Related Research Articles

Inflammation is part of the complex biological response of body tissues to harmful stimuli, such as pathogens, damaged cells, or irritants, and is a protective response involving immune cells, blood vessels, and molecular mediators. The function of inflammation is to eliminate the initial cause of cell injury, clear out necrotic cells and tissues damaged from the original insult and the inflammatory process, and initiate tissue repair.

A granuloma is an aggregation of macrophages that forms in response to chronic inflammation. This occurs when the immune system attempts to isolate foreign substances that it is otherwise unable to eliminate. Such substances include infectious organisms including bacteria and fungi, as well as other materials such as foreign objects, keratin, and suture fragments.
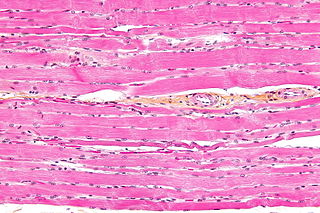
Striated muscle tissue is a muscle tissue that features repeating functional units called sarcomeres. The presence of sarcomeres manifests as a series of bands visible along the muscle fibers, which is responsible for the striated appearance observed in microscopic images of this tissue. There are two types of striated muscle:

Kupffer cells, also known as stellate macrophages and Kupffer–Browicz cells, are specialized cells localized in the liver within the lumen of the liver sinusoids and are adhesive to their endothelial cells which make up the blood vessel walls. Kupffer cells comprise the largest population of tissue-resident macrophages in the body. Gut bacteria, bacterial endotoxins, and microbial debris transported to the liver from the gastrointestinal tract via the portal vein will first come in contact with Kupffer cells, the first immune cells in the liver. It is because of this that any change to Kupffer cell functions can be connected to various liver diseases such as alcoholic liver disease, viral hepatitis, intrahepatic cholestasis, steatohepatitis, activation or rejection of the liver during liver transplantation and liver fibrosis. They form part of the mononuclear phagocyte system.
Pattern recognition receptors (PRRs) play a crucial role in the proper function of the innate immune system. PRRs are germline-encoded host sensors, which detect molecules typical for the pathogens. They are proteins expressed, mainly, by cells of the innate immune system, such as dendritic cells, macrophages, monocytes, neutrophils and epithelial cells, to identify two classes of molecules: pathogen-associated molecular patterns (PAMPs), which are associated with microbial pathogens, and damage-associated molecular patterns (DAMPs), which are associated with components of host's cells that are released during cell damage or death. They are also called primitive pattern recognition receptors because they evolved before other parts of the immune system, particularly before adaptive immunity. PRRs also mediate the initiation of antigen-specific adaptive immune response and release of inflammatory cytokines.

The innate, or nonspecific, immune system is one of the two main immunity strategies in vertebrates. The innate immune system is an older evolutionary defense strategy, relatively speaking, and is the dominant immune system response found in plants, fungi, insects, and primitive multicellular organisms.
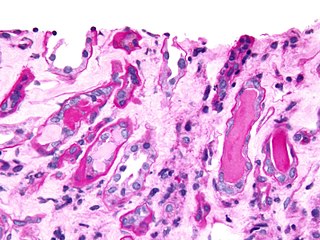
Urinary casts are microscopic cylindrical structures produced by the kidney and present in the urine in certain disease states. They form in the distal convoluted tubule and collecting ducts of nephrons, then dislodge and pass into the urine, where they can be detected by microscopy.
Feline lower urinary tract disease (FLUTD) is a generic category term to describe any disorder affecting the bladder or urethra of cats.
Phosphate nephropathy or nephrocalcinosis is an adverse renal condition that arises with a formation of phosphate crystals within the kidney's tubules. This renal insufficiency is associated with the use of oral sodium phosphate (OSP) such as C.B. Fleet's Phospho soda and Salix's Visocol, for bowel cleansing prior to a colonoscopy.

Bladder stones or uroliths are a common occurrence in animals, especially in domestic animals such as dogs and cats. Occurrence in other species, including tortoises, has been reported as well. The stones form in the urinary bladder in varying size and numbers secondary to infection, dietary influences, and genetics. Stones can form in any part of the urinary tract in dogs and cats, but unlike in humans, stones of the kidney are less common and do not often cause significant disease, although they can contribute to pyelonephritis and chronic kidney disease. Types of stones include struvite, calcium oxalate, urate, cystine, calcium phosphate, and silicate. Struvite and calcium oxalate stones are by far the most common.

NLR family pyrin domain containing 3 (NLRP3), is a protein that in humans is encoded by the NLRP3 gene located on the long arm of chromosome 1.
Pyroptosis is a highly inflammatory form of lytic programmed cell death that occurs most frequently upon infection with intracellular pathogens and is likely to form part of the antimicrobial response. This process promotes the rapid clearance of various bacterial, viral, fungal and protozoan infections by removing intracellular replication niches and enhancing the host's defensive responses. Pyroptosis can take place in immune cells and is also reported to occur in keratinocytes and some epithelial cells.
Nephrocalcinosis, once known as Albright's calcinosis after Fuller Albright, is a term originally used to describe deposition of calcium salts in the renal parenchyma due to hyperparathyroidism. The term nephrocalcinosis is used to describe the deposition of both calcium oxalate and calcium phosphate. It may cause acute kidney injury. It is now more commonly used to describe diffuse, fine, renal parenchymal calcification in radiology. It is caused by multiple different conditions and is determined progressive kidney dysfunction. These outlines eventually come together to form a dense mass. During its early stages, nephrocalcinosis is visible on x-ray, and appears as a fine granular mottling over the renal outlines. It is most commonly seen as an incidental finding with medullary sponge kidney on an abdominal x-ray. However, it may be severe enough to cause renal tubular acidosis or even end stage kidney disease, due to disruption of the kidney tissue by the deposited calcium.

