
Nicotinamide adenine dinucleotide (NAD) is a coenzyme central to metabolism. Found in all living cells, NAD is called a dinucleotide because it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an adenine nucleobase and the other, nicotinamide. NAD exists in two forms: an oxidized and reduced form, abbreviated as NAD+ and NADH (H for hydrogen), respectively.
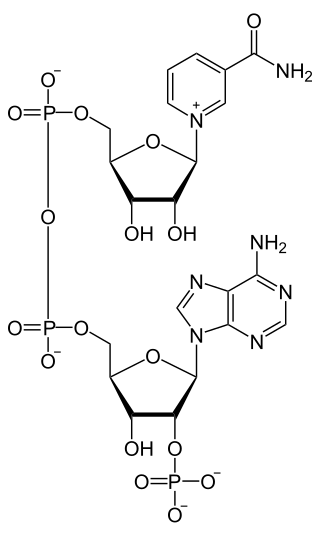
Nicotinamide adenine dinucleotide phosphate, abbreviated NADP+ or, in older notation, TPN (triphosphopyridine nucleotide), is a cofactor used in anabolic reactions, such as the Calvin cycle and lipid and nucleic acid syntheses, which require NADPH as a reducing agent ('hydrogen source'). NADPH is the reduced form of NADP+, the oxidized form. NADP+ is used by all forms of cellular life.

In biochemistry, mixed acid fermentation is the metabolic process by which a six-carbon sugar is converted into a complex and variable mixture of acids. It is an anaerobic (non-oxygen-requiring) fermentation reaction that is common in bacteria. It is characteristic for members of the Enterobacteriaceae, a large family of Gram-negative bacteria that includes E. coli.
In molecular biology, the protein domain Saccharopine dehydrogenase (SDH), also named Saccharopine reductase, is an enzyme involved in the metabolism of the amino acid lysine, via an intermediate substance called saccharopine. The Saccharopine dehydrogenase enzyme can be classified under EC 1.5.1.7, EC 1.5.1.8, EC 1.5.1.9, and EC 1.5.1.10. It has an important function in lysine metabolism and catalyses a reaction in the alpha-Aminoadipic acid pathway. This pathway is unique to fungal organisms therefore, this molecule could be useful in the search for new antibiotics. This protein family also includes saccharopine dehydrogenase and homospermidine synthase. It is found in prokaryotes, eukaryotes and archaea.

In enzymology, a 3-hydroxyacyl-CoA dehydrogenase (EC 1.1.1.35) is an enzyme that catalyzes the chemical reaction
In enzymology, a mycothiol-dependent formaldehyde dehydrogenase (EC 1.1.1.306) is an enzyme that catalyzes the chemical reaction
In enzymology, 6,7-dihydropteridine reductase (EC 1.5.1.34, also Dihydrobiopterin reductase) is an enzyme that catalyzes the chemical reaction
In enzymology, a 2-methylcitrate synthase (EC 2.3.3.5) is an enzyme that catalyzes the chemical reaction
3-hydroxypropionate dehydrogenase (NADP+) (EC 1.1.1.298) is an enzyme with systematic name 3-hydroxypropionate:NADP+ oxidoreductase. This enzyme catalyses the following chemical reaction
D-xylose reductase (EC 1.1.1.307, XylR, XyrA, msXR, dsXR, monospecific xylose reductase, dual specific xylose reductase, NAD(P)H-dependent xylose reductase, xylose reductase) is an enzyme with systematic name xylitol:NAD(P)+ oxidoreductase. This enzyme catalyses the following chemical reaction
Acrylyl-CoA reductase (NADPH) (EC 1.3.1.84) is an enzyme with systematic name propanoyl-CoA:NADP+ oxidoreductase. This enzyme catalyses the following chemical reaction
Flavin reductase (NADH) (EC 1.5.1.36, NADH-dependent flavin reductase, flavin:NADH oxidoreductase) is an enzyme with systematic name flavin:NAD+ oxidoreductase. This enzyme catalyses the following chemical reaction
FAD reductase (NADH) (EC 1.5.1.37, NADH-FAD reductase, NADH-dependent FAD reductase) is an enzyme with systematic name FADH2:NAD+ oxidoreductase. This enzyme catalyses the following chemical reaction
FMN reductase (NADH) (EC 1.5.1.42, NADH-FMN reductase) is an enzyme with systematic name FMNH2:NAD+ oxidoreductase. This enzyme catalyses the following chemical reaction

NADH:ubiquinone reductase (non-electrogenic) (EC 1.6.5.9, NDH-2, ubiquinone reductase, coenzyme Q reductase, dihydronicotinamide adenine dinucleotide-coenzyme Q reductase, DPNH-coenzyme Q reductase, DPNH-ubiquinone reductase, NADH-coenzyme Q oxidoreductase, NADH-coenzyme Q reductase, NADH-CoQ oxidoreductase, NADH-CoQ reductase) is an enzyme with systematic name NADH:ubiquinone oxidoreductase. This enzyme catalyses the following chemical reaction:
Putidaredoxin—NAD+ reductase (EC 1.18.1.5, putidaredoxin reductase, camA (gene)) is an enzyme with systematic name putidaredoxin:NAD+ oxidoreductase. This enzyme catalyses the following chemical reaction
3-hydroxypropionyl-CoA dehydratase (EC 4.2.1.116) is an enzyme with systematic name 3-hydroxypropionyl-CoA hydro-lyase. This enzyme catalyses the following chemical reaction
Cortisone reductase deficiency is caused by dysregulation of the 11β-hydroxysteroid dehydrogenase type 1 enzyme (11β-HSD1), otherwise known as cortisone reductase, a bi-directional enzyme, which catalyzes the interconversion of cortisone to cortisol in the presence of NADH as a co-factor. If levels of NADH are low, the enzyme catalyses the reverse reaction, from cortisol to cortisone, using NAD+ as a co-factor.
Cortisol is a glucocorticoid that plays a variety of roles in many different biochemical pathways, including, but not limited to: gluconeogenesis, suppressing immune system responses and carbohydrate metabolism.
One of the symptoms of cortisone reductase deficiency is hyperandrogenism, resulting from activation of the Hypothalamic–pituitary–adrenal axis. The deficiency has been known to exhibit symptoms of other disorders such as Polycystic Ovary Syndrome in women. Cortisone Reductase Deficiency alone has been reported in fewer than ten cases in total, all but one case were women. Elevated activity of 11β-HSD1 can lead to obesity or Type II Diabetes, because of the role of cortisol in carbohydrate metabolism and gluconeogenesis.

Biliverdin reductase B is a protein that in humans is encoded by the BLVRB gene.

Chlorophyllide a and Chlorophyllide b are the biosynthetic precursors of chlorophyll a and chlorophyll b respectively. Their propionic acid groups are converted to phytyl esters by the enzyme chlorophyll synthase in the final step of the pathway. Thus the main interest in these chemical compounds has been in the study of chlorophyll biosynthesis in plants, algae and cyanobacteria. Chlorophyllide a is also an intermediate in the biosynthesis of bacteriochlorophylls.










