
Adenocarcinoma is a type of cancerous tumor that can occur in several parts of the body. It is defined as neoplasia of epithelial tissue that has glandular origin, glandular characteristics, or both. Adenocarcinomas are part of the larger grouping of carcinomas, but are also sometimes called by more precise terms omitting the word, where these exist. Thus invasive ductal carcinoma, the most common form of breast cancer, is adenocarcinoma but does not use the term in its name—however, esophageal adenocarcinoma does to distinguish it from the other common type of esophageal cancer, esophageal squamous cell carcinoma. Several of the most common forms of cancer are adenocarcinomas, and the various sorts of adenocarcinoma vary greatly in all their aspects, so that few useful generalizations can be made about them.
This is a list of terms related to oncology. The original source for this list was the US National Cancer Institute's public domain Dictionary of Cancer Terms.
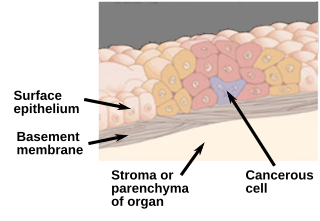
Carcinoma in situ (CIS) is a group of abnormal cells. While they are a form of neoplasm, there is disagreement over whether CIS should be classified as cancer. This controversy also depends on the exact CIS in question. Some authors do not classify them as cancer, however, recognizing that they can potentially become cancer. Others classify certain types as a non-invasive form of cancer. The term "pre-cancer" has also been used.
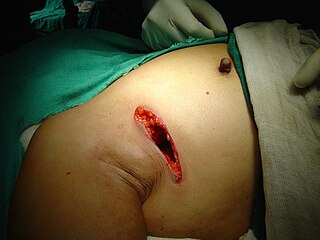
Lumpectomy is a surgical removal of a discrete portion or "lump" of breast tissue, usually in the treatment of a malignant tumor or breast cancer. It is considered a viable breast conservation therapy, as the amount of tissue removed is limited compared to a full-breast mastectomy, and thus may have physical and emotional advantages over more disfiguring treatment. Sometimes a lumpectomy may be used to either confirm or rule out that cancer has actually been detected. A lumpectomy is usually recommended to patients whose cancer has been detected early and who do not have enlarged tumors. Although a lumpectomy is used to allow for most of the breast to remain intact, the procedure may result in adverse affects that can include sensitivity and result in scar tissue, pain, and possible disfiguration of the breast if the lump taken out is significant. According to National Comprehensive Cancer Network guidelines, lumpectomy may be performed for ductal carcinoma in situ (DCIS), invasive ductal carcinoma, or other conditions.

Invasive carcinoma of no special type, invasive breast carcinoma of no special type (IBC-NST), invasive ductal carcinoma (IDC), infiltrating ductal carcinoma (IDC) or invasive ductal carcinoma, not otherwise specified (NOS) is a disease. For international audiences this article will use "invasive carcinoma NST" because it is the preferred term of the World Health Organization (WHO).

Ductal carcinoma in situ (DCIS), also known as intraductal carcinoma, is a pre-cancerous or non-invasive cancerous lesion of the breast. DCIS is classified as Stage 0. It rarely produces symptoms or a breast lump one can feel, typically being detected through screening mammography. It has been diagnosed in a significant percentage of men.

Endometrial intraepithelial neoplasia (EIN) is a premalignant lesion of the uterine lining that predisposes to endometrioid endometrial adenocarcinoma. It is composed of a collection of abnormal endometrial cells, arising from the glands that line the uterus, which have a tendency over time to progress to the most common form of uterine cancer—endometrial adenocarcinoma, endometrioid type.

Fibrocystic breast changes is a condition of the breasts where there may be pain, breast cysts, and breast masses. The breasts may be described as "lumpy" or "doughy". Symptoms may worsen during certain parts of the menstrual cycle due to hormonal stimulation. These are normal breast changes, not associated with cancer.
Atypical hyperplasia is a benign (noncancerous) cellular hyperplasia in which cells show some atypia. In this condition, cells look abnormal under a microscope and are increased in number.

