Related Research Articles
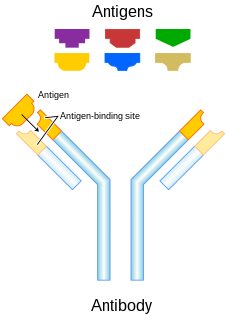
An antibody (Ab), also known as an immunoglobulin (Ig), is a large, Y-shaped protein used by the immune system to identify and neutralize foreign objects such as pathogenic bacteria and viruses. The antibody recognizes a unique molecule of the pathogen, called an antigen. Each tip of the "Y" of an antibody contains a paratope that is specific for one particular epitope on an antigen, allowing these two structures to bind together with precision. Using this binding mechanism, an antibody can tag a microbe or an infected cell for attack by other parts of the immune system, or can neutralize it directly.

The immune system is a network of biological processes that protects an organism against disease. It detects and responds to a wide variety of pathogens, from viruses to parasitic worms, as well as cancer cells and objects such as wood splinters, distinguishing them from the organism's own healthy tissue. Many species have two major subsystems of the immune system. The innate immune system provides a preconfigured response to broad groups of situations and stimuli. The adaptive immune system provides a tailored response to each stimulus by learning to recognize molecules it has previously encountered. Both use molecules and cells to perform their functions.
Natural killer cells, also known as NK cells or large granular lymphocytes (LGL), are a type of cytotoxic lymphocyte critical to the innate immune system. The role of NK cells is analogous to that of cytotoxic T cells in the vertebrate adaptive immune response. NK cells provide rapid responses to virus-infected cells, acting at around 3 days after infection, and respond to tumor formation. Typically, immune cells detect the major histocompatibility complex (MHC) presented on infected cell surfaces, triggering cytokine release, causing the death of the infected cell by lysis or apoptosis. NK cells are unique, however, as they have the ability to recognize and kill stressed cells in the absence of antibodies and MHC, allowing for a much faster immune reaction. They were named "natural killers" because of the notion that they do not require activation to kill cells that are missing "self" markers of MHC class 1. This role is especially important because harmful cells that are missing MHC I markers cannot be detected and destroyed by other immune cells, such as T lymphocyte cells.

An opsonin is any molecule that enhances phagocytosis by marking an antigen for an immune response or marking dead cells for recycling. Opson in ancient Greece referred to the delicious side-dish of any meal, versus the sitos, or the staple of the meal.
Cancer immunotherapy is the artificial stimulation of the immune system to treat cancer, improving on the immune system's natural ability to fight the disease. It is an application of the fundamental research of cancer immunology and a growing subspeciality of oncology.

The adaptive immune system, also referred as the acquired immune system, is a subsystem of the immune system that is composed of specialized, systemic cells and processes that eliminates pathogens by preventing their growth. The acquired immune system is one of the two main immunity strategies found in vertebrates.

CD23, also known as Fc epsilon RII, or FcεRII, is the "low-affinity" receptor for IgE, an antibody isotype involved in allergy and resistance to parasites, and is important in regulation of IgE levels. Unlike many of the antibody receptors, CD23 is a C-type lectin. It is found on mature B cells, activated macrophages, eosinophils, follicular dendritic cells, and platelets.

A Fc receptor is a protein found on the surface of certain cells – including, among others, B lymphocytes, follicular dendritic cells, natural killer cells, macrophages, neutrophils, eosinophils, basophils, human platelets, and mast cells – that contribute to the protective functions of the immune system. Its name is derived from its binding specificity for a part of an antibody known as the Fc region. Fc receptors bind to antibodies that are attached to infected cells or invading pathogens. Their activity stimulates phagocytic or cytotoxic cells to destroy microbes, or infected cells by antibody-mediated phagocytosis or antibody-dependent cell-mediated cytotoxicity. Some viruses such as flaviviruses use Fc receptors to help them infect cells, by a mechanism known as antibody-dependent enhancement of infection.

An immune complex, sometimes called an antigen-antibody complex or antigen-bound antibody, is a molecule formed from the binding of multiple antigens to antibodies. The bound antigen and antibody act as a unitary object, effectively an antigen of its own with a specific epitope. After an antigen-antibody reaction, the immune complexes can be subject to any of a number of responses, including complement deposition, opsonization, phagocytosis, or processing by proteases. Red blood cells carrying CR1-receptors on their surface may bind C3b-coated immune complexes and transport them to phagocytes, mostly in liver and spleen, and return to the general circulation.

