Related Research Articles

Acute disseminated encephalomyelitis (ADEM), or acute demyelinating encephalomyelitis, is a rare autoimmune disease marked by a sudden, widespread attack of inflammation in the brain and spinal cord. As well as causing the brain and spinal cord to become inflamed, ADEM also attacks the nerves of the central nervous system and damages their myelin insulation, which, as a result, destroys the white matter. The cause is often a trigger such as from viral infection or vaccinations.
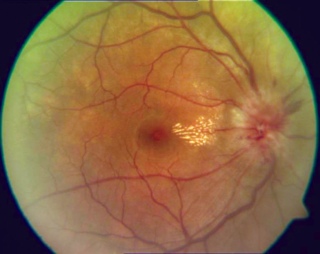
Optic neuritis describes any condition that causes inflammation of the optic nerve; it may be associated with demyelinating diseases, or infectious or inflammatory processes.
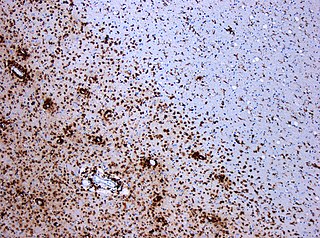
Multiplesclerosis (MS) is an autoimmune disease in which the insulating covers of nerve cells in the brain and spinal cord are damaged. This damage disrupts the ability of parts of the nervous system to transmit signals, resulting in a range of signs and symptoms, including physical, mental, and sometimes psychiatric problems. Specific symptoms can include double vision, vision loss, eye pain, muscle weakness, and loss of sensation or coordination. MS takes several forms, with new symptoms either occurring in isolated attacks or building up over time. In the relapsing forms of MS, between attacks, symptoms may disappear completely, although some permanent neurological problems often remain, especially as the disease advances. In the progressive forms of MS, bodily function slowly deteriorates and disability worsens once symptoms manifest and will steadily continue to do so if the disease is left untreated.

Interferon beta-1a is a cytokine in the interferon family used to treat multiple sclerosis (MS). It is produced by mammalian cells, while interferon beta-1b is produced in modified E. coli. Some research indicates that interferon injections may result in an 18–38% reduction in the rate of MS relapses.
Neuromyelitis optica spectrum disorders (NMOSD) are a spectrum of autoimmune diseases characterized by acute inflammation of the optic nerve and the spinal cord (myelitis). Episodes of ON and myelitis can be simultaneous or successive. A relapsing disease course is common, especially in untreated patients.
Interferon beta-1b is a cytokine in the interferon family used to treat the relapsing-remitting and secondary-progressive forms of multiple sclerosis (MS). It is approved for use after the first MS event. Closely related is interferon beta 1a, also indicated for MS, with a very similar drug profile.

Myelin oligodendrocyte glycoprotein (MOG) is a glycoprotein believed to be important in the myelination of nerves in the central nervous system (CNS). In humans this protein is encoded by the MOG gene. It is speculated to serve as a necessary "adhesion molecule" to provide structural integrity to the myelin sheath and is known to develop late on the oligodendrocyte.
Experimental autoimmune encephalomyelitis, sometimes experimental allergic encephalomyelitis (EAE), is an animal model of brain inflammation. It is an inflammatory demyelinating disease of the central nervous system (CNS). It is mostly used with rodents and is widely studied as an animal model of the human CNS demyelinating diseases, including multiple sclerosis (MS) and acute disseminated encephalomyelitis (ADEM). EAE is also the prototype for T-cell-mediated autoimmune disease in general.
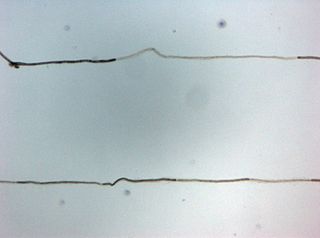
Chronic inflammatory demyelinating polyneuropathy (CIDP) is an acquired autoimmune disease of the peripheral nervous system characterized by progressive weakness and impaired sensory function in the legs and arms. The disorder is sometimes called chronic relapsing polyneuropathy (CRP) or chronic inflammatory demyelinating polyradiculoneuropathy. CIDP is closely related to Guillain–Barré syndrome and it is considered the chronic counterpart of that acute disease. Its symptoms are also similar to progressive inflammatory neuropathy. It is one of several types of neuropathy.

