 | |
| Identifiers | |
|---|---|
| |
3D model (JSmol) |
|
PubChem CID |
|
| |
| |
| Properties | |
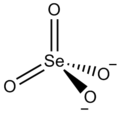
| Ce2(SeO4)3 | |
| Molar mass | 709.121 |
| Density | 4.456g/cm3 (octahydrate) [1] |
| 39.5g(0 °C) 2.51(100 °C) | |
| Related compounds | |
Other anions | cerium(III) sulfate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Cerium(III) selenate is an inorganic compound with the chemical formula Ce2(SeO4)3. It can be obtained by reacting selenic acid and cerium(III) carbonate, and the solvent is evaporated to precipitate crystals. [2] The double salt CsCe(SeO4)2·4H2O can be obtained from mixing cerium(III) selenate and cesium selenate in an aqueous solution, and then evaporating and crystallizing the solution. [3]