DNA ligase 1 is an enzyme that in humans is encoded by the LIG1 gene. DNA ligase I is the only known eukaryotic DNA ligase involved in both DNA replication and repair, making it the most studied of the ligases.
DNA ligase 1 is an enzyme that in humans is encoded by the LIG1 gene. DNA ligase I is the only known eukaryotic DNA ligase involved in both DNA replication and repair, making it the most studied of the ligases.
It was known that DNA replication occurred through the breakage of the double DNA strand, but the enzyme responsible for ligating the strands back together, and mechanism of action, was unknown until Lehman, Gellert, Richardson, and Hurwitz laboratories, made significant contributions to the discovery of DNA ligase in 1967. [5]

The LIG1 gene encodes a, 120kDa enzyme, 919 residues long, known as DNA ligase I. The DNA ligase I polypeptide contains an N-terminal replication factory-targeting sequence (RFTS), followed by a nuclear localization sequence (NLS), and three functional domains. [6] The three domains consist of an N-terminal DNA binding domain (DBD), and catalytic nucleotidyltransferase (NTase), and C-terminal oligonucleotide / oligosaccharide binding (OB) domains. Although the N-terminus of the peptide has no catalytic activity it is needed for activity within the cells. The N-terminus of the protein contains a replication factory-targeting sequence that is used to recruit it to sites of DNA replication known as replication factories.
Activation and recruitment of DNA ligase I seem to be associated with posttranslational modifications. N-terminal domain is completed through phosphorylation of four serine residues on this domain, Ser51, Ser76, and Ser91 by cyclin-dependent kinase (CDK) and Ser66 by casein kinase II (CKII). Phosphorylation of these residues (Ser66 in particular) has been shown to possibly regulate the interaction between the RFTS to the proliferating cell nuclear antigen (PCNA) when ligase I is recruited to the replication factories during S-phase. [6] [7] Rossi et al. proposed that when Ser66 is dephosphorylated, the RFTS of ligase I interact with PCNA, which was confirmed in vitro by Tom et al. Both data sets provide plausible evidence the N-terminal region of ligase I plays a regulatory role in the enzymes in vivo function in the nucleus. [7] [8] Moreover, the identification of a cyclin binding (Cy) motif in the catalytic C-terminus domain was shown by mutational analysis to play a role in the phosphorylation of serines 91 and 76. Together, the N-terminal serines are substrates of the CDK and CKII, which appear to play an important regulatory role DNA ligase I recruitment to the replication factory during S-phase of the cell cycle. [6] [9]

LIG1 encodes DNA ligase I, which functions in DNA replication and the base excision repair process. [10]
Eukaryotic DNA ligase 1 catalyzes a reaction that is chemically universal to all ligases. DNA ligase 1 utilizes adenosine triphosphate (ATP) to catalyze the energetically favorable ligation events in both DNA replication and repair. During the synthesis phase (S-phase) of the eukaryotic cell cycle, DNA replication occurs. DNA ligase 1 is responsible for joining Okazaki fragments formed during discontinuous DNA synthesis on the DNA's lagging strand after DNA polymerase δ has replaced the RNA primer nucleotides with DNA nucleotides. If the Okazaki fragments are not properly ligated together, the unligated DNA (containing a ‘nick’) could easily degrade to a double strand break, a phenomenon known to cause genetic mutations. In order to ligate these fragments together, the ligase progresses through three steps:

