
The design of experiments, also known as experiment design or experimental design, is the design of any task that aims to describe and explain the variation of information under conditions that are hypothesized to reflect the variation. The term is generally associated with experiments in which the design introduces conditions that directly affect the variation, but may also refer to the design of quasi-experiments, in which natural conditions that influence the variation are selected for observation.
In statistics, a unit is one member of a set of entities being studied. It is the main source for the mathematical abstraction of a "random variable". Common examples of a unit would be a single person, animal, plant, manufactured item, or country that belongs to a larger collection of such entities being studied.

An experiment is a procedure carried out to support or refute a hypothesis, or determine the efficacy or likelihood of something previously untried. Experiments provide insight into cause-and-effect by demonstrating what outcome occurs when a particular factor is manipulated. Experiments vary greatly in goal and scale but always rely on repeatable procedure and logical analysis of the results. There also exist natural experimental studies.

A randomized controlled trial is a form of scientific experiment used to control factors not under direct experimental control. Examples of RCTs are clinical trials that compare the effects of drugs, surgical techniques, medical devices, diagnostic procedures or other medical treatments.
In statistics, the power of a binary hypothesis test is the probability that the test correctly rejects the null hypothesis when a specific alternative hypothesis is true. It is commonly denoted by , and represents the chances of a true positive detection conditional on the actual existence of an effect to detect. Statistical power ranges from 0 to 1, and as the power of a test increases, the probability of making a type II error by wrongly failing to reject the null hypothesis decreases.

Field experiments are experiments carried out outside of laboratory settings.
In the design of experiments, hypotheses are applied to experimental units in a treatment group. In comparative experiments, members of a control group receive a standard treatment, a placebo, or no treatment at all. There may be more than one treatment group, more than one control group, or both.
In the statistical theory of the design of experiments, blocking is the arranging of experimental units that are similar to one another in groups (blocks). Blocking can be used to tackle the problem of pseudoreplication.
Clinical study design is the formulation of trials and experiments, as well as observational studies in medical, clinical and other types of research involving human beings. The goal of a clinical study is to assess the safety, efficacy, and / or the mechanism of action of an investigational medicinal product (IMP) or procedure, or new drug or device that is in development, but potentially not yet approved by a health authority. It can also be to investigate a drug, device or procedure that has already been approved but is still in need of further investigation, typically with respect to long-term effects or cost-effectiveness.

In causal inference, a confounder is a variable that influences both the dependent variable and independent variable, causing a spurious association. Confounding is a causal concept, and as such, cannot be described in terms of correlations or associations. The existence of confounders is an important quantitative explanation why correlation does not imply causation. Some notations are explicitly designed to identify the existence, possible existence, or non-existence of confounders in causal relationships between elements of a system.

In science, randomized experiments are the experiments that allow the greatest reliability and validity of statistical estimates of treatment effects. Randomization-based inference is especially important in experimental design and in survey sampling.

In fields such as epidemiology, social sciences, psychology and statistics, an observational study draws inferences from a sample to a population where the independent variable is not under the control of the researcher because of ethical concerns or logistical constraints. One common observational study is about the possible effect of a treatment on subjects, where the assignment of subjects into a treated group versus a control group is outside the control of the investigator. This is in contrast with experiments, such as randomized controlled trials, where each subject is randomly assigned to a treated group or a control group. Observational studies, for lacking an assignment mechanism, naturally present difficulties for inferential analysis.

A quasi-experiment is an empirical interventional study used to estimate the causal impact of an intervention on target population without random assignment. Quasi-experimental research shares similarities with the traditional experimental design or randomized controlled trial, but it specifically lacks the element of random assignment to treatment or control. Instead, quasi-experimental designs typically allow the researcher to control the assignment to the treatment condition, but using some criterion other than random assignment.
Repeated measures design is a research design that involves multiple measures of the same variable taken on the same or matched subjects either under different conditions or over two or more time periods. For instance, repeated measurements are collected in a longitudinal study in which change over time is assessed.
A glossary of terms used in experimental research.
In medical research, a dynamic treatment regime (DTR), adaptive intervention, or adaptive treatment strategy is a set of rules for choosing effective treatments for individual patients. Historically, medical research and the practice of medicine tended to rely on an acute care model for the treatment of all medical problems, including chronic illness. Treatment choices made for a particular patient under a dynamic regime are based on that individual's characteristics and history, with the goal of optimizing his or her long-term clinical outcome. A dynamic treatment regime is analogous to a policy in the field of reinforcement learning, and analogous to a controller in control theory. While most work on dynamic treatment regimes has been done in the context of medicine, the same ideas apply to time-varying policies in other fields, such as education, marketing, and economics.
An N of 1 trial (N=1) is a clinical trial in which a single patient is the entire trial, a single case study. A trial in which random allocation can be used to determine the order in which an experimental and a control intervention are given to a patient is an N of 1 randomized controlled trial. The order of experimental and control interventions can also be fixed by the researcher.
The phases of clinical research are the stages in which scientists conduct experiments with a health intervention to obtain sufficient evidence for a process considered effective as a medical treatment. For drug development, the clinical phases start with testing for drug safety in a few human subjects, then expand to many study participants to determine if the treatment is effective. Clinical research is conducted on drug candidates, vaccine candidates, new medical devices, and new diagnostic assays.
In medicine, a stepped-wedge trial is a type of randomised controlled trial (RCT). An RCT is a scientific experiment that is designed to reduce bias when testing a new medical treatment, a social intervention, or another testable hypothesis.
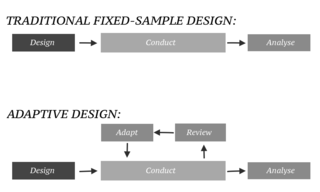
In an adaptive design of a clinical trial, the parameters and conduct of the trial for a candidate drug or vaccine may be changed based on an interim analysis. Adaptive design typically involves advanced statistics to interpret a clinical trial endpoint. This is in contrast to traditional single-arm clinical trials or randomized clinical trials (RCTs) that are static in their protocol and do not modify any parameters until the trial is completed. The adaptation process takes place at certain points in the trial, prescribed in the trial protocol. Importantly, this trial protocol is set before the trial begins with the adaptation schedule and processes specified. Adaptions may include modifications to: dosage, sample size, drug undergoing trial, patient selection criteria and/or "cocktail" mix. The PANDA provides not only a summary of different adaptive designs, but also comprehensive information on adaptive design planning, conduct, analysis and reporting.







