
CinnaGen is an Iran based biotechnology company that was founded in 1994 by four scientists. As of 2016, it manufactured biosimilar drugs, laboratory diagnostic reagents for in vitro use, and recombinant proteins, and exported to other countries in the Middle East and to countries in the Commonwealth of Independent States.

Novavax, Inc. is an American biotechnology company based in Gaithersburg, Maryland, that develops vaccines to counter serious infectious diseases. Prior to 2020, company scientists developed experimental vaccines for influenza and respiratory syncytial virus (RSV), as well as Ebola and other emerging infectious diseases. During 2020, the company redirected its efforts to focus on development and approval of its NVX-CoV2373 vaccine for COVID-19.

The Novavax COVID-19 vaccine, sold under the brand names Nuvaxovid and Covovax, among others, is a subunit COVID-19 vaccine developed by Novavax and the Coalition for Epidemic Preparedness Innovations (CEPI).

ZF2001, trade-named Zifivax or ZF-UZ-VAC-2001, is an adjuvanted protein subunit COVID-19 vaccine developed by Anhui Zhifei Longcom in collaboration with the Institute of Microbiology at the Chinese Academy of Sciences. The vaccine candidate is in Phase III trials with 29,000 participants in China, Ecuador, Malaysia, Pakistan, and Uzbekistan.

CoVLP was a COVID-19 vaccine developed by Medicago in Canada and GlaxoSmithKline (GSK). The product and Medicago, Inc. were owned by Mitsubishi who terminated the company and program in February 2023 due to high international market competition for COVID-19 vaccines.

Soberana 02 or Soberana 2, technical name FINLAY-FR-2, is a COVID-19 vaccine produced by the Finlay Institute, a Cuban epidemiological research institute. The vaccine is known as PastoCovac in Iran, where it has been developed in collaboration with the Pasteur Institute of Iran.
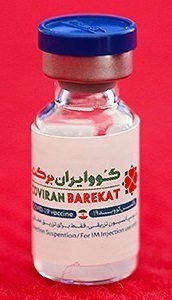
COVIran Barekat is a COVID-19 vaccine developed in Iran by Shifa Pharmed Industrial Group, a subsidiary of the Barkat Pharmaceutical Group. It is an inactivated virus-based vaccine. Iranian authorities have authorized its emergency use. This makes it the first locally developed COVID-19 vaccine to be approved for emergency use in the Middle East.

SCB-2019 is a protein subunit COVID-19 vaccine developed by Clover Biopharmaceuticals using an adjuvant from Dynavax technologies. Positive results of Phase I trials for the vaccine were published in The Lancet and the vaccine completed enrollment of 29,000 participants in Phase II/III trials in July 2021. In September 2021, SCB-2019 announced Phase III results showing 67% efficacy against all cases of COVID-19 and 79% efficacy against all cases of the Delta variant. Additionally, the vaccine was 84% effective against moderate cases and 100% effective against hospitalization.

The Sanofi–GSK COVID-19 vaccine sold under the brand name VidPrevtyn Beta, is a COVID-19 vaccine developed by Sanofi Pasteur and GSK.

Nanocovax is a Vietnamese COVID-19 vaccine candidate developed by Nanogen Pharmaceutical Biotechnology JSC. It is a subunit vaccine.

The MVC COVID-19 vaccine, designated MVC-COV1901 and also known as the Medigen COVID-19 vaccine, is a protein subunit COVID-19 vaccine developed by Medigen Vaccine Biologics Corporation in Taiwan, American company Dynavax Technologies, and the U.S. National Institutes of Health.

AWcorna, originally termed ARCoV and also known as the Walvax COVID-19 vaccine, is an mRNA COVID-19 vaccine developed by Walvax Biotechnology, Suzhou Abogen Biosciences, and the PLA Academy of Military Science. In contrast to other mRNA COVID vaccines, such as those by Pfizer-BioNtech and Moderna, this vaccine primarily targets the Sars-CoV-2 receptor-binding domain of the spike protein, rather than the entire spike protein. It is approved for Phase III trials in China, Mexico, Indonesia, and Nepal.

V-01 is a protein subunit COVID-19 vaccine candidate developed by a subsidiary of Livzon Pharmaceutical Group Inc.
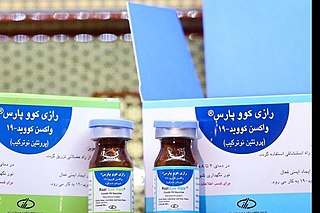
Razi Cov Pars is a COVID-19 vaccine developed by the Iranian Razi Vaccine and Serum Research Institute Razi Cov Pars is a covid-19 vaccine based on recombinant protein, which is being produced by Razi Vaccine and Serum Research Institute, Iran. This vaccine is the first injectable-intranasal recombinant protein corona vaccine.It's the second Iranian COVID-19 vaccine reaching human trials and is currently in phase III of clinical research during which it's compared to the Sinopharm BIBP vaccine.

FAKHRAVAC is a COVID-19 vaccine developed in Iran by the Organization of Defensive Innovation and Research, a subsidiary of Iran's Ministry of Defense. It is the third Iranian COVID-19 vaccine reaching clinical trials. It is currently in phase III. It received emergency use authorization in Iran on 9 September 2021.

COVID-19 vaccine clinical research uses clinical research to establish the characteristics of COVID-19 vaccines. These characteristics include efficacy, effectiveness, and safety. As of November 2022, 40 vaccines are authorized by at least one national regulatory authority for public use:

Noora is a COVID-19 vaccine candidate developed by Baqiyatallah University of Medical Sciences in collaboration with Plasma Darman Sarv Sepid Co. in Iran.

Soberana Plus, technical name FINLAY-FR-1A, is a COVID-19 candidate vaccine produced by the Finlay Institute, a Cuban epidemiological research institute.

S-268019-b is a protein subunit COVID-19 vaccine candidate developed by Shionogi.

LYB001 is a COVID-19 vaccine candidate developed by Yantai Patronus Biotech Co., Ltd.