The B-cell receptor (BCR) is a transmembrane protein on the surface of a B cell. B-cell receptors are composed of immunoglobulin molecules that form a type 1 transmembrane receptor protein, and are typically located on the outer surface of these lymphocyte cells. Through biochemical signaling and by physically acquiring antigens from the immune synapses, the BCR controls the activation of the B cell. B cells are able to gather and grab antigens by engaging biochemical modules for receptor clustering, cell spreading, generation of pulling forces, and receptor transport, which eventually culminates in endocytosis and antigen presentation. B cells' mechanical activity adheres to a pattern of negative and positive feedbacks that regulate the quantity of removed antigen by manipulating the dynamic of BCR–antigen bonds directly. Particularly, grouping and spreading increase the relation of antigen with BCR, thereby proving sensitivity and amplification. On the other hand, pulling forces delinks the antigen from the BCR, thus testing the quality of antigen binding.
The neonatal Fc receptor is a protein that in humans is encoded by the FCGRT gene.. It is an Fc receptor which is similar in structure to the MHC class I molecule and also associates with beta-2-microglobulin. Further studies revealed a similar receptor in humans, leading to the naming as a neonatal Fc receptor. In humans, however, it is found in the placenta to help facilitate transport of mother's IgG to the growing fetus. It has also been shown to play a role in monitoring IgG and serum albumin turnover. Neonatal Fc receptor expression is up-regulated by the proinflammatory cytokine, TNF-α, and down-regulated by IFN-γ.

Fc receptor-like protein 3 is a protein that in humans is encoded by the FCRL3 gene.

Lymphocyte-activation gene 3, also known as LAG-3, is a protein which in humans is encoded by the LAG3 gene. LAG3, which was discovered in 1990 and was designated CD223 after the Seventh Human Leucocyte Differentiation Antigen Workshop in 2000, is a cell surface molecule with diverse biologic effects on T cell function. It is an immune checkpoint receptor and as such is the target of various drug development programs by pharmaceutical companies seeking to develop new treatments for cancer and autoimmune disorders. In soluble form it is also being developed as a cancer drug in its own right.

Fc receptor-like protein 5 is a protein that in humans is encoded by the FCRL5 gene. FCRL5 has also been designated as CD307.
Fc fragment of IgG receptor IIb is a low affinity inhibitory receptor for the Fc region of immunoglobulin gamma (IgG). FCGR2B participates in the phagocytosis of immune complexes and in the regulation of antibody production by B lymphocytes.

Fc receptor-like A is a protein that in humans is encoded by the FCRLA gene.

Fc fragment of IgA receptor (FCAR) is a human gene that codes for the transmembrane receptor FcαRI, also known as CD89. FcαRI binds the heavy-chain constant region of Immunoglubulin A (IgA) antibodies. FcαRI is present on the cell surface of myeloid lineage cells, including neutrophils, monocytes, macrophages, and eosinophils, though it is notably absent from intestinal macrophages and does not appear on mast cells. FcαRI plays a role in both pro- and anti-inflammatory responses depending on the state of IgA bound. Inside-out signaling primes FcαRI in order for it to bind its ligand, while outside-in signaling caused by ligand binding depends on FcαRI association with the Fc receptor gamma chain.

Fc receptor-like protein 4 is a protein that in humans is encoded by the FCRL4 gene. FCRL4 is an inhibitory receptor expressed on human memory B cells which resides in epithelial tissues.

Fc receptor-like protein 1 is a protein that in humans is encoded by the FCRL1 gene.
The following outline is provided as an overview of and topical guide to immunology:
References
- ↑ Davis RS (2007). "Fc receptor-like molecules". Annual Review of Immunology. 25: 525–60. doi:10.1146/annurev.immunol.25.022106.141541. PMID 17201682.
- ↑ Butovsky O, Jedrychowski MP, Moore CS, Cialic R, Lanser AJ, Gabriely G, Koeglsperger T, Dake B, Wu PM, Doykan CE, Fanek Z, Liu L, Chen Z, Rothstein JD, Ransohoff RM, Gygi SP, Antel JP, Weiner HL (January 2014). "Identification of a unique TGF-β-dependent molecular and functional signature in microglia" (PDF). Nature Neuroscience. 17 (1): 131–43. doi:10.1038/nn.3599. PMC 4066672 . PMID 24316888.
- ↑ Santiago T, Kulemzin SV, Reshetnikova ES, Chikaev NA, Volkova OY, Mechetina LV, Zhao M, Davis RS, Taranin AV, Najakshin AM, Hendershot LM, Burrows PD (January 2011). "FCRLA is a resident endoplasmic reticulum protein that associates with intracellular Igs, IgM, IgG and IgA". International Immunology . 23 (1): 43–53. doi:10.1093/intimm/dxq456. PMC 3003704 . PMID 21149418.