Medullary breast carcinoma is a rare type of breast cancer that is characterized as a relatively circumscribed tumor with pushing, rather than infiltrating, margins. It is histologically characterized as poorly differentiated cells with abundant cytoplasm and pleomorphic high grade vesicular nuclei. It involves lymphocytic infiltration in and around the tumor and can appear to be brown in appearance with necrosis and hemorrhage. Prognosis is measured through staging but can often be treated successfully and has a better prognosis than other infiltrating breast carcinomas.
Comedocarcinoma is a kind of breast cancer that demonstrates comedonecrosis, which is the central necrosis of cancer cells within involved ducts. Comedocarcinomas are usually non-infiltrating and intraductal tumors, characterized as a comedo-type, high-grade ductal carcinoma in situ (DCIS). However, there have been accounts of comedocarcinoma which has then diversified into other cell types and developed into infiltrating (invasive) ductal carcinoma. Recurrence and survival rates differ for invasive breast cancer which has originated as comedocarcinoma compared with other types of cancer cells.

High-grade prostatic intraepithelial neoplasia (HGPIN) is an abnormality of prostatic glands and believed to precede the development of prostate adenocarcinoma.
Breast cancer classification divides breast cancer into categories according to different schemes criteria and serving a different purpose. The major categories are the histopathological type, the grade of the tumor, the stage of the tumor, and the expression of proteins and genes. As knowledge of cancer cell biology develops these classifications are updated.

Male breast cancer (MBC) is a cancer in males that originates in their breasts. Males account for less than 1% of new breast cancers with about 20,000 new cases being diagnosed worldwide every year. Its incidence rates in males vs. females are, respectively, 0.4 and 66.7 per 100,000 person-years. The worldwide incidences of male as well as female breast cancers have been increasing over the last few decades. Currently, one of every 800 men are estimated to develop this cancer during their lifetimes.
The Bethesda system (TBS), officially called The Bethesda System for Reporting Cervical Cytology, is a system for reporting cervical or vaginal cytologic diagnoses, used for reporting Pap smear results. It was introduced in 1988 and revised in 1991, 2001, and 2014. The name comes from the location of the conference, sponsored by the National Institutes of Health, that established the system.

Invasive lobular carcinoma (ILC) is breast cancer arising from the lobules of the mammary glands. It accounts for 5–10% of invasive breast cancer. Rare cases of this carcinoma have been diagnosed in men.

Atypical ductal hyperplasia (ADH) is the term used for a benign lesion of the breast that indicates an increased risk of breast cancer.
A preventive mastectomy or prophylactic mastectomy or risk-reducing mastectomy (RRM) is an elective operation to remove the breasts so that the risk of breast cancer is reduced.
Hereditary lobular breast cancer is a rare inherited cancer predisposition associated with pathogenic CDH1 (gene) germline mutations, and without apparent correlation with the hereditary diffuse gastric cancer syndrome. Research studies identified novel CDH1 germline variants in women with diagnosed lobular breast cancer and without any family history of gastric carcinoma. Firstly, in 2018 Giovanni Corso et al. defined this syndrome as a new cancer predisposition and the Authors suggested additional clinical criteria to testing CDH1 in lobular breast cancer patients. In 2020, the International Gastric Cancer Linkage Consortium recognized officially that the hereditary lobular breast cancer is a possible independent syndrome. To date, there are reported about 40 families clustering for lobular breast cancer and associated with CDH1 germline mutations but without association with diffuse gastric cancer. Other recent studies demonstrated a possible correlation between hereditary lobular breast cancer and gastric cancer risk.
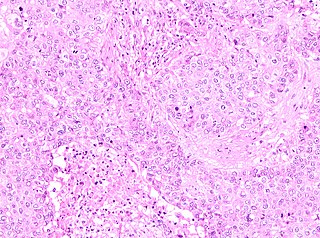
Pure apocrine carcinoma of the breast (PACB) is a rare carcinoma derived from the epithelial cells in the lactiferous ducts of the mammary gland. The mammary gland is an apocrine gland. Its lactiferous ducts have two layers of epithelial cells, a luminal layer which faces the duct's lumen and a basal layer which lies beneath the luminal layer. There are at least 4 subtypes of epithelial cells in these ducts: luminal progenitor cells and luminal mature cells which reside in the luminal layer and mammary stem cells and basal cells which reside in the basal layer. Examination of the genes expressed in PACB cancer cells indicate that most of these tumors consist of cells derived from luminal cells but a minority of these tumors consist of cells derived from basal cells.















