| Uroporphyrinogen-III synthase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 Uroporphyrinogen-III synthase monomer, Thermus thermophilus | |||||||||
| Identifiers | |||||||||
| EC no. | 4.2.1.75 | ||||||||
| CAS no. | 37340-55-9 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
| Uroporphyrinogen III synthase | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | UROS | ||||||
| NCBI gene | 7390 | ||||||
| HGNC | 12592 | ||||||
| OMIM | 606938 | ||||||
| RefSeq | NM_000375 | ||||||
| UniProt | P10746 | ||||||
| Other data | |||||||
| EC number | 4.2.1.75 | ||||||
| Locus | Chr. 10 q25.2-26.3 | ||||||
| |||||||
| Uroporphyrinogen-III synthase HemD | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 crystal structure of uroporphyrinogen iii synthase from an extremely thermophilic bacterium thermus thermophilus hb8 (wild type, native, form-2 crystal) | |||||||||
| Identifiers | |||||||||
| Symbol | HEM4 | ||||||||
| Pfam | PF02602 | ||||||||
| InterPro | IPR003754 | ||||||||
| SCOP2 | 1jr2 / SCOPe / SUPFAM | ||||||||
| |||||||||
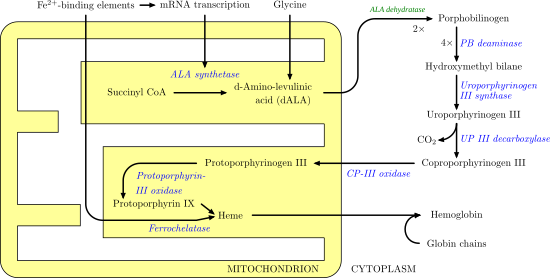
Uroporphyrinogen III synthase (EC 4.2.1.75) is an enzyme involved in the metabolism of the cyclic tetrapyrrole compound porphyrin. It is involved in the conversion of hydroxymethylbilane into uroporphyrinogen III. This enzyme catalyses the inversion of the final pyrrole unit (ring D) of the linear tetrapyrrole molecule, linking it to the first pyrrole unit (ring A), thereby generating a large macrocyclic structure, uroporphyrinogen III. [1] The enzyme folds into two alpha/beta domains connected by a beta-ladder, the active site being located between the two domains. [2]
Contents