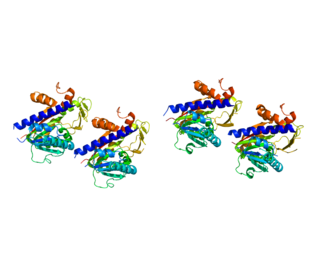Histone methyltransferases (HMT) are histone-modifying enzymes, that catalyze the transfer of one, two, or three methyl groups to lysine and arginine residues of histone proteins. The attachment of methyl groups occurs predominantly at specific lysine or arginine residues on histones H3 and H4. Two major types of histone methyltranferases exist, lysine-specific and arginine-specific. In both types of histone methyltransferases, S-Adenosyl methionine (SAM) serves as a cofactor and methyl donor group.
The genomic DNA of eukaryotes associates with histones to form chromatin. The level of chromatin compaction depends heavily on histone methylation and other post-translational modifications of histones. Histone methylation is a principal epigenetic modification of chromatin that determines gene expression, genomic stability, stem cell maturation, cell lineage development, genetic imprinting, DNA methylation, and cell mitosis.

Prenylation is the addition of hydrophobic molecules to a protein or a biomolecule. It is usually assumed that prenyl groups (3-methylbut-2-en-1-yl) facilitate attachment to cell membranes, similar to lipid anchors like the GPI anchor, though direct evidence of this has not been observed. Prenyl groups have been shown to be important for protein–protein binding through specialized prenyl-binding domains.

Methyltransferases are a large group of enzymes that all methylate their substrates but can be split into several subclasses based on their structural features. The most common class of methyltransferases is class I, all of which contain a Rossmann fold for binding S-Adenosyl methionine (SAM). Class II methyltransferases contain a SET domain, which are exemplified by SET domain histone methyltransferases, and class III methyltransferases, which are membrane associated. Methyltransferases can also be grouped as different types utilizing different substrates in methyl transfer reactions. These types include protein methyltransferases, DNA/RNA methyltransferases, natural product methyltransferases, and non-SAM dependent methyltransferases. SAM is the classical methyl donor for methyltransferases, however, examples of other methyl donors are seen in nature. The general mechanism for methyl transfer is a SN2-like nucleophilic attack where the methionine sulfur serves as the leaving group and the methyl group attached to it acts as the electrophile that transfers the methyl group to the enzyme substrate. SAM is converted to S-Adenosyl homocysteine (SAH) during this process. The breaking of the SAM-methyl bond and the formation of the substrate-methyl bond happen nearly simultaneously. These enzymatic reactions are found in many pathways and are implicated in genetic diseases, cancer, and metabolic diseases. Another type of methyl transfer is the radical S-Adenosyl methionine (SAM) which is the methylation of unactivated carbon atoms in primary metabolites, proteins, lipids, and RNA.
Protein L-isoaspartyl methyltransferase , also called S-adenosyl-L-methionine:protein-L-isoaspartate O-methyltransferase, is an enzyme which recognizes and catalyzes the repair of damaged L-isoaspartyl and D-aspartyl groups in proteins. It is a highly conserved enzyme which is present in nearly all eukaryotes, archaebacteria, and Gram-negative eubacteria.
In enzymology, a fatty-acid O-methyltransferase is an enzyme that catalyzes the chemical reaction
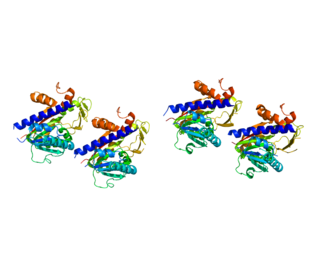
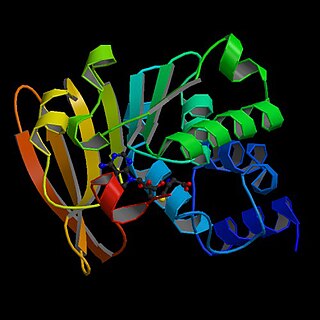
In enzymology, a mRNA (guanine-N7-)-methyltransferase also known as mRNA cap guanine-N7 methyltransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a mRNA (nucleoside-2'-O-)-methyltransferase is an enzyme that catalyzes the chemical reaction

In enzymology, a protein-glutamate O-methyltransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a sterol 24-C-methyltransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a trans-aconitate 2-methyltransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a tRNA (cytosine-5-)-methyltransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a tRNA (uracil-5-)-methyltransferase is an enzyme that catalyzes the chemical reaction
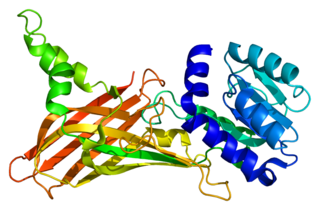
Protein arginine N-methyltransferase 1 is an enzyme that in humans is encoded by the PRMT1 gene. The HRMT1L2 gene encodes a protein arginine methyltransferase that functions as a histone methyltransferase specific for histone H4.
In enzymology, a mRNA guanylyltransferase is an enzyme that catalyzes the chemical reaction

Protein-L-isoaspartate(D-aspartate) O-methyltransferase is an enzyme that in humans is encoded by the PCMT1 gene.

Protein-S-isoprenylcysteine O-methyltransferase is an enzyme that in humans is encoded by the ICMT gene.
Steven G. Clarke, an American biochemist, is a director of the UCLA Molecular Biology Institute, a professor of chemistry and biochemistry at UCLA biochemistry department. Clarke heads a laboratory at UCLA's department of chemistry and biochemistry. Clarke is famous for his work on molecular damage and discoveries of novel molecular repair mechanisms.
Multisite-specific tRNA:(cytosine-C5)-methyltransferase is an enzyme with systematic name S-adenosyl-L-methionine:tRNA (cytosine-C5)-methyltransferase. This enzyme catalyses the following chemical reaction
(phosphatase 2A protein)-leucine-carboxy methyltransferase is an enzyme with systematic name S-adenosyl-L-methionine:(phosphatase 2A protein)-leucine O-methyltransferase. This enzyme catalyses the following chemical reaction
Protein methylation is a type of post-translational modification featuring the addition of methyl groups to proteins. It can occur on the nitrogen-containing side-chains of arginine and lysine, but also at the amino- and carboxy-termini of a number of different proteins. In biology, methyltransferases catalyze the methylation process, activated primarily by S-adenosylmethionine. Protein methylation has been most studied in histones, where the transfer of methyl groups from S-adenosyl methionine is catalyzed by histone methyltransferases. Histones that are methylated on certain residues can act epigenetically to repress or activate gene expression.