During adenylylation, there is a nucleophilic attack on the alpha phosphate of ATP from a catalytic lysine resulting in the production of inorganic pyrophosphate (PPi) and a covalently bound lysine-AMP intermediate in the active site of DNA ligase 1.
During the AMP transfer step, the DNA ligase becomes associated with the DNA, locates a nick and catalyzes a reaction at the 5’ phosphate site of the DNA nick. An anionic oxygen on the 5’ phosphate of the DNA nick serves as the nucleophile, attacking the alpha phosphate of the covalently bound AMP causing the AMP to be covalently bound intermediate (DNA-AMP intermediate).
In order for the phosphodiester bond to be formed, the DNA-AMP intermediate must be cleaved off. To accomplish this task, there is a nucleophilic attack on the 5’-phosphate from the upstream 3’-hydroxyl which results in the formation of the phosphodiester bond. During this nucleophilic attack, the AMP group is pushed off the 5’ phosphate as the leaving group allowing for the nick to seal and the AMP to be released, completing one cycle of DNA ligation.
Under suboptimal conditions the ligase can disassociate from the DNA before the full reaction is complete. It has been shown that magnesium levels can slow the nick sealing process, causing the ligase to disassociate from the DNA, leaving an aborted adenylylated intermediate incapable of being fixed without the aid of a phosphodiesterase. Aprataxin (a phosphodiesterase) has been shown to act on aborted DNA intermediates via hydrolysis of the AMP-phosphate bond, restoring the DNA to its initial state before the ligase had reacted. [12] [13]

DNA ligase I functions to ligate single stranded DNA breaks in the final step of the base excision repair (BER) pathway. [14] The nitrogenous bases of DNA are commonly damaged by environmental hazards such as reactive oxygen species, toxins, and ionizing radiation. BER is the major repair pathway responsible for excising and replacing damaged bases. Ligase I is involved in the LP-BER pathway, whereas ligase III is involved in the major SN-BER pathway(2). [15] LP-BER proceeds in 4 catalytic steps. First, a DNA glycosylase cleaves the N-glycosidic bond, releasing the damaged base and creating an AP site– a site that lacks a purine or pyrimidine base. In the next step, an AP endonuclease creates a nick at the 5' end of the AP site, generating a hanging deoxyribose phosphate (dRP) residue in place of the AP site. DNA polymerase then synthesizes several new bases in the 5' to 3' direction, generating a hanging stretch of DNA with the dRP at its 5' end. It is at this step that SN-BER and LP-BER diverge in mechanism – in SNBER, only a single nucleotide is added and DNA Polymerase acts as a lyase to excise the AP site. In LP-BER, several bases are synthesized, generating a hanging flap of DNA, which is cleaved by a flap endonuclease. This leaves behind a nicked DNA strand that is sensed and ligated by DNA ligase. [14] [15] [16] The action of ligase I is stimulated by other LP-BER enzymes, particularly AP-endonuclease and DNA polymerase. [16]
Mutations in LIG1 that lead to DNA ligase I deficiency result in immunodeficiency and increased sensitivity to DNA-damaging agents. [10]
There are rare reports of patients exhibiting ligase I deficiency which resulted from inherited mutant alleles. The first case manifested as stunted growth and development and an immunodeficiency. A mouse model was made based on cell lines derived from the patient, confirming that the mutant ligase confers replication errors leading to genomic instability. Notably the mutant mice also showed increases in tumorigenesis. [8] Molecular, cellular, and clinical features of 5 patients from 3 kindreds with biallelic mutations were reported. The patients exhibited hypogammaglobulinemia, lymphopenia, increased proportions of circulating γδT cells, and very large red cells (macrocytosis.) Clinical severity ranged from a mild antibody deficiency to a combined immunodeficiency requiring hematopoietic stem cell transplantation. Chemical and radiation defects were demonstrated to impair the DNA repair pathways. Defects in DNA ligase 1 can thus lead to different forms of autosomal recessive, partial DNA ligase 1 deficiency leading to an immunodeficiency of variable severity. [17]
Ligase I has also been found to be upregulated in proliferating tumor cells, as opposed to benign tumor cell lines and normal human cells. Furthermore, it has been shown that inhibiting ligase I expression in these cells can have a cytotoxic effect, suggesting that ligase I inhibitors may be viable chemotherapeutic agents. [18]
Deficiencies in aprataxin, a phosphodiesterase responsible for reconditioning the DNA (after DNA ligase I aborts the adenylylated DNA intermediate), has been linked to neurodegeneration. This suggests that DNA is incapable of reentering the repair pathway without additional back-up machinery to correct for ligase errors. [13]
With the structure of DNA being well known and many of the components necessary for its manipulation, repair, and usage becoming identified and characterized, researchers are beginning to look into the development of nanoscopic machinery that would be incorporated into a living organism that would possess the ability to treat diseases, fight cancer, and release medications based on a biological stimulus provided by the organism to the nanosocpic machinery. DNA ligase would most likely have to be incorporated into such a machine. [19]