Multiple sclerosis is an inflammatory demyelinating disease of the CNS in which activated immune cells invade the central nervous system and cause inflammation, neurodegeneration, and tissue damage. The underlying cause is currently unknown. Current research in neuropathology, neuroimmunology, neurobiology, and neuroimaging, together with clinical neurology, provide support for the notion that MS is not a single disease but rather a spectrum.
Multiple sclerosis (MS) is a chronic inflammatory demyelinating disease that affects the central nervous system (CNS). Several therapies for it exist, although there is no known cure.
Inflammatory demyelinating diseases (IDDs), sometimes called Idiopathic (IIDDs) due to the unknown etiology of some of them, are a heterogenous group of demyelinating diseases - conditions that cause damage to myelin, the protective sheath of nerve fibers - that occur against the background of an acute or chronic inflammatory process. IDDs share characteristics with and are often grouped together under Multiple Sclerosis. They are sometimes considered different diseases from Multiple Sclerosis, but considered by others to form a spectrum differing only in terms of chronicity, severity, and clinical course.

Tumefactive multiple sclerosis is a condition in which the central nervous system of a person has multiple demyelinating lesions with atypical characteristics for those of standard multiple sclerosis (MS). It is called tumefactive as the lesions are "tumor-like" and they mimic tumors clinically, radiologically and sometimes pathologically.

Current standards for diagnosing multiple sclerosis (MS) are based on the 2018 revision of McDonald criteria. They rely on MRI detection of demyelinating lesions in the CNS, which are distributed in space (DIS) and in time (DIT). It is also a requirement that any possible known disease that produces demyelinating lesions is ruled out before applying McDonald's criteria.

Stephen L. Hauser is a professor of the Department of Neurology at the University of California, San Francisco (UCSF) specializing in immune mechanisms and multiple sclerosis (MS). He has contributed to the establishment of consortia that have identified more than 50 gene variants that contribute to MS risk.

Multiple sclerosis (MS) can be pathologically defined as the presence of distributed glial scars (scleroses) in the central nervous system that must show dissemination in time (DIT) and in space (DIS) to be considered MS lesions.
MOG antibody disease (MOGAD) or MOG antibody-associated encephalomyelitis (MOG-EM) is an inflammatory demyelinating disease of the central nervous system. Serum anti-myelin oligodendrocyte glycoprotein antibodies are present in up to half of patients with an acquired demyelinating syndrome and have been described in association with a range of phenotypic presentations, including acute disseminated encephalomyelitis, optic neuritis, transverse myelitis, and neuromyelitis optica.
There are several ways for pharmaceuticals for treating multiple sclerosis (MS) to reach the market.
Anti-AQP4 diseases, are a group of diseases characterized by auto-antibodies against aquaporin 4.
Brenda Banwell is Chief of the Division of Neurology and Co-Director of the Neuroscience Center, and Professor of Neurology at Children's Hospital of Philadelphia and holder of the Grace R. Loeb Endowed Chair in Neurosciences. She also holds the title of Professor of Pediatrics and Neurology at the Perelman School of Medicine at the University of Pennsylvania.
References
- 1 2 Cohen JA (July 2009). "Emerging therapies for relapsing multiple sclerosis". Arch. Neurol. 66 (7): 821–8. doi:10.1001/archneurol.2009.104. PMID 19597083.
- ↑ Kazutoshi Sato Douglas; et al. (Feb 2014). "Distinction between MOG antibody-positive and AQP4 antibody-positive NMO spectrum disorders". Neurology. 82 (6): 474–481. doi:10.1212/WNL.0000000000000101. PMC 3937859 . PMID 24415568.
