
Threonine is an amino acid that is used in the biosynthesis of proteins. It contains an α-amino group, a carboxyl group, and a side chain containing a hydroxyl group, making it a polar, uncharged amino acid. It is essential in humans, meaning the body cannot synthesize it: it must be obtained from the diet. Threonine is synthesized from aspartate in bacteria such as E. coli. It is encoded by all the codons starting AC.

Aconitase is an enzyme that catalyses the stereo-specific isomerization of citrate to isocitrate via cis-aconitate in the tricarboxylic acid cycle, a non-redox-active process.
Propionyl-CoA is a coenzyme A derivative of propionic acid. It is composed of a 24 total carbon chain and its production and metabolic fate depend on which organism it is present in. Several different pathways can lead to its production, such as through the catabolism of specific amino acids or the oxidation of odd-chain fatty acids. It later can be broken down by propionyl-CoA carboxylase or through the methylcitrate cycle. In different organisms, however, propionyl-CoA can be sequestered into controlled regions, to alleviate its potential toxicity through accumulation. Genetic deficiencies regarding the production and breakdown of propionyl-CoA also have great clinical and human significance.
In enzymology, a 3-hydroxy-2-methylbutyryl-CoA dehydrogenase (EC 1.1.1.178) is an enzyme that catalyzes the chemical reaction
In enzymology, a trans-aconitate 2-methyltransferase is an enzyme that catalyzes the chemical reaction
In enzymology, a trans-aconitate 3-methyltransferase is an enzyme that catalyzes the chemical reaction
In enzymology, an aconitate Δ-isomerase is an enzyme that catalyzes the chemical reaction

The enzyme methylisocitrate lyase catalyzes the chemical reaction
The enzyme 2-methylcitrate dehydratase (EC 4.2.1.79) catalyzes the chemical reaction
The enzyme 2-methylisocitrate dehydratase (EC 4.2.1.99) catalyzes the chemical reaction
In enzymology, a citrate dehydratase (EC 4.2.1.4) is an enzyme that catalyzes the chemical reaction
The enzyme dihydroxy-acid dehydratase (EC 4.2.1.9) catalyzes the chemical reaction
The enzyme ectoine synthase (EC ) catalyzes the chemical reaction
In enzymology, a 2-methylcitrate synthase (EC 2.3.3.5) is an enzyme that catalyzes the chemical reaction
Decylcitrate synthase (EC 2.3.3.2) is an enzyme that catalyzes the chemical reaction in enzymology.
3-Isopropylmalate dehydrogenase (EC 1.1.1.85) is an enzyme that is a part of the isopropylmalate dehydrogenase family, which catalyzes the chemical reactions:

In molecular biology, the citrate synthase family of proteins includes the enzymes citrate synthase EC 2.3.3.1, and the related enzymes 2-methylcitrate synthase EC 2.3.3.5 and ATP citrate lyase EC 2.3.3.8.
Linalool dehydratase (EC 4.2.1.127, linalool hydro-lyase (myrcene-forming)) is an enzyme with systematic name (3S)-linalool hydro-lyase (myrcene-forming). This enzyme catalyses the following chemical reaction
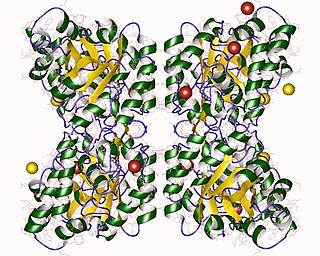
4-Hydroxy-tetrahydrodipicolinate synthase (EC 4.3.3.7, dihydrodipicolinate synthase, dihydropicolinate synthetase, dihydrodipicolinic acid synthase, L-aspartate-4-semialdehyde hydro-lyase (adding pyruvate and cyclizing), dapA (gene)) is an enzyme with the systematic name L-aspartate-4-semialdehyde hydro-lyase (adding pyruvate and cyclizing; (4S)-4-hydroxy-2,3,4,5-tetrahydro-(2S)-dipicolinate-forming). This enzyme catalyses the following chemical reaction