DNA ligase is a type of enzyme that facilitates the joining of DNA strands together by catalyzing the formation of a phosphodiester bond. It plays a role in repairing single-strand breaks in duplex DNA in living organisms, but some forms may specifically repair double-strand breaks. Single-strand breaks are repaired by DNA ligase using the complementary strand of the double helix as a template, with DNA ligase creating the final phosphodiester bond to fully repair the DNA.

Okazaki fragments are short sequences of DNA nucleotides which are synthesized discontinuously and later linked together by the enzyme DNA ligase to create the lagging strand during DNA replication. They were discovered in the 1960s by the Japanese molecular biologists Reiji and Tsuneko Okazaki, along with the help of some of their colleagues.

DNA repair is a collection of processes by which a cell identifies and corrects damage to the DNA molecules that encode its genome. In human cells, both normal metabolic activities and environmental factors such as radiation can cause DNA damage, resulting in tens of thousands of individual molecular lesions per cell per day. Many of these lesions cause structural damage to the DNA molecule and can alter or eliminate the cell's ability to transcribe the gene that the affected DNA encodes. Other lesions induce potentially harmful mutations in the cell's genome, which affect the survival of its daughter cells after it undergoes mitosis. As a consequence, the DNA repair process is constantly active as it responds to damage in the DNA structure. When normal repair processes fail, and when cellular apoptosis does not occur, irreparable DNA damage may occur. This can eventually lead to malignant tumors, or cancer as per the two-hit hypothesis.

A molecular lesion or point lesion is damage to the structure of a biological molecule such as DNA, RNA, or protein. This damage may result in the reduction or absence of normal function, and in rare cases the gain of a new function. Lesions in DNA may consist of breaks or other changes in chemical structure of the helix, ultimately preventing transcription. Meanwhile, lesions in proteins consist of both broken bonds and improper folding of the amino acid chain. While many nucleic acid lesions are general across DNA and RNA, some are specific to one, such as thymine dimers being found exclusively in DNA. Several cellular repair mechanisms exist, ranging from global to specific, in order to prevent lasting damage resulting from lesions.

Non-homologous end joining (NHEJ) is a pathway that repairs double-strand breaks in DNA. It is called "non-homologous" because the break ends are directly ligated without the need for a homologous template, in contrast to homology directed repair (HDR), which requires a homologous sequence to guide repair. NHEJ is active in both non-dividing and proliferating cells, while HDR is not readily accessible in non-dividing cells. The term "non-homologous end joining" was coined in 1996 by Moore and Haber.
DNA glycosylases are a family of enzymes involved in base excision repair, classified under EC number EC 3.2.2. Base excision repair is the mechanism by which damaged bases in DNA are removed and replaced. DNA glycosylases catalyze the first step of this process. They remove the damaged nitrogenous base while leaving the sugar-phosphate backbone intact, creating an apurinic/apyrimidinic site, commonly referred to as an AP site. This is accomplished by flipping the damaged base out of the double helix followed by cleavage of the N-glycosidic bond.