- ↑ Gahr M, Lauda F, Wigand ME, Connemann BJ, Rosenbohm A, Tumani H, Reindl M, Uzelac Z, Lewerenz J (2015). "Periventricular white matter lesion and incomplete MRZ reaction in a male patient with anti-N-methyl-D-aspartate receptor encephalitis presenting with dysphoric mania". BMJ Case Reports. 2015: bcr2014209075. doi:10.1136/bcr-2014-209075. PMC 4422915 . PMID 25917068.
- ↑ Li Y, Xie P, Lv F, et al. (2008). "Brain magnetic resonance imaging abnormalities in neuromyelitis optica". Acta Neurologica Scandinavica. 118 (4): 218–25. doi:10.1111/j.1600-0404.2008.01012.x. PMID 18384459. S2CID 22270592.
- ↑ Reindl M, Di Pauli F, Rostásy K, Berger T (Aug 2013). "The spectrum of MOG autoantibody-associated demyelinating diseases". Nature Reviews Neurology. 9 (8): 455–61. doi:10.1038/nrneurol.2013.118. PMID 23797245. S2CID 7219279.
- ↑ Jarius S, Metz I, König FB, Ruprecht K, Reindl M, Paul F, Brück W, Wildemann B (Feb 2016). "Screening for MOG-IgG and 27 other anti-glial and anti-neuronal autoantibodies in 'pattern II multiple sclerosis' and brain biopsy findings in a MOG-IgG-positive case". Multiple Sclerosis Journal. 22 (12): 1541–1549. doi:10.1177/1352458515622986. PMID 26869529. S2CID 1387384.
- 1 2 Spadaro M, et al. (2016). "Autoantibodies to MOG in a distinct subgroup of adult multiple sclerosis". Neurology: Neuroimmunology & Neuroinflammation. 3 (5): e257. doi:10.1212/NXI.0000000000000257. PMC 4949775 . PMID 27458601.
- ↑ Kitagawa S, Osada T, Kaneko K, Takahashi T, Suzuki N, Nakahara J (Nov 2018). "Clinical analysis of opticospinal multiple sclerosis (OSMS) presentation detecting anti-myelin oligodendrocyte glycoprotein (MOG) antibody". Rinsho Shinkeigaku. 58 (12): 737–744. doi: 10.5692/clinicalneurol.cn-001184 . PMID 30487359.
- ↑ Ikeda, Ken; Ito, Hirono; Hidaka, Takanobu; Takazawa, Takanori; Sekine, Tokinori; Yoshii, Yasuhiro; Hirayama, Takehisa; Kawabe, Kiyokazu; Kano, Osamu; Iwasaki, Yasuo (2011). "Repeated Non-enhancing Tumefactive Lesions in a Patient with a Neuromyelitis Optica Spectrum Disorder". Internal Medicine. 50 (9): 1061–1064. doi: 10.2169/internalmedicine.50.4295 . PMID 21532234.
- ↑ Pröbstel AK, et al. (Mar 2015). "Anti-MOG antibodies are present in a subgroup of patients with a neuromyelitis optica phenotype". Journal of Neuroinflammation. 12 (1): 46. doi: 10.1186/s12974-015-0256-1 . PMC 4359547 . PMID 25889963.
- ↑ Spadaro Melania; et al. (2015). "Histopathology and clinical course of MOG-antibody-associated encephalomyelitis". Annals of Clinical and Translational Neurology. 2 (3): 295–301. doi:10.1002/acn3.164. PMC 4369279 . PMID 25815356.
- ↑ Ya Y, et al. (2015). "Autoantibody to MOG suggests two distinct clinical subtypes of NMOSD". Science China Life Sciences. 59 (12): 1270–1281. doi:10.1007/s11427-015-4997-y. PMC 5101174 . PMID 26920678.
