Background radiation is a measure of the level of ionizing radiation present in the environment at a particular location which is not due to deliberate introduction of radiation sources.

The hertz is the unit of frequency in the International System of Units (SI), often described as being equivalent to one event per second. The hertz is an SI derived unit whose formal expression in terms of SI base units is s−1, meaning that one hertz is one per second or the reciprocal of one second. It is used only in the case of periodic events. It is named after Heinrich Rudolf Hertz (1857–1894), the first person to provide conclusive proof of the existence of electromagnetic waves. For high frequencies, the unit is commonly expressed in multiples: kilohertz (kHz), megahertz (MHz), gigahertz (GHz), terahertz (THz).

Polonium is a chemical element; it has symbol Po and atomic number 84. A rare and highly radioactive metal with no stable isotopes, polonium is a chalcogen and chemically similar to selenium and tellurium, though its metallic character resembles that of its horizontal neighbors in the periodic table: thallium, lead, and bismuth. Due to the short half-life of all its isotopes, its natural occurrence is limited to tiny traces of the fleeting polonium-210 in uranium ores, as it is the penultimate daughter of natural uranium-238. Though longer-lived isotopes exist, such as the 124 years half-life of polonium-209, they are much more difficult to produce. Today, polonium is usually produced in milligram quantities by the neutron irradiation of bismuth. Due to its intense radioactivity, which results in the radiolysis of chemical bonds and radioactive self-heating, its chemistry has mostly been investigated on the trace scale only.

Radium is a chemical element; it has symbol Ra and atomic number 88. It is the sixth element in group 2 of the periodic table, also known as the alkaline earth metals. Pure radium is silvery-white, but it readily reacts with nitrogen (rather than oxygen) upon exposure to air, forming a black surface layer of radium nitride (Ra3N2). All isotopes of radium are radioactive, the most stable isotope being radium-226 with a half-life of 1,600 years. When radium decays, it emits ionizing radiation as a by-product, which can excite fluorescent chemicals and cause radioluminescence. For this property, it was widely used in self-luminous paints following its discovery. Of the radioactive elements that occur in quantity, radium is considered particularly toxic, and it is carcinogenic due to the radioactivity of both it and its immediate decay product radon as well as its tendency to accumulate in the bones.

The becquerel is the unit of radioactivity in the International System of Units (SI). One becquerel is defined as an activity of one per second, on average, for aperiodic activity events referred to a radionuclide. For applications relating to human health this is a small quantity, and SI multiples of the unit are commonly used.
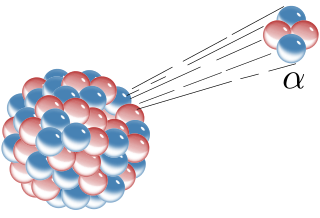
Radioactive decay is the process by which an unstable atomic nucleus loses energy by radiation. A material containing unstable nuclei is considered radioactive. Three of the most common types of decay are alpha, beta, and gamma decay. The weak force is the mechanism that is responsible for beta decay, while the other two are governed by the electromagnetic and nuclear forces.
A period 7 element is one of the chemical elements in the seventh row of the periodic table of the chemical elements. The periodic table is laid out in rows to illustrate recurring (periodic) trends in the chemical behavior of the elements as their atomic number increases: a new row is begun when chemical behavior begins to repeat, meaning that elements with similar behavior fall into the same vertical columns. The seventh period contains 32 elements, tied for the most with period 6, beginning with francium and ending with oganesson, the heaviest element currently discovered. As a rule, period 7 elements fill their 7s shells first, then their 5f, 6d, and 7p shells in that order, but there are exceptions, such as uranium.

Specific activity is the activity per unit mass of a radionuclide and is a physical property of that radionuclide. It is usually given in units of becquerel per kilogram (Bq/kg), but another commonly used unit of specific activity is the curie per gram (Ci/g).
The measurement of ionizing radiation is sometimes expressed as being a rate of counts per unit time as registered by a radiation monitoring instrument, for which counts per minute (cpm) and counts per second (cps) are commonly used quantities.
The rad is a unit of absorbed radiation dose, defined as 1 rad = 0.01 Gy = 0.01 J/kg. It was originally defined in CGS units in 1953 as the dose causing 100 ergs of energy to be absorbed by one gram of matter. The material absorbing the radiation can be human tissue, air, water, or any other substance.

