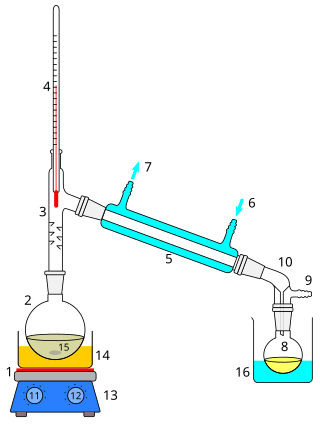
Distillation, also classical distillation, is the process of separating the component substances of a liquid mixture of two or more chemically discrete substances; the separation process is realized by way of the selective boiling of the mixture and the condensation of the vapors in a still.
Kerosene, or paraffin, is a combustible hydrocarbon liquid which is derived from petroleum. It is widely used as a fuel in aviation as well as households. Its name derives from Greek: κηρός (kērós) meaning "wax", and was registered as a trademark by Nova Scotia geologist and inventor Abraham Gesner in 1854 before evolving into a generic trademark. It is sometimes spelled kerosine in scientific and industrial usage.
Perfume is a mixture of fragrant essential oils or aroma compounds (fragrances), fixatives and solvents, usually in liquid form, used to give the human body, animals, food, objects, and living-spaces an agreeable scent. Perfumes can be defined as substances that emit and diffuse a pleasant and fragrant odor. They consist of manmade mixtures of aromatic chemicals and essential oils. The 1939 Nobel Laureate for Chemistry, Leopold Ružička stated in 1945 that "right from the earliest days of scientific chemistry up to the present time, perfumes have substantially contributed to the development of organic chemistry as regards methods, systematic classification, and theory."

An essential oil is a concentrated hydrophobic liquid containing volatile chemical compounds from plants. Essential oils are also known as volatile oils, ethereal oils, aetheroleum, or simply as the oil of the plant from which they were extracted, such as oil of clove. An essential oil is essential in the sense that it contains the essence of the plant's fragrance—the characteristic fragrance of the plant from which it is derived. The term "essential" used here does not mean indispensable or usable by the human body, as with the terms essential amino acid or essential fatty acid, which are so called because they are nutritionally required by a living organism.

A tincture is typically an extract of plant or animal material dissolved in ethanol. Solvent concentrations of 25–60% are common, but may run as high as 90%. In chemistry, a tincture is a solution that has ethanol as its solvent. In herbal medicine, alcoholic tinctures are made with various ethanol concentrations, which should be at least 20% alcohol for preservation purposes.

Used in perfumery and aromatherapy, absolutes are similar to essential oils. They are concentrated, highly aromatic, oily mixtures extracted from plants. Whereas essential oils are produced by distillation, boiling, or pressing, absolutes are produced through solvent extraction, or more traditionally, through enfleurage.
Enfleurage is a process that uses odorless fats that are solid at room temperature to capture the fragrant compounds, such as volatile oils, produced by plants. The process can be "cold" enfleurage or "hot" enfleurage.

Steam distillation is a separation process that consists of distilling water together with other volatile and non-volatile components. The steam from the boiling water carries the vapor of the volatiles to a condenser; both are cooled and return to the liquid or solid state, while the non-volatile residues remain behind in the boiling container.
Supercritical fluid extraction (SFE) is the process of separating one component (the extractant) from another (the matrix) using supercritical fluids as the extracting solvent. Extraction is usually from a solid matrix, but can also be from liquids. SFE can be used as a sample preparation step for analytical purposes, or on a larger scale to either strip unwanted material from a product (e.g. decaffeination) or collect a desired product (e.g. essential oils). These essential oils can include limonene and other straight solvents. Carbon dioxide (CO2) is the most used supercritical fluid, sometimes modified by co-solvents such as ethanol or methanol. Extraction conditions for supercritical carbon dioxide are above the critical temperature of 31 °C and critical pressure of 74 bar. Addition of modifiers may slightly alter this. The discussion below will mainly refer to extraction with CO2, except where specified.

Rose oil is the essential oil extracted from the petals of various types of rose. Rose ottos are extracted through steam distillation, while rose absolutes are obtained through solvent extraction, the absolute being used more commonly in perfumery. The production technique originated in Greater Iran. Even with their high price and the advent of organic synthesis, rose oils are still perhaps the most widely used essential oil in perfumery.

Neroli oil is an essential oil produced from the blossom of the bitter orange tree. Its scent is sweet, honeyed and somewhat metallic with green and spicy facets. Orange blossom is also extracted from the same blossom and both extracts are extensively used in perfumery. Orange blossom can be described as smelling sweeter, warmer and more floral than neroli. The difference between how neroli and orange blossom smell and why they are referred to with different names, is a result of the process of extraction that is used to obtain the oil from the blooms. Neroli is extracted by steam distillation and orange blossom is extracted via a process of enfleurage or solvent extraction.
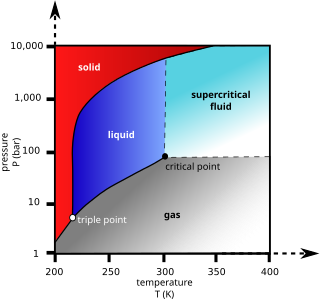
Supercritical carbon dioxide is a fluid state of carbon dioxide where it is held at or above its critical temperature and critical pressure.

An extract (essence) is a substance made by extracting a part of a raw material, often by using a solvent such as ethanol, oil or water. Extracts may be sold as tinctures, absolutes or in powder form.

A florentine flask, also known as florentine receiver, florentine separator or essencier, other shapes called florentine vase or florentine vessel, is an oil–water separator fed with condensed vapors of a steam distillation in a fragrance extraction process.

Plant oils or vegetable oils are oils derived from plant sources, as opposed to animal fats or petroleum. There are three primary types of plant oil, differing both the means of extracting the relevant parts of the plant, and in the nature of the resulting oil:
- Vegetable fats and oils were historically extracted by putting part of the plant under pressure, squeezing out the oil.
- Macerated oils consist of a base oil to which parts of plants are added.
- Essential oils are composed of volatile aromatic compounds, extracted from plants by distillation.

Superheated water is liquid water under pressure at temperatures between the usual boiling point, 100 °C (212 °F) and the critical temperature, 374 °C (705 °F). It is also known as "subcritical water" or "pressurized hot water". Superheated water is stable because of overpressure that raises the boiling point, or by heating it in a sealed vessel with a headspace, where the liquid water is in equilibrium with vapour at the saturated vapor pressure. This is distinct from the use of the term superheating to refer to water at atmospheric pressure above its normal boiling point, which has not boiled due to a lack of nucleation sites.
Winterizationof oil is a process that uses a solvent and cold temperatures to separate lipids and other desired oil compounds from waxes. Winterization is a type of fractionation, the general process of separating the triglycerides found in fats and oils, using the difference in their melting points, solubility, and volatility.

Hash oil or cannabis oil is an oleoresin obtained by the extraction of cannabis or hashish. It is a cannabis concentrate containing many of its resins and terpenes – in particular, tetrahydrocannabinol (THC), cannabidiol (CBD), and other cannabinoids. Hash oil is usually consumed by smoking, vaporizing or eating. Preparations of hash oil may be solid or semi-liquid colloids depending on both production method and temperature and are usually identified by their appearance or characteristics. Color most commonly ranges from transparent golden or light brown, to tan or black. There are various extraction methods, most involving a solvent, such as butane or ethanol.
Concrete, in perfumery, is a waxy mass obtained by solvent extraction of fresh plant material. It is usually used for the production of absolutes.