- ↑ Marcus Vinicius, Magno Goncalves, Yara Dadalti Fragoso, The involvement of anti-neurofascin 155 antibodies in central and peripheral demyelinating diseases, Neuroimmunol Neuroinflammation, 8 Apr 2019;6:6.10.20517/2347-8659.2019.08
- 1 2 3 4 Kira, Jun-Ichi; Yamasaki, Ryo; Ogata, Hidenori (2019). "Anti-neurofascin autoantibody and demyelination". Neurochemistry International. 130: 104360. doi: 10.1016/j.neuint.2018.12.011 . PMID 30582947.
- ↑ Stich O, Perera S, Berger B, Jarius S, Wildemann B, Baumgartner A, Rauer S (March 2016). "Prevalence of neurofascin-155 antibodies in patients with multiple sclerosis". Journal of the Neurological Sciences. 364: 29–32. doi:10.1016/j.jns.2016.03.004. PMID 27084211. S2CID 29204735.
- ↑ "Progressive MS: a new perspective | Multiple Sclerosis Society UK". www.mssociety.org.uk. Archived from the original on 2007-06-21.
- ↑ Engel S, Luessi F, Mueller A, Schopf RE, Zipp F, Bittner S (2020). "PPMS onset upon adalimumab treatment extends the spectrum of anti-TNF-α therapy-associated demyelinating disorders". Therapeutic Advances in Neurological Disorders. 13: 175628641989515. doi: 10.1177/1756286419895155 . PMC 6940603 . PMID 31921355.
- ↑ Alnasser Alsukhni Rana; Jriekh Ziena; Aboras Yasmin (2016). "Adalimumab Induced or Provoked MS in Patient with Autoimmune Uveitis: A Case Report and Review of the Literature". Case Reports in Medicine. 2016: 1–6. doi: 10.1155/2016/1423131 . PMC 5093248 . PMID 27840642.
- 1 2 Vicente L. Gómez; Viqueira B. Rubio; Cobos R. Garcia; Moreno J. Pardo; Gonzalez R. Arroyo (2016). "P04.07 Relapse in a paucisymptomatic form of multiple sclerosis in a patient treated with nivolumab". Neuro-Oncology. 18: iv25. doi: 10.1093/neuonc/now188.085 . PMC 5782583 .
- 1 2 3 Höftberger R, Leisser M, Bauer J, Lassmann H (Dec 2015). "Autoimmune encephalitis in humans: how closely does it reflect multiple sclerosis?". Acta Neuropathologica Communications. 3 (1): 80. doi: 10.1186/s40478-015-0260-9 . PMC 4670499 . PMID 26637427.
- ↑ Marzia Anita Lucia Romeo et al, Multiple sclerosis associated with pembrolizumab in a patient with non-small cell lung cancer, Journal of Neurology, pp 1–4, 04 October 2019
- 1 2 Lassman Hans (Feb 2010). "Acute disseminated encephalomyelitis and multiple sclerosis". Brain. 133 (2): 317–319. doi: 10.1093/brain/awp342 . PMID 20129937.
- ↑ Kalinowska-Lyszczarz, Alicja; Fereidan-Esfahani, Mahboobeh; Guo, Yong; Lucchinetti, Claudia F; Tobin, W Oliver (December 17, 2019). "Pathological findings in central nervous system demyelination associated with infliximab". Multiple Sclerosis Journal. 26 (9): 1124–1129. doi:10.1177/1352458519894710. PMC 7297659 . PMID 31845616.
- ↑ Young NP, Weinshenker BG, Parisi JE, Scheithauer B, Giannini C, Roemer SF, Thomsen KM, Mandrekar JN, Erickson BJ, Lucchinetti CF (2010). "Perivenous demyelination: association with clinically defined acute disseminated encephalomyelitis and comparison with pathologically confirmed multiple sclerosis". Brain. 133 (2): 333–348. doi:10.1093/brain/awp321. PMC 2822631 . PMID 20129932.
- ↑ Bargiela, David; Chinnery, Patrick F (September 25, 2019). "Mitochondria in neuroinflammation – Multiple sclerosis (MS), leber hereditary optic neuropathy (LHON) and LHON-MS". Neuroscience Letters. 710: 132932. doi: 10.1016/j.neulet.2017.06.051 . PMID 28668384. S2CID 38817368.
