
Nickel is a chemical element; it has symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel is a hard and ductile transition metal. Pure nickel is chemically reactive, but large pieces are slow to react with air under standard conditions because a passivation layer of nickel oxide forms on the surface that prevents further corrosion. Even so, pure native nickel is found in Earth's crust only in tiny amounts, usually in ultramafic rocks, and in the interiors of larger nickel–iron meteorites that were not exposed to oxygen when outside Earth's atmosphere.

Vanadium is a chemical element; it has symbol V and atomic number 23. It is a hard, silvery-grey, malleable transition metal. The elemental metal is rarely found in nature, but once isolated artificially, the formation of an oxide layer (passivation) somewhat stabilizes the free metal against further oxidation.
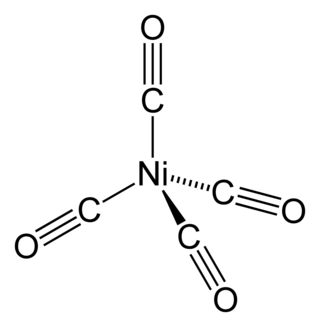
Nickel carbonyl (IUPAC name: tetracarbonylnickel) is a nickel(0) organometallic compound with the formula Ni(CO)4. This colorless liquid is the principal carbonyl of nickel. It is an intermediate in the Mond process for producing very high-purity nickel and a reagent in organometallic chemistry, although the Mond Process has fallen out of common usage due to the health hazards in working with the compound. Nickel carbonyl is one of the most dangerous substances yet encountered in nickel chemistry due to its very high toxicity, compounded with high volatility and rapid skin absorption.

Group 5 is a group of elements in the periodic table. Group 5 contains vanadium (V), niobium (Nb), tantalum (Ta) and dubnium (Db). This group lies in the d-block of the periodic table. This group is sometimes called the vanadium group or vanadium family after its lightest member; however, the group itself has not acquired a trivial name because it belongs to the broader grouping of the transition metals.

Sudan I is an organic compound, typically classified as an azo dye. It is an intensely orange-red solid that is added to colourise waxes, oils, petrol, solvents, and polishes. Sudan I has also been adopted for colouring various foodstuffs, especially curry powder and chili powder, although the use of Sudan I in foods is now banned in many countries, because Sudan I, Sudan III, and Sudan IV have been classified as category 3 carcinogens by the International Agency for Research on Cancer. Sudan I is still used in some orange-coloured smoke formulations and as a colouring for cotton refuse used in chemistry experiments.

Nickel(II) chloride (or just nickel chloride) is the chemical compound NiCl2. The anhydrous salt is yellow, but the more familiar hydrate NiCl2·6H2O is green. Nickel(II) chloride, in various forms, is the most important source of nickel for chemical synthesis. The nickel chlorides are deliquescent, absorbing moisture from the air to form a solution. Nickel salts have been shown to be carcinogenic to the lungs and nasal passages in cases of long-term inhalation exposure.

1,4-Dioxane is a heterocyclic organic compound, classified as an ether. It is a colorless liquid with a faint sweet odor similar to that of diethyl ether. The compound is often called simply dioxane because the other dioxane isomers are rarely encountered.

Ethylbenzene is an organic compound with the formula C6H5CH2CH3. It is a highly flammable, colorless liquid with an odor similar to that of gasoline. This monocyclic aromatic hydrocarbon is important in the petrochemical industry as a reaction intermediate in the production of styrene, the precursor to polystyrene, a common plastic material. In 2012, more than 99% of ethylbenzene produced was consumed in the production of styrene.
Indium gallium arsenide (InGaAs) is a ternary alloy of indium arsenide (InAs) and gallium arsenide (GaAs). Indium and gallium are group III elements of the periodic table while arsenic is a group V element. Alloys made of these chemical groups are referred to as "III-V" compounds. InGaAs has properties intermediate between those of GaAs and InAs. InGaAs is a room-temperature semiconductor with applications in electronics and photonics.

Benzyl butyl phthalate (BBP) is an organic compound historically used a plasticizer, but which has now been largely phased out due to health concerns. It is a phthalate ester of containing benzyl alcohol, and n-butanol tail groups. Like most phthalates, BBP is non-volatile and remains liquid over a wide range of temperatures. It was mostly used as a plasticizer for PVC, but was also a common plasticizer for PVCA and PVB.

Nickel(II) sulfate, or just nickel sulfate, usually refers to the inorganic compound with the formula NiSO4(H2O)6. This highly soluble blue green coloured salt is a common source of the Ni2+ ion for electroplating.
Dense inert metal explosive (DIME) is an experimental type of explosive that has a relatively small but effective blast radius. It is manufactured by producing a homogeneous mixture of an explosive material and small particles of a chemically inert material such as tungsten. It is intended to limit the effective distance of the explosion, to avoid collateral damage in warfare.

In molecular genetics, a DNA adduct is a segment of DNA bound to a cancer-causing chemical. This process could lead to the development of cancerous cells, or carcinogenesis. DNA adducts in scientific experiments are used as biomarkers of exposure. They are especially useful in quantifying an organism's exposure to a carcinogen. The presence of such an adduct indicates prior exposure to a potential carcinogen, but it does not necessarily indicate the presence of cancer in the subject animal.

o-Toluidine (ortho-toluidine) is an organic compound with the chemical formula CH3C6H4NH2. It is the most important of the three isomeric toluidines. It is a colorless liquid although commercial samples are often yellowish. It is a precursor to the herbicides metolachlor and acetochlor.

1-Nitropyrene is a by-product of combustion and is the predominant nitrated polycyclic aromatic hydrocarbon (pyrene) emitted in a diesel engine. 1-Nitropyrene is listed as an IARC Group 2B carcinogen, indicating it is possibly carcinogenic to humans.

Riddelliine is a chemical compound classified as a pyrrolizidine alkaloid. It was first isolated from Senecio riddellii and is also found in a variety of plants including Jacobaea vulgaris, Senecio vulgaris, and others plants in the genus Senecio.

Diepoxybutane is an epoxide which is a colorless liquid at room temperature. It is therefore highly reactive, more than other ethers. An epoxide is a cyclic ether that contains a three atom ring that comes close to an equilateral triangle. The primary structure of an epoxide contains two carbon atoms and a hydrocarbon attached to an oxygen atom. It polymerizes in the presence of catalysts or when heated. It’s hydrophilic, very flammable and easily ignited by heat or sparks.
Methacrylonitrile, MeAN in short, is a chemical compound that is an unsaturated aliphatic nitrile, widely used in the preparation of homopolymers, copolymers, elastomers, and plastics and as a chemical intermediate in the preparation of acids, amides, amines, esters, and other nitriles. MeAN is also used as a replacement for acrylonitrile in the manufacture of an acrylonitrile/butadiene/styrene-like polymer. It is a clear and colorless liquid, that has a bitter almond smell.

2-Mercaptobenzothiazole is an organosulfur compound with the formula C6H4(NH)SC=S. A white solid, it is used in the sulfur vulcanization of rubber.

Glycidamide is an organic compound with the formula H2NC(O)C2H3O. It is a colorless oil. Structurally, it contains adjacent amides and epoxide functional groups. It is a bioactive, potentially toxic or even carcinogenic metabolite of acrylonitrile and acrylamide. It is a chiral molecule.





