Allograft inflammatory factor 1 (AIF-1) also known as ionized calcium-binding adapter molecule 1 (IBA1) is a protein that in humans is encoded by the AIF1 gene.

Inflammasomes are cytosolic multiprotein oligomers of the innate immune system responsible for the activation of inflammatory responses. Activation and assembly of the inflammasome promotes proteolytic cleavage, maturation and secretion of pro-inflammatory cytokines interleukin 1β (IL-1β) and interleukin 18 (IL-18), as well as cleavage of Gasdermin-D. The N-terminal fragment resulting from this cleavage induces a pro-inflammatory form of programmed cell death distinct from apoptosis, referred to as pyroptosis, and is responsible for secretion of the mature cytokines, presumably through the formation of pores in the plasma membrane.
An inflammatory cytokine or proinflammatory cytokine is a type of signaling molecule that is secreted from immune cells like helper T cells (Th) and macrophages, and certain other cell types that promote inflammation. They include interleukin-1 (IL-1), IL-6, IL-12, and IL-18, tumor necrosis factor alpha (TNF-α), interferon gamma (IFNγ), and granulocyte-macrophage colony stimulating factor (GM-CSF) and play an important role in mediating the innate immune response. Inflammatory cytokines are predominantly produced by and involved in the upregulation of inflammatory reactions.

Necroptosis is a programmed form of necrosis, or inflammatory cell death. Conventionally, necrosis is associated with unprogrammed cell death resulting from cellular damage or infiltration by pathogens, in contrast to orderly, programmed cell death via apoptosis. The discovery of necroptosis showed that cells can execute necrosis in a programmed fashion and that apoptosis is not always the preferred form of cell death. Furthermore, the immunogenic nature of necroptosis favors its participation in certain circumstances, such as aiding in defence against pathogens by the immune system. Necroptosis is well defined as a viral defense mechanism, allowing the cell to undergo "cellular suicide" in a caspase-independent fashion in the presence of viral caspase inhibitors to restrict virus replication. In addition to being a response to disease, necroptosis has also been characterized as a component of inflammatory diseases such as Crohn's disease, pancreatitis, and myocardial infarction.
Neuroinflammation is inflammation of the nervous tissue. It may be initiated in response to a variety of cues, including infection, traumatic brain injury, toxic metabolites, or autoimmunity. In the central nervous system (CNS), including the brain and spinal cord, microglia are the resident innate immune cells that are activated in response to these cues. The CNS is typically an immunologically privileged site because peripheral immune cells are generally blocked by the blood–brain barrier (BBB), a specialized structure composed of astrocytes and endothelial cells. However, circulating peripheral immune cells may surpass a compromised BBB and encounter neurons and glial cells expressing major histocompatibility complex molecules, perpetuating the immune response. Although the response is initiated to protect the central nervous system from the infectious agent, the effect may be toxic and widespread inflammation as well as further migration of leukocytes through the blood–brain barrier.

Dapansutrile (OLT1177) is an inhibitor of the NLRP3 inflammasome.
Autoinflammatory diseases (AIDs) are a group of rare disorders caused by a dysfunction of the innate immune system.They are characterised by a perdiodic or chronic systemic inflammation usually without the involvement of adaptive immunity.
References
- 1 2 3 4 5 6 7 8 9 10 11 Mulay, Shrikant R.; Longo, Dan L.; Anders, Hans-Joachim (2016). "Crystallopathies". New England Journal of Medicine. 374 (25): 2465–2476. doi:10.1056/NEJMra1601611. ISSN 0028-4793. PMID 27332905.
- ↑ Mulay, Shrikant R.; Steiger, Stefanie; Shi, Chongxu; Anders, Hans‐Joachim (2020). "A guide to crystal‐related and nano‐ or microparticle‐related tissue responses". The FEBS Journal. 287 (5): 818–832. doi: 10.1111/febs.15174 . ISSN 1742-464X. PMID 31829497.
- ↑ Steiger, Stefanie; Grill, Julia Felicitas; Ma, Qiuyue; Bäuerle, Tobias; Jordan, Jutta; Smolle, Michaela; Böhland, Claudia; Lech, Maciej; Anders, Hans-Joachim (2018). "Anti-Transforming Growth Factor β IgG Elicits a Dual Effect on Calcium Oxalate Crystallization and Progressive Nephrocalcinosis-Related Chronic Kidney Disease". Frontiers in Immunology. 9: 619. doi: 10.3389/fimmu.2018.00619 . ISSN 1664-3224. PMC 5884871 . PMID 29651290.