- ↑ Nikoskelainen EK, Marttila RJ, Huoponen K, et al. (August 1995). "Leber's "plus": neurological abnormalities in patients with Leber's hereditary optic neuropathy". J. Neurol. Neurosurg. Psychiatry. 59 (2): 160–4. doi:10.1136/jnnp.59.2.160. PMC 485991 . PMID 7629530.
- ↑ "cardiac arrythmia". Archived from the original on 2020-01-30. Retrieved 2020-02-05.
- ↑ "Mayo Clinic: Multiple Sclerosis".
- ↑ "FDA approves new oral treatment for multiple sclerosis". fda.gov. Retrieved 2019-05-11.
- 1 2 3 4 5 6 Miller AE (2011). "Multiple sclerosis: where will we be in 2020?". Mt. Sinai J. Med. 78 (2): 268–79. doi: 10.1002/msj.20242 . PMID 21425270.
- ↑ Jeffrey, susan (9 Aug 2012). "CONCERTO: A Third Phase 3 Trial for Laquinimod in MS". Medscape Medical News. Retrieved 21 May 2013.
- ↑ He, Dian; Han, Kai; Gao, Xiangdong; Dong, Shuai; Chu, Lan; Feng, ZhanHui; Wu, Shan (2013-08-06). Chu, Lan (ed.). "Laquinimod for multiple sclerosis". The Cochrane Database of Systematic Reviews (8): CD010475. doi:10.1002/14651858.CD010475.pub2. ISSN 1469-493X. PMID 23922214.
- ↑ Sicotte, Nancy L.; Liva, Stephanie M.; Klutch, Rochelle; Pfeiffer, Paul; Bouvier, Seth; Odesa, Sylvia; Wu, T. C. Jackson; Voskuhl, Rhonda R. (2002-10-01). "Treatment of multiple sclerosis with the pregnancy hormone estriol". Annals of Neurology. 52 (4): 421–428. doi:10.1002/ana.10301. ISSN 1531-8249. PMID 12325070. S2CID 5000678.
- ↑ Gold, Stefan M.; Voskuhl, Rhonda R. (2009). "Estrogen treatment in multiple sclerosis". Journal of the Neurological Sciences. 286 (1–2): 99–103. doi:10.1016/j.jns.2009.05.028. PMC 2760629 . PMID 19539954.
- ↑ Voskuhl, Rhonda R; Wang, HeJing; Wu, T C Jackson; Sicotte, Nancy L; Nakamura, Kunio; Kurth, Florian; Itoh, Noriko; Bardens, Jenny; Bernard, Jacqueline T (2016). "Estriol combined with glatiramer acetate for women with relapsing-remitting multiple sclerosis: a randomised, placebo-controlled, phase 2 trial". The Lancet Neurology. 15 (1): 35–46. doi:10.1016/s1474-4422(15)00322-1. PMID 26621682. S2CID 30418205.
- ↑ Mendoza, RL (2014). "Pharmacoeconomics and clinical trials in multiple sclerosis: baseline data from the European Union". Journal of Public Health. 22 (3): 211–218. doi:10.1007/s10389-013-0561-z. S2CID 25533962.
- ↑ Kieseier, BC; Calabresi, PA (March 2012). "PEGylation of interferon-β-1a: a promising strategy in multiple sclerosis". CNS Drugs. 26 (3): 205–14. doi:10.2165/11596970-000000000-00000. PMID 22201341. S2CID 34290702.
- ↑ "Biogen Idec Announces Positive Top-Line Results from Phase 3 Study of Peginterferon Beta-1a in Multiple Sclerosis" (Press release). Biogen Idec. 2013-01-24. Archived from the original on 2013-10-04. Retrieved 2013-05-21.
- ↑ "Drug Approval Package: Plegridy (peginterferon beta-1a) prefilled syringe) NDA #125499". U.S. Food and Drug Administration (FDA). 17 September 2014. Retrieved 30 March 2020.
