reaction