DNA mismatch repair (MMR) is a system for recognizing and repairing erroneous insertion, deletion, and mis-incorporation of bases that can arise during DNA replication and recombination, as well as repairing some forms of DNA damage.
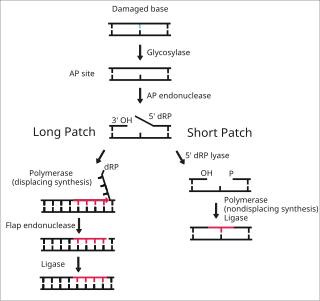
Base excision repair (BER) is a cellular mechanism, studied in the fields of biochemistry and genetics, that repairs damaged DNA throughout the cell cycle. It is responsible primarily for removing small, non-helix-distorting base lesions from the genome. The related nucleotide excision repair pathway repairs bulky helix-distorting lesions. BER is important for removing damaged bases that could otherwise cause mutations by mispairing or lead to breaks in DNA during replication. BER is initiated by DNA glycosylases, which recognize and remove specific damaged or inappropriate bases, forming AP sites. These are then cleaved by an AP endonuclease. The resulting single-strand break can then be processed by either short-patch or long-patch BER.
A nick is a discontinuity in a double stranded DNA molecule where there is no phosphodiester bond between adjacent nucleotides of one strand typically through damage or enzyme action. Nicks allow DNA strands to untwist during replication, and are also thought to play a role in the DNA mismatch repair mechanisms that fix errors on both the leading and lagging daughter strands.

Werner syndrome ATP-dependent helicase, also known as DNA helicase, RecQ-like type 3, is an enzyme that in humans is encoded by the WRN gene. WRN is a member of the RecQ Helicase family. Helicase enzymes generally unwind and separate double-stranded DNA. These activities are necessary before DNA can be copied in preparation for cell division. Helicase enzymes are also critical for making a blueprint of a gene for protein production, a process called transcription. Further evidence suggests that Werner protein plays a critical role in repairing DNA. Overall, this protein helps maintain the structure and integrity of a person's DNA.

Proliferating cell nuclear antigen (PCNA) is a DNA clamp that acts as a processivity factor for DNA polymerase δ in eukaryotic cells and is essential for replication. PCNA is a homotrimer and achieves its processivity by encircling the DNA, where it acts as a scaffold to recruit proteins involved in DNA replication, DNA repair, chromatin remodeling and epigenetics.
DNA repair protein XRCC1, also known as X-ray repair cross-complementing protein 1, is a protein that in humans is encoded by the XRCC1 gene. XRCC1 is involved in DNA repair, where it complexes with DNA ligase III.

Double-strand break repair protein MRE11 is an enzyme that in humans is encoded by the MRE11 gene. The gene has been designated MRE11A to distinguish it from the pseudogene MRE11B that is nowadays named MRE11P1.

DNA ligase 4 is an enzyme that in humans is encoded by the LIG4 gene.
Artemis is a protein that in humans is encoded by the DCLRE1C gene.

DNA polymerase beta, also known as POLB, is an enzyme present in eukaryotes. In humans, it is encoded by the POLB gene.

Flap endonuclease 1 is an enzyme that in humans is encoded by the FEN1 gene.
Uracil-DNA glycosylase is an enzyme. Its most important function is to prevent mutagenesis by eliminating uracil from DNA molecules by cleaving the N-glycosidic bond and initiating the base-excision repair (BER) pathway.

DNA ligase 3 is an enzyme that, in humans, is encoded by the LIG3 gene. The human LIG3 gene encodes ATP-dependent DNA ligases that seal interruptions in the phosphodiester backbone of duplex DNA.

Ligation is the joining of two nucleic acid fragments through the action of an enzyme. It is an essential laboratory procedure in the molecular cloning of DNA, whereby DNA fragments are joined to create recombinant DNA molecules (such as when a foreign DNA fragment is inserted into a plasmid). The ends of DNA fragments are joined by the formation of phosphodiester bonds between the 3'-hydroxyl of one DNA terminus with the 5'-phosphoryl of another. RNA may also be ligated similarly. A co-factor is generally involved in the reaction, and this is usually ATP or NAD+. Eukaryotic cells ligases belong to ATP type, and NAD+ - dependent are found in bacteria (e.g. E. coli).
{{cite journal}}: CS1 maint: DOI inactive as of April 2024 (link)