- ↑ Xiao, Yousheng; Huang, Jianyi; Luo, Hongye; Wang, Jin (2014-02-07). "Mycophenolate mofetil for relapsing-remitting multiple sclerosis". The Cochrane Database of Systematic Reviews (2): CD010242. doi:10.1002/14651858.CD010242.pub2. ISSN 1469-493X. PMID 24505016.
- ↑ Gray, O.; McDonnell, G. V.; Forbes, R. B. (2003). "Intravenous immunoglobulins for multiple sclerosis". The Cochrane Database of Systematic Reviews. 2010 (4): CD002936. doi:10.1002/14651858.CD002936. ISSN 1469-493X. PMC 8407393 . PMID 14583956.
- ↑ Saidha S, Eckstein C, Calabresi PA (January 2012). "New and emerging disease modifying therapies for multiple sclerosis". Annals of the New York Academy of Sciences. 1247 (1): 117–37. Bibcode:2012NYASA1247..117S. doi:10.1111/j.1749-6632.2011.06272.x. PMID 22224673. S2CID 10837693.
- ↑ Kappos, Ludwig; Wiendl, Heinz; Selmaj, Krzysztof; Arnold, Douglas L.; Havrdova, Eva; Boyko, Alexey; Kaufman, Michael; Rose, John; Greenberg, Steven; Sweetser, Marianne; Riester, Katherine; o'Neill, Gilmore; Elkins, Jacob (2015). "Daclizumab HYP versus Interferon Beta-1a in Relapsing Multiple Sclerosis". New England Journal of Medicine. 373 (15): 1418–1428. doi: 10.1056/nejmoa1501481 . PMID 26444729.
- 1 2 3 4 Milo R, Panitch H (February 2011). "Combination therapy in multiple sclerosis". J. Neuroimmunol. 231 (1–2): 23–31. doi:10.1016/j.jneuroim.2010.10.021. PMID 21111490. S2CID 31753224.
- ↑ Luessi F, Siffrin V, Zipp F (September 2012). "Neurodegeneration in multiple sclerosis: novel treatment strategies". Expert Rev Neurother. 12 (9): 1061–76, quiz 1077. doi: 10.1586/ern.12.59 . PMID 23039386.
- ↑ Lee Mendoza R (2014). "Pharmacoeconomics and clinical trials in multiple sclerosis: baseline data from the European Union". Journal of Public Health. 22 (3): 211–218. doi:10.1007/s10389-013-0561-z. S2CID 25533962.
- ↑ "Opexa shares lose most of value on study data". Forbes .[ permanent dead link ]
- ↑ "Opexa Therapeutics Announces Completion Of Mid Study Descriptive Analysis On Phase IIb Trial Of Tovaxin". Archived from the original on 2009-01-03. Retrieved 2008-03-14.
- ↑ "Opexa Therapeutics - Home". www.opexatherapeutics.com.
- ↑ "Significant Advances in Multiple Sclerosis Treatment". Pharmacy Times. Archived from the original on August 19, 2016.
- ↑ Gladstone DE, Zamkoff KW, Krupp L, et al. (2006). "High-dose cyclophosphamide for moderate to severe refractory multiple sclerosis". Arch. Neurol. 63 (10): 1388–93. doi:10.1001/archneur.63.10.noc60076. PMID 16908728.
- ↑ Zipoli V, Portaccio E, Hakiki B, Siracusa G, Sorbi S, Pia Amato M (2007). "Intravenous mitoxantrone and cyclophosphamide as second-line therapy in multiple sclerosis: An open-label comparative study of efficacy and safety". Journal of the Neurological Sciences. 266 (1–2): 25–30. doi:10.1016/j.jns.2007.08.023. PMID 17870094. S2CID 24283817.
- ↑ Krishnan C, Kaplin AI, Brodsky RA, et al. (June 2008). "Reduction of Disease Activity and Disability With High-Dose Cyclophosphamide in Patients With Aggressive Multiple Sclerosis". Arch. Neurol. 65 (8): 1044–51. doi:10.1001/archneurol.65.8.noc80042. PMC 2574697 . PMID 18541787.
