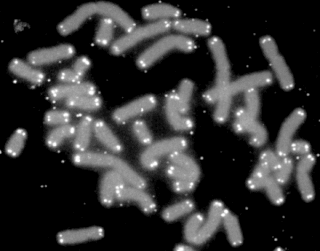
A telomere is a region of repetitive nucleotide sequences associated with specialized proteins at the ends of linear chromosomes. Telomeres are a widespread genetic feature most commonly found in eukaryotes. In most, if not all species possessing them, they protect the terminal regions of chromosomal DNA from progressive degradation and ensure the integrity of linear chromosomes by preventing DNA repair systems from mistaking the very ends of the DNA strand for a double-strand break.

Elizabeth Helen Blackburn, is an Australian-American Nobel laureate who is the former president of the Salk Institute for Biological Studies. In 1984, Blackburn co-discovered telomerase, the enzyme that replenishes the telomere, with Carol W. Greider. For this work, she was awarded the 2009 Nobel Prize in Physiology or Medicine, sharing it with Carol W. Greider and Jack W. Szostak, becoming the first Australian woman Nobel laureate.
Telomeric repeat-binding factor 2 is a protein that is present at telomeres throughout the cell cycle. It is also known as TERF2, TRF2, and TRBF2, and is encoded in humans by the TERF2 gene. It is a component of the shelterin nucleoprotein complex and a second negative regulator of telomere length, playing a key role in the protective activity of telomeres. It was first reported in 1997 in the lab of Titia de Lange, where a DNA sequence similar, but not identical, to TERF1 was discovered, with respect to the Myb-domain. De Lange isolated the new Myb-containing protein sequence and called it TERF2.

Telomeric repeat-binding factor 1 is a protein that in humans is encoded by the TERF1 gene.

Protection of telomeres protein 1 is a protein that in humans is encoded by the POT1 gene.
Tankyrase, also known as tankyrase 1, is an enzyme that in humans is encoded by the TNKS gene. It inhibits the binding of TERF1 to telomeric DNA. Tankyrase attracts substantial interest in cancer research through its interaction with AXIN1 and AXIN2, which are negative regulators of pro-oncogenic β-catenin signaling. Importantly, activity in the β-catenin destruction complex can be increased by tankyrase inhibitors and thus such inhibitors are a potential therapeutic option to reduce the growth of β-catenin-dependent cancers.

TERF1-interacting nuclear factor 2 is a protein that in humans is encoded by the TINF2 gene. TINF2 is a component of the shelterin protein complex found at the end of telomeres.

Adrenocortical dysplasia protein homolog is a protein that in humans is encoded by the ACD gene.

Telomeric repeat-binding factor 2-interacting protein 1 also known as repressor activator protein 1 (Rap1) is a protein that in humans is encoded by the TERF2IP gene.

