 | |
| Identifiers | |
|---|---|
3D model (JSmol) | |
| ChemSpider | |
| ECHA InfoCard | 100.030.635 |
| EC Number |
|
PubChem CID | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
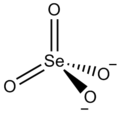
| Cs2SeO4 | |
| Molar mass | 408.77 |
| Appearance | colourless crystals [1] |
| insoluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Caesium selanate is an inorganic compound, with the chemical formula of Cs2SeO4. It can form colourless crystals of the orthorhombic crystal system.