- ↑ Chataway, J (2014). "Effect of high-dose simvastatin on brain atrophy and disability in secondary progressive multiple sclerosis (MS-STAT): a randomised, placebo-controlled, phase 2 trial". The Lancet. 383 (9936): 2213–2221. doi: 10.1016/s0140-6736(13)62242-4 . hdl: 10044/1/26298 . PMID 24655729.
- ↑ de Araújo EA, de Freitas MR (June 2008). "Benefit with methylprednisolone in continuous pulsetherapy in progressive primary form of multiple sclerosis: study of 11 cases in 11 years". Arq Neuropsiquiatr. 66 (2B): 350–3. doi: 10.1590/S0004-282X2008000300013 . PMID 18641870.
- ↑ Killestein J, Kalkers NF, Polman CH (June 2005). "Glutamate inhibition in MS: the neuroprotective properties of riluzole". J Neurol Sci. 233 (1–2): 113–5. doi:10.1016/j.jns.2005.03.011. PMID 15949499. S2CID 20607353.
- ↑ Gironi M, Martinelli-Boneschi F, Sacerdote P, Solaro C, Zaffaroni M, Cavarretta R, Moiola L, Bucello S, Radaelli M, Pilato V, Rodegher M, Cursi M, Franchi S, Martinelli V, Nemni R, Comi G, Martino G (2008). "A pilot trial of low-dose naltrexone in primary progressive multiple sclerosis". Multiple Sclerosis. 14 (8): 1076–83. doi:10.1177/1352458508095828. PMID 18728058. S2CID 3548490.
- ↑ Gajofatto A, Turatti M, Benedetti MD (2016). "Primary progressive multiple sclerosis: current therapeutic strategies and future perspectives". Expert Rev Neurother. 17 (4): 1–14. doi:10.1080/14737175.2017.1257385. PMID 27813441. S2CID 24034132.
- ↑ Castro-Borrero Wanda; et al. (2012). "Current and emerging therapies in multiple sclerosis: a systematic review". Therapeutic Advances in Neurological Disorders. 5 (4): 205–220. doi:10.1177/1756285612450936. PMC 3388530 . PMID 22783370.
- ↑ "Statin may slow progressive MS".[ permanent dead link ]
- ↑ Wang, Z (2016). "Nuclear Receptor NR1H3 in Familial Multiple Sclerosis". Neuron. 90 (5): 948–954. doi:10.1016/j.neuron.2016.04.039. PMC 5092154 . PMID 27253448.
- ↑ First Oral Treatment For Highly Active Relapsing Remitting Multiple Sclerosis Provides New Choice For UK Patients Failing On Injections,
- ↑ Weiner HL, Cohen JA (April 2002). "Treatment of multiple sclerosis with cyclophosphamide: critical review of clinical and immunologic effects". Mult. Scler. 8 (2): 142–54. doi:10.1191/1352458502ms790oa. PMID 11990872. S2CID 35767740.
- ↑ Margoni, Monica; Franciotta, Silvia; Poggiali, Davide; Riccardi, Alice; Rinaldi, Francesca; Nosadini, Margherita; Sartori, Stefano; Anglani, Maria Giulia; Causin, Francesco; Perini, Paola; Gallo, Paolo (5 March 2020). "Cerebellar gray matter lesions are common in pediatric multiple sclerosis at clinical onset". Journal of Neurology. 267 (6): 1824–1829. doi:10.1007/s00415-020-09776-6. PMID 32140864. S2CID 212404981.
- ↑ Eshaghi, Arman (4 March 2020). "First approved treatment in children with multiple sclerosis slows brain atrophy". Journal of Neurology, Neurosurgery & Psychiatry. 91 (5): jnnp-2019-322519. doi:10.1136/jnnp-2019-322519. PMID 32132223. S2CID 212416856.
- ↑ Matthews, Paul M. (2015). "New drugs and personalized medicine for multiple sclerosis". Nature Reviews Neurology. 11 (11): 614–616. doi:10.1038/nrneurol.2015.200. PMID 26503926. S2CID 40121167.
