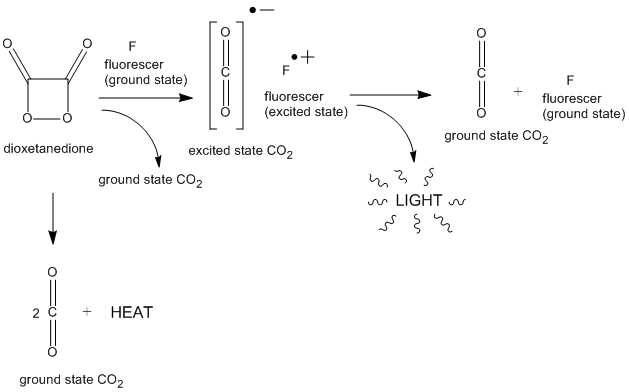
Peroxyoxalates are esters initially formed by the reaction of hydrogen peroxide with oxalate diesters or oxalyl chloride, with or without a base, although the reaction is faster with base. However, they are intermediates that will rapidly transform into 1,2-dioxetanedione, another high-energy intermediate. The likely mechanism of 1,2-dioxetanedione formation from peroxyoxalate in base is illustrated below:

As seen above, 1,2-Dioxetanedione will rapidly decompose into carbon dioxide (CO2). If there is no fluorescer present, only heat will be released. However, in the presence of a fluorescer, light can be generated (chemiluminescence).
Peroxyoxalate chemiluminescence (CL) was first reported by Rauhut in 1967 [1] in the reaction of diphenyl oxalate. The emission is generated by the reaction of an oxalate ester with hydrogen peroxide in the presence of a suitably fluorescent energy acceptor. This reaction is used in glow sticks.

The three most widely used oxalates are bis(2,4,6-trichlorophenyl)oxalate (TCPO), Bis(2,4,5-trichlorophenyl-6-carbopentoxyphenyl)oxalate (CPPO) and bis(2,4-dinitrophenyl) oxalate (DNPO). Other aryl oxalates have been synthesized and evaluated with respect to their possible analytical applications. [2] Divanillyl oxalate, a more eco-friendly or "green" oxalate for chemiluminescence, decomposes into the nontoxic, biodegradable compound vanillin and works in nontoxic, biodegradable triacetin. [3] Peroxyoxalate CL is an example of indirect or sensitized chemiluminescence in which the energy from an excited intermediate is transferred to a suitable fluorescent molecule, which relaxes to the ground state by emitting a photon. Rauhut and co-workers have reported that the intermediate responsible for providing the energy of excitation is 1,2-dioxetanedione. [1] [4] The peroxyoxalate reaction is able to excite many different compounds, having emissions spanning the visible and infrared regions of the spectrum, [4] [5] and the reaction can supply up to 440 kJ mol-1, corresponding to excitation at 272 nm. [6] It has been found, however, that the chemiluminescence intensity corrected for quantum yield decreases as the singlet excitation energy of the fluorescent molecule increases. [7] There is also a linear relationship between the corrected chemiluminescence intensity and the oxidation potential of the molecule. [7] This suggests the possibility of an electron transfer step in the mechanism, as demonstrated in several other chemiluminescence systems. [8] [9] [10] [11] It has been postulated that a transient charge transfer complex is formed between the intermediate 1,2-dioxetanedione and the fluorescer, [12] and a modified mechanism was proposed involving the transfer of an electron from the fluorescer to the reactive intermediate. [13] The emission of light is thought to result from the annihilation of the fluorescer radical cation with the carbon dioxide radical anion formed when the 1,2-dioxetanedione decomposes. [14] This process is called chemically induced electron exchange luminescence (CIEEL).
Chemiluminescent reactions are widely used in analytical chemistry. [15] [16]