Daniela Bargellini Rhodes FRS is an Italian structural and molecular biologist. She was a senior scientist at the Laboratory of Molecular Biology in Cambridge, England, where she worked, and later studied for her PhD under the supervision of Nobel laureate Aaron Klug. Continuing her work under the tutelage of Aaron Klug at Cambridge, she was appointed group leader in 1983, obtained tenure in 1987 and was promoted to senior scientist in 1994. Subsequently, she served as director of studies between 2003 and 2006. She has also been visiting professor at both "La Sapienza" in Rome, Italy and the Rockefeller University in NY, USA.
Virginia Zakian is the Harry C. Wiess Professor in the Life Sciences in the Department of Molecular Biology at Princeton University. She is the director of the Zakian Lab, which has done important research in topics such as telomere-binding protein, telomere recombination, and telomere position effects, at Princeton University. She is a fellow at the American Academy of Microbiology and the American Association for the Advancement of Science., and is an elected member of the National Academy of Sciences (2018). Zakian served as the chair of "Princeton's Task force on the Status of Women Faculty in the Natural Sciences and Engineering at Princeton" from 2001-2003, in 2003 Zakian became Princeton University's representative to Nine Universities, Gender Equity Analysis She was elected as a member of the American Academy of Arts and Sciences in 2019.
Xiaowei Zhuang is a Chinese-American biophysicist who is the David B. Arnold Jr. Professor of Science, Professor of Chemistry and Chemical Biology, and Professor of Physics at Harvard University, and an Investigator at the Howard Hughes Medical Institute. She is best known for her work in the development of Stochastic Optical Reconstruction Microscopy (STORM), a super-resolution fluorescence microscopy method, and the discoveries of novel cellular structures using STORM. She received a 2019 Breakthrough Prize in Life Sciences for developing super-resolution imaging techniques that get past the diffraction limits of traditional light microscopes, allowing scientists to visualize small structures within living cells. She was elected a Member of the American Philosophical Society in 2019 and was awarded a Vilcek Foundation Prize in Biomedical Science in 2020.
Telomere-binding proteins function to bind telomeric DNA in various species. In particular, telomere-binding protein refers to TTAGGG repeat binding factor-1 (TERF1) and TTAGGG repeat binding factor-2 (TERF2). Telomere sequences in humans are composed of TTAGGG sequences which provide protection and replication of chromosome ends to prevent degradation. Telomere-binding proteins can generate a T-loop to protect chromosome ends. TRFs are double-stranded proteins which are known to induce bending, looping, and pairing of DNA which aids in the formation of T-loops. They directly bind to TTAGGG repeat sequence in the DNA. There are also subtelomeric regions present for regulation. However, in humans, there are six subunits forming a complex known as shelterin.
Shelterin is a protein complex known to protect telomeres in many eukaryotes from DNA repair mechanisms, as well as to regulate telomerase activity. In mammals and other vertebrates, telomeric DNA consists of repeating double-stranded 5'-TTAGGG-3' (G-strand) sequences along with the 3'-AATCCC-5' (C-strand) complement, ending with a 50-400 nucleotide 3' (G-strand) overhang. Much of the final double-stranded portion of the telomere forms a T-loop (Telomere-loop) that is invaded by the 3' (G-strand) overhang to form a small D-loop (Displacement-loop).

Telomeric repeat–containing RNA (TERRA) is a long non-coding RNA transcribed from telomeres - repetitive nucleotide regions found on the ends of chromosomes that function to protect DNA from deterioration or fusion with neighboring chromosomes. TERRA has been shown to be ubiquitously expressed in almost all cell types containing linear chromosomes - including humans, mice, and yeasts. While the exact function of TERRA is still an active area of research, it is generally believed to play a role in regulating telomerase activity as well as maintaining the heterochromatic state at the ends of chromosomes. TERRA interaction with other associated telomeric proteins has also been shown to help regulate telomere integrity in a length-dependent manner.

María Antonia Blasco Marhuenda, known as María Blasco, is a Spanish molecular biologist. She is the current director of the Spanish National Cancer Research Centre.

Victoria Lundblad is an American geneticist whose work focuses on the genetic control of chromosome behavior in yeast. Many of her discoveries have concerned telomerase, the RNA-containing enzyme that completes the ends of chromosomes. She works at the Salk Institute in La Jolla, California.
Agata Smogorzewska is a Polish-born scientist. She is an associate professor at Rockefeller University, heading the Laboratory of Genome Maintenance. Her work primarily focuses on DNA interstrand crosslink repair and the diseases resulting from deficiencies in this repair pathway, including Fanconi anemia and karyomegalic interstitial nephritis.
Telomeres, the caps on the ends of eukaryotic chromosomes, play critical roles in cellular aging and cancer. An important facet to how telomeres function in these roles is their involvement in cell cycle regulation.

Jan Karlseder an Austrian molecular biologist, is the Chief Science Officer and a Senior Vice President at the Salk Institute for Biological Studies. He is also a professor in the Molecular and Cellular Biology Laboratory, the Director of the Paul F. Glenn Center for Biology of Aging Research and the holder of the Donald and Darlene Shiley Chair at the Salk Institute for Biological Studies.