- 1 2 Hottenrott, T.; Dersch, R.; Berger, B.; Rauer, S.; Eckenweiler, M.; Huzly, D.; Stich, O. (2015). "The intrathecal, polyspecific antiviral immune response in neurosarcoidosis, acute disseminated encephalomyelitis and autoimmune encephalitis compared to multiple sclerosis in a tertiary hospital cohort". Fluids and Barriers of the CNS. 12: 27. doi: 10.1186/s12987-015-0024-8 . PMC 4677451 . PMID 26652013.
- ↑ Duranti, Fabio; Pieri, Massimo; Zenobi, Rossella; Centonze, Diego; Buttari, Fabio; Bernardini, Sergio; Dessi, Mariarita. "kFLC Index: a novel approach in early diagnosis of Multiple Sclerosis". International Journal of Scientific Research. 4 (8). Archived from the original on 2016-08-28. Retrieved 2018-08-27.
- ↑ Laura A, Eero R, Juha OR (December 2015). "Imaging neuroinflammation in multiple sclerosis using TSPO-PET". Clinical and Translational Imaging. 3 (6): 461–473. doi:10.1007/s40336-015-0147-6. PMC 4887541 . PMID 27331049.
- ↑ "fda-drug-safety-communication-fda-warns-gadolinium-based-contrast-agents-gbcas-are-retained-body; requires new class warnings". USA FDA . 2018-05-16.
- ↑ Rudie, J. D.; Mattay, R. R.; Schindler, M.; Steingall, S.; Cook, T. S.; Loevner, L. A.; Schnall, M. D.; Mamourian, A. C.; Bilello, M. (2018-05-01). "Initiative to Reduce Unnecessary Gadolinium-Based Contrast in Multiple Sclerosis Patients". Journal of the American College of Radiology. 16 (9 Pt A): 1158–1164. doi:10.1016/j.jacr.2019.04.005. PMC 6732018 . PMID 31092348.
- ↑ Filippi, M.; Agosta, F. (2010-04-01). "Imaging biomarkers in multiple sclerosis". Journal of Magnetic Resonance Imaging. 31 (4): 770–88. doi:10.1002/jmri.22102. PMID 20373420. S2CID 15893040.
- 1 2 Cohen JA, Reingold SC, Polman CH, Wolinsky JS (May 2012). "Disability outcome measures in multiple sclerosis clinical trials: current status and future prospects". Lancet Neurol. 11 (5): 467–76. doi:10.1016/S1474-4422(12)70059-5. PMID 22516081. S2CID 19562110.
- ↑ Puthenparampil, Marco; Cazzola, Chiara; Zywicki, Sofia; Federle, Lisa; Stropparo, Erica; Anglani, Mariagiulia; Rinaldi, Francesca; Perini, Paola; Gallo, Paolo (2018). "NEDA-3 status including cortical lesions in the comparative evaluation of natalizumab versus fingolimod efficacy in multiple sclerosis". Therapeutic Advances in Neurological Disorders. 11: 175628641880571. doi:10.1177/1756286418805713. PMC 6204617 . PMID 30386435.
- ↑ Giovannoni, Gavin; Hawkes, Christopher H.; Lechner-Scott, Jeannette; Levy, Michael; Yeh, E. Ann (1 July 2022). "Multiple sclerosis is one disease". Multiple Sclerosis and Related Disorders. 63: 103961. doi:10.1016/j.msard.2022.103961. PMID 35714573.
- ↑ Pitt, David; Lo, Chih Hung; Gauthier, Susan A.; Hickman, Richard A.; Longbrake, Erin; Airas, Laura M.; Mao-Draayer, Yang; Riley, Claire; Jager, Philip Lawrence De; Wesley, Sarah; Boster, Aaron; Topalli, Ilir; Bagnato, Francesca; Mansoor, Mohammad; Stuve, Olaf; Kister, Ilya; Pelletier, Daniel; Stathopