Related Research Articles

Electrochemistry is the branch of physical chemistry concerned with the relationship between electrical potential difference and identifiable chemical change. These reactions involve electrons moving via an electronically-conducting phase between electrodes separated by an ionically conducting and electronically insulating electrolyte.

An electrode is an electrical conductor used to make contact with a nonmetallic part of a circuit. Electrodes are essential parts of batteries that can consist of a variety of materials depending on the type of battery.
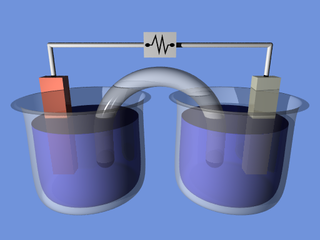
An electrochemical cell is a device that generates electrical energy from chemical reactions. Electrical energy can also be applied to these cells to cause chemical reactions to occur. Electrochemical cells that generate an electric current are called voltaic or galvanic cells and those that generate chemical reactions, via electrolysis for example, are called electrolytic cells.

The voltaic pile was the first electrical battery that could continuously provide an electric current to a circuit. It was invented by Italian chemist Alessandro Volta, who published his experiments in 1799. Its invention can be traced back to an argument between Volta and Luigi Galvani, Volta's fellow Italian scientist who had conducted experiments on frogs' legs. The voltaic pile then enabled a rapid series of other discoveries including the electrical decomposition (electrolysis) of water into oxygen and hydrogen by William Nicholson and Anthony Carlisle (1800) and the discovery or isolation of the chemical elements sodium (1807), potassium (1807), calcium (1808), boron (1808), barium (1808), strontium (1808), and magnesium (1808) by Humphry Davy.

In chemistry and manufacturing, electrolysis is a technique that uses direct electric current (DC) to drive an otherwise non-spontaneous chemical reaction. Electrolysis is commercially important as a stage in the separation of elements from naturally occurring sources such as ores using an electrolytic cell. The voltage that is needed for electrolysis to occur is called the decomposition potential. The word "lysis" means to separate or break, so in terms, electrolysis would mean "breakdown via electricity".
In electrochemistry, electrode potential is the voltage of a galvanic cell built from a standard reference electrode and another electrode to be characterized. By convention, the reference electrode is the standard hydrogen electrode (SHE). It is defined to have a potential of zero volts. It may also be defined as the potential difference between the charged metallic rods and salt solution.
In chemistry, a half reaction is either the oxidation or reduction reaction component of a redox reaction. A half reaction is obtained by considering the change in oxidation states of individual substances involved in the redox reaction. Often, the concept of half reactions is used to describe what occurs in an electrochemical cell, such as a Galvanic cell battery. Half reactions can be written to describe both the metal undergoing oxidation and the metal undergoing reduction.

Electroplating, also known as electrochemical deposition or electrodeposition, is a process for producing a metal coating on a solid substrate through the reduction of cations of that metal by means of a direct electric current. The part to be coated acts as the cathode of an electrolytic cell; the electrolyte is a solution of a salt of the metal to be coated; and the anode is usually either a block of that metal, or of some inert conductive material. The current is provided by an external power supply.

Redox is a type of chemical reaction in which the oxidation states of a reactant change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a decrease in the oxidation state.
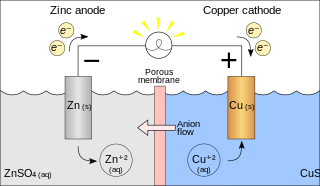
A galvanic cell or voltaic cell, named after the scientists Luigi Galvani and Alessandro Volta, respectively, is an electrochemical cell in which an electric current is generated from spontaneous oxidation–reduction reactions. A common apparatus generally consists of two different metals, each immersed in separate beakers containing their respective metal ions in solution that are connected by a salt bridge or separated by a porous membrane.
In electrochemistry, standard electrode potential, or , is a measure of the reducing power of any element or compound. The IUPAC "Gold Book" defines it as; "the value of the standard emf of a cell in which molecular hydrogen under standard pressure is oxidized to solvated protons at the left-hand electrode".

The alkaline fuel cell (AFC), also known as the Bacon fuel cell after its British inventor, Francis Thomas Bacon, is one of the most developed fuel cell technologies. Alkaline fuel cells consume hydrogen and pure oxygen, to produce potable water, heat, and electricity. They are among the most efficient fuel cells, having the potential to reach 70%.

The copper–copper(II) sulfate electrode is a reference electrode of the first kind, based on the redox reaction with participation of the metal (copper) and its salt, copper(II) sulfate. It is used for measuring electrode potential and is the most commonly used reference electrode for testing cathodic protection corrosion control systems. The corresponding equation can be presented as follow:
A silver chloride electrode is a type of reference electrode, commonly used in electrochemical measurements. For environmental reasons it has widely replaced the saturated calomel electrode. For example, it is usually the internal reference electrode in pH meters and it is often used as reference in reduction potential measurements. As an example of the latter, the silver chloride electrode is the most commonly used reference electrode for testing cathodic protection corrosion control systems in sea water environments.

The Daniell cell is a type of electrochemical cell invented in 1836 by John Frederic Daniell, a British chemist and meteorologist, and consists of a copper pot filled with a copper (II) sulfate solution, in which is immersed an unglazed earthenware container filled with sulfuric acid and a zinc electrode. He was searching for a way to eliminate the hydrogen bubble problem found in the voltaic pile, and his solution was to use a second electrolyte to consume the hydrogen produced by the first. Zinc sulfate may be substituted for the sulfuric acid. The Daniell cell was a great improvement over the existing technology used in the early days of battery development. A later variant of the Daniell cell called the gravity cell or crowfoot cell was invented in the 1860s by a Frenchman named Callaud and became a popular choice for electrical telegraphy.

A zinc–carbon battery (or carbon zinc battery in U.S. English) is a dry cell primary battery that provides direct electric current from the electrochemical reaction between zinc (Zn) and manganese dioxide (MnO2) in the presence of an ammonium chloride (NH4Cl) electrolyte. It produces a voltage of about 1.5 volts between the zinc anode, which is typically constructed as a cylindrical container for the battery cell, and a carbon rod surrounded by a compound with a higher Standard electrode potential (positive polarity), known as the cathode, that collects the current from the manganese dioxide electrode. The name "zinc-carbon" is slightly misleading as it implies that carbon is acting as the oxidizing agent rather than the manganese dioxide.
Cell notation or cell representation in chemistry is a shorthand method of expressing a reaction in an electrochemical cell.

The Leclanché cell is a battery invented and patented by the French scientist Georges Leclanché in 1866. The battery contained a conducting solution (electrolyte) of ammonium chloride, a cathode of carbon, a depolarizer of manganese dioxide (oxidizer), and an anode of zinc (reductant). The chemistry of this cell was later successfully adapted to manufacture a dry cell.

The penny battery is a voltaic pile which uses various coinage as the metal disks (pennies) of a traditional voltaic pile. The coins are stacked with pieces of electrolyte soaked paper in between. The penny battery experiment is common during electrochemistry units in an educational setting.
In chemistry, ion transport number, also called the transference number, is the fraction of the total electric current carried in an electrolyte by a given ionic species i:
References
- ↑ "an introduction to redox equilibria and electrode potentials". www.chemguide.co.uk. Retrieved 2024-02-11.
- ↑ "Untitled Document". old.iupac.org. Retrieved 2024-02-11.