Caesium-137, cesium-137 (US), or radiocaesium, is a radioactive isotope of caesium that is formed as one of the more common fission products by the nuclear fission of uranium-235 and other fissionable isotopes in nuclear reactors and nuclear weapons. Trace quantities also originate from spontaneous fission of uranium-238. It is among the most problematic of the short-to-medium-lifetime fission products. Caesium-137 has a relatively low boiling point of 671 °C (1,240 °F) and easily becomes volatile when released suddenly at high temperature, as in the case of the Chernobyl nuclear accident and with atomic explosions, and can travel very long distances in the air. After being deposited onto the soil as radioactive fallout, it moves and spreads easily in the environment because of the high water solubility of caesium's most common chemical compounds, which are salts. Caesium-137 was discovered by Glenn T. Seaborg and Margaret Melhase.
The rutherford is a non-SI unit of radioactive decay. It is defined as the activity of a quantity of radioactive material in which one million nuclei decay per second. It is therefore equivalent to one megabecquerel, and one becquerel equals one microrutherford. One rutherford is equivalent to 2.702×10−5 curie, or 37000 rutherfords for one curie.
In health physics, whole-body counting refers to the measurement of radioactivity within the human body. The technique is primarily applicable to radioactive material that emits gamma rays. Alpha particle decays can also be detected indirectly by their coincident gamma radiation. In certain circumstances, beta emitters can be measured, but with degraded sensitivity. The instrument used is normally referred to as a whole body counter.

Uranium tiles have been used in the ceramics industry for many centuries, as uranium oxide makes an excellent ceramic glaze, and is reasonably abundant. In addition to its medical usage, radium was used in the 1920s and 1930s for making watch, clock and aircraft dials. Because it takes approximately three metric tons of uranium to extract 1 gram of radium, prodigious quantities of uranium were mined to sustain this new industry. The uranium ore itself was considered a waste product and taking advantage of this newly abundant resource, the tile and pottery industry had a relatively inexpensive and abundant source of glazing material. Vibrant colors of orange, yellow, red, green, blue, black, mauve, etc. were produced, and some 25% of all houses and apartments constructed during that period used bathroom or kitchen tiles that had been glazed with uranium. These can now be detected by a Geiger counter that detects the beta radiation emitted by uranium's decay chain.
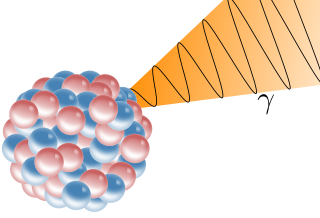
A gamma ray, also known as gamma radiation (symbol
γ
), is a penetrating form of electromagnetic radiation arising from the radioactive decay of atomic nuclei. It consists of the shortest wavelength electromagnetic waves, typically shorter than those of X-rays. With frequencies above 30 exahertz (3×1019 Hz) and wavelengths less than 10 picometers (1×10−11 m), gamma ray photons have the highest photon energy of any form of electromagnetic radiation. Paul Villard, a French chemist and physicist, discovered gamma radiation in 1900 while studying radiation emitted by radium. In 1903, Ernest Rutherford named this radiation gamma rays based on their relatively strong penetration of matter; in 1900, he had already named two less penetrating types of decay radiation (discovered by Henri Becquerel) alpha rays and beta rays in ascending order of penetrating power.

Alpha particles, also called alpha rays or alpha radiation, consist of two protons and two neutrons bound together into a particle identical to a helium-4 nucleus. They are generally produced in the process of alpha decay but may also be produced in other ways. Alpha particles are named after the first letter in the Greek alphabet, α. The symbol for the alpha particle is α or α2+. Because they are identical to helium nuclei, they are also sometimes written as He2+ or 4
2He2+ indicating a helium ion with a +2 charge (missing its two electrons). Once the ion gains electrons from its environment, the alpha particle becomes a normal (electrically neutral) helium atom 4
2He.

The roentgen or röntgen is a legacy unit of measurement for the exposure of X-rays and gamma rays, and is defined as the electric charge freed by such radiation in a specified volume of air divided by the mass of that air . In 1928, it was adopted as the first international measurement quantity for ionizing radiation to be defined for radiation protection, as it was then the most easily replicated method of measuring air ionization by using ion chambers. It is named after the German physicist Wilhelm Röntgen, who discovered X-rays and was awarded the first Nobel Prize in Physics for the discovery.

Banana equivalent dose (BED) is an informal unit of measurement of ionizing radiation exposure, intended as a general educational example to compare a dose of radioactivity to the dose one is exposed to by eating one average-sized banana. Bananas contain naturally occurring radioactive isotopes, particularly potassium-40 (40K), one of several naturally occurring isotopes of potassium. One BED is often correlated to 10−7 sievert ; however, in practice, this dose is not cumulative, as the potassium in foods is excreted in urine to maintain homeostasis. The BED is only meant as an educational exercise and is not a formally adopted dose measurement.

A radioactive source is a known quantity of a radionuclide which emits ionizing radiation, typically one or more of the radiation types gamma rays, alpha particles, beta particles, and neutron radiation.
Since its introduction in 1960, the base units for the International system of units, known as SI, have changed several times. Tables in this article summarize those changes.











