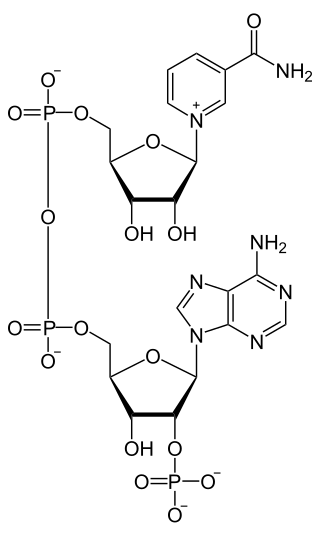
Nicotinamide adenine dinucleotide phosphate, abbreviated NADP+ or, in older notation, TPN (triphosphopyridine nucleotide), is a cofactor used in anabolic reactions, such as the Calvin cycle and lipid and nucleic acid syntheses, which require NADPH as a reducing agent ('hydrogen source'). NADPH is the reduced form of NADP+, the oxidized form. NADP+ is used by all forms of cellular life.

5α-Reductases, also known as 3-oxo-5α-steroid 4-dehydrogenases, are enzymes involved in steroid metabolism. They participate in three metabolic pathways: bile acid biosynthesis, androgen and estrogen metabolism. There are three isozymes of 5α-reductase encoded by the genes SRD5A1, SRD5A2, and SRD5A3.

In enzymology, aldose reductase is a cytosolic NADPH-dependent oxidoreductase that catalyzes the reduction of a variety of aldehydes and carbonyls, including monosaccharides. It is primarily known for catalyzing the reduction of glucose to sorbitol, the first step in polyol pathway of glucose metabolism.

Dicarbonyl/L-xylulose reductase, also known as carbonyl reductase II, is an enzyme that in human is encoded by the DCXR gene located on chromosome 17.
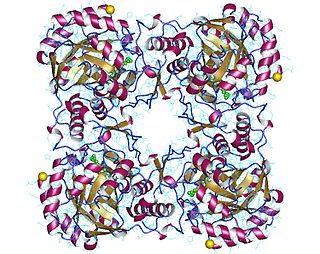
GMP reductase EC 1.7.1.7 is an enzyme that catalyzes the irreversible and NADPH-dependent reductive deamination of GMP into IMP.
In enzymology, a dihydrokaempferol 4-reductase (EC 1.1.1.219) is an enzyme that catalyzes the chemical reaction
In enzymology, a GDP-L-fucose synthase (EC 1.1.1.271) is an enzyme that catalyzes the chemical reaction

Sepiapterin reductase is an enzyme that in humans is encoded by the SPR gene.
In enzymology, a 2,5-didehydrogluconate reductase (EC 1.1.1.274) is an enzyme that catalyzes the chemical reaction

3-dehydrosphinganine reductase also known as 3-ketodihydrosphingosine reductase (KDSR) or follicular variant translocation protein 1 (FVT1) is an enzyme that in humans is encoded by the KDSR gene.
In enzymology, a precorrin-6A reductase (EC 1.3.1.54) is an enzyme that catalyzes the chemical reaction

In enzymology, an aspartate-semialdehyde dehydrogenase is an enzyme that is very important in the biosynthesis of amino acids in prokaryotes, fungi, and some higher plants. It forms an early branch point in the metabolic pathway forming lysine, methionine, leucine and isoleucine from aspartate. This pathway also produces diaminopimelate which plays an essential role in bacterial cell wall formation. There is particular interest in ASADH as disabling this enzyme proves fatal to the organism giving rise to the possibility of a new class of antibiotics, fungicides, and herbicides aimed at inhibiting it.
A glutamyl-tRNA reductase (EC 1.2.1.70) is an enzyme that catalyzes the chemical reaction
In enzymology, a ferredoxin-NADP+ reductase (EC 1.18.1.2) abbreviated FNR, is an enzyme that catalyzes the chemical reaction

[Methionine synthase] reductase, or Methionine synthase reductase, encoded by the gene MTRR, is an enzyme that is responsible for the reduction of methionine synthase inside human body. This enzyme is crucial for maintaining the one carbon metabolism, specifically the folate cycle. The enzyme employs one coenzyme, flavoprotein.
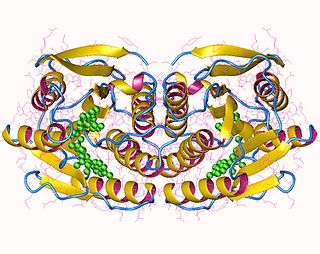
In enzymology, 6,7-dihydropteridine reductase (EC 1.5.1.34, also Dihydrobiopterin reductase) is an enzyme that catalyzes the chemical reaction

Adrenodoxin reductase, was first isolated from bovine adrenal cortex where it functions as the first enzyme in the mitochondrial P450 systems that catalyze essential steps in steroid hormone biosynthesis. Examination of complete genome sequences revealed that adrenodoxin reductase gene is present in most metazoans and prokaryotes.

Steroid 5-alpha-reductase 3, also known as 3-oxo-5-alpha-steroid 4-dehydrogenase 3, is an enzyme that in humans is encoded by the SRD5A3 gene. It is one of three forms of 5α-reductase.
Ditrans,polycis-polyprenyl diphosphate synthase is an enzyme with systematic name (2E,6E)-farnesyl-diphosphate:isopentenyl-diphosphate cistransferase . This enzyme catalyses the following chemical reaction

Chlorophyllide a and Chlorophyllide b are the biosynthetic precursors of chlorophyll a and chlorophyll b respectively. Their propionic acid groups are converted to phytyl esters by the enzyme chlorophyll synthase in the final step of the pathway. Thus the main interest in these chemical compounds has been in the study of chlorophyll biosynthesis in plants, algae and cyanobacteria. Chlorophyllide a is also an intermediate in the biosynthesis of bacteriochlorophylls.













