
Shigellosis is an infection of the intestines caused by Shigella bacteria. Symptoms generally start one to two days after exposure and include diarrhea, fever, abdominal pain, and feeling the need to pass stools even when the bowels are empty. The diarrhea may be bloody. Symptoms typically last five to seven days and it may take several months before bowel habits return entirely to normal. Complications can include reactive arthritis, sepsis, seizures, and hemolytic uremic syndrome.

An agar plate is a Petri dish that contains a growth medium solidified with agar, used to culture microorganisms. Sometimes selective compounds are added to influence growth, such as antibiotics.

A microbiological culture, or microbial culture, is a method of multiplying microbial organisms by letting them reproduce in predetermined culture medium under controlled laboratory conditions. Microbial cultures are foundational and basic diagnostic methods used as research tools in molecular biology.

Bacteriological water analysis is a method of analysing water to estimate the numbers of bacteria present and, if needed, to find out what sort of bacteria they are. It represents one aspect of water quality. It is a microbiological analytical procedure which uses samples of water and from these samples determines the concentration of bacteria. It is then possible to draw inferences about the suitability of the water for use from these concentrations. This process is used, for example, to routinely confirm that water is safe for human consumption or that bathing and recreational waters are safe to use.

Coliform bacteria are defined as either motile or non-motile Gram-negative non-spore forming bacilli that possess β-galactosidase to produce acids and gases under their optimal growth temperature of 35–37 °C. They can be aerobes or facultative aerobes, and are a commonly used indicator of low sanitary quality of foods, milk, and water. Coliforms can be found in the aquatic environment, in soil and on vegetation; they are universally present in large numbers in the feces of warm-blooded animals as they are known to inhabit the gastrointestinal system. While coliform bacteria are not normally the cause of serious illness, they are easy to culture, and their presence is used to infer that other pathogenic organisms of fecal origin may be present in a sample, or that said sample is not safe to consume. Such pathogens include disease-causing bacteria, viruses, or protozoa and many multicellular parasites. Every drinking water source must be tested for the presence of these total coliform bacteria.

Eosin methylene blue is a selective and differential media used for the identification of Gram-negative bacteria, specifically the Enterobacteriaceae. EMB inhibits the growth of most Gram-positive bacteria. EMB is often used to confirm the presence of coliforms in a sample. It contains two dyes, eosin and methylene blue in the ratio of 6:1. EMB is a differential microbiological media, which inhibits the growth of Gram-positive bacteria and differentiates bacteria that ferment lactose from those that do not. Organisms that ferment lactose appear dark/black or green often with "nucleated colonies"—colonies with dark centers. Organisms that do not ferment lactose will appear pink and often mucoid.

A growth medium or culture medium is a solid, liquid, or semi-solid designed to support the growth of a population of microorganisms or cells via the process of cell proliferation or small plants like the moss Physcomitrella patens. Different types of media are used for growing different types of cells.

MacConkey agar is a selective and differential culture medium for bacteria. It is designed to selectively isolate Gram-negative and enteric bacteria and differentiate them based on lactose fermentation. Lactose fermenters turn red or pink on MacConkey agar, and nonfermenters do not change color. The media inhibits growth of Gram-positive organisms with crystal violet and bile salts, allowing for the selection and isolation of gram-negative bacteria. The media detects lactose fermentation by enteric bacteria with the pH indicator neutral red.
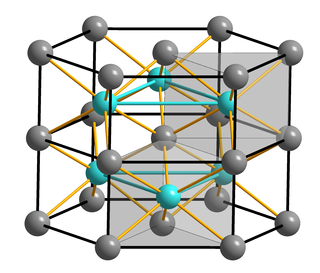
Iron(II) sulfide or ferrous sulfide is one of a family of chemical compounds and minerals with the approximate formula FeS. Iron sulfides are often iron-deficient non-stoichiometric. All are black, water-insoluble solids.

Shigella dysenteriae is a species of the rod-shaped bacterial genus Shigella. Shigella species can cause shigellosis. Shigellae are Gram-negative, non-spore-forming, facultatively anaerobic, nonmotile bacteria. S. dysenteriae has the ability to invade and replicate in various species of epithelial cells and enterocytes.
Sorbitol-MacConkey agar is a variant of traditional MacConkey agar used in the detection of E. coli O157:H7. Traditionally, MacConkey agar has been used to distinguish those bacteria that ferment lactose from those that do not. This is important because gut bacteria, such as Escherichia coli, can typically ferment lactose, while important gut pathogens, such as Salmonella enterica and most shigellas are unable to ferment lactose. Shigella sonnei can ferment lactose, but only after prolonged incubation, so it is referred to as a late-lactose fermenter.

CLED agar is a valuable non-inhibitory growth medium used in the isolation and differentiation of urinary microbes. It contains cystine and lactose and is electrolyte-deficient; the latter trait prevents the swarming of Proteus species. Cystine promotes the formation of cystine-dependent dwarf colonies. Bromothymol blue is the indicator used in the agar, it changes to yellow in case of acid production during fermentation of lactose or changes to deep blue in case of alkalinization. Lactose-positive bacteria build yellow colonies. Bacteria which decarboxylate L-cystine cause an alkaline reaction and build deep blue colonies.

Xylose Lysine Deoxycholate agar is a selective growth medium used in the isolation of Salmonella and Shigella species from clinical samples and from food. The agar was developed by Welton Taylor in 1965. It has a pH of approximately 7.4, leaving it with a bright pink or red appearance due to the indicator phenol red. Sugar fermentation lowers the pH and the phenol red indicator registers this by changing to yellow. Most gut bacteria, including Salmonella, can ferment the sugar xylose to produce acid; Shigella colonies cannot do this and therefore remain red. After exhausting the xylose supply Salmonella colonies will decarboxylate lysine, increasing the pH once again to alkaline and mimicking the red Shigella colonies. Salmonellae metabolise thiosulfate to produce hydrogen sulfide, which leads to the formation of colonies with black centers and allows them to be differentiated from the similarly coloured Shigella colonies.
Leuconostoc mesenteroides is a species of lactic acid bacteria associated with fermentation, under conditions of salinity and low temperatures. In some cases of vegetable and food storage, it was associated with pathogenicity. L. mesenteroides is approximately 0.5-0.7 μm in diameter and has a length of 0.7-1.2 μm, producing small grayish colonies that are typically less than 1.0 mm in diameter. It is facultatively anaerobic, Gram-positive, non-motile, non-sporogenous, and spherical. It often forms lenticular coccoid cells in pairs and chains, however, it can occasionally form short rods with rounded ends in long chains, as its shape can differ depending on what media the species is grown on. L. mesenteroides grows best at 30 °C, but can survive in temperatures ranging from 10 °C to 30 °C. Its optimum pH is 5.5, but can still show growth in pH of 4.5-7.0.

The Triple Sugar Iron (TSI) test is a microbiological test roughly named for its ability to test a microorganism's ability to ferment sugars and to produce hydrogen sulfide. It is often used to differentiate enteric bacteria including Salmonella and Shigella.

Endo agar is a microbiological growth medium with a faint pink colour. Originally developed for the isolation of Salmonella typhi, it is now used mostly as a coliform medium. Most gram-negative organisms grow well in this medium, while growth of gram-positive organisms is inhibited. Coliform organisms ferment the lactose in this medium, producing a green metallic sheen, whereas non-lactose-fermenting organisms produce clear, colourless colonies, i.e. Salmonella species.

Thiosulfate–citrate–bile salts–sucrose agar, or TCBS agar, is a type of selective agar culture plate that is used in microbiology laboratories to isolate Vibrio species. TCBS agar is highly selective for the isolation of V. cholerae and V. parahaemolyticus as well as other Vibrio species. Apart from TCBS agar, other rapid testing dipsticks like immunochromatographic dipstick is also used in endemic areas such as Asia, Africa and Latin America. Though, TCBS agar study is required for confirmation. This becomes immensely important in cases of gastroenteritis caused by campylobacter species, whose symptoms mimic that of cholera. Since no yellow bacterial growth is observed in case of campylobacter species on TCBS agar, chances of incorrect diagnosis can be rectified. TCBS agar contains high concentrations of sodium thiosulfate and sodium citrate to inhibit the growth of Enterobacteriaceae. Inhibition of gram-positive bacteria is achieved by the incorporation of ox gall, which is a naturally occurring substance containing a mixture of bile salts and sodium cholate, a pure bile salt. Sodium thiosulfate also serves as a sulfur source and its presence, in combination with ferric citrate, allows for the easy detection of hydrogen sulfide production. Saccharose (sucrose) is included as a fermentable carbohydrate for metabolism by Vibrio species. The alkaline pH of the medium enhances the recovery of V. cholerae and inhibits the growth of others. Thymol blue and bromothymol blue are included as indicators of pH changes.
Oxidative/fermentation glucose test is a biological technique. It was developed in 1953 by Hugh and Leifson to be utilized in microbiology to determine the way a microorganism metabolizes a carbohydrate such as glucose (dextrose). OF-glucose deeps contain glucose as a carbohydrate, peptones, bromothymol blue indicator for Hugh-Leifson's OF medium or phenol red for King's OF medium, and 0.5% agar.
Thermotoga naphthophila is a hyperthermophilic, anaerobic, non-spore-forming, rod-shaped fermentative heterotroph, with type strain RKU-10T.
Diagnostic microbiology is the study of microbial identification. Since the discovery of the germ theory of disease, scientists have been finding ways to harvest specific organisms. Using methods such as differential media or genome sequencing, physicians and scientists can observe novel functions in organisms for more effective and accurate diagnosis of organisms. Methods used in diagnostic microbiology are often used to take advantage of a particular difference in organisms and attain information about what species it can be identified as, which is often through a reference of previous studies. New studies provide information that others can reference so that scientists can attain a basic understanding of the organism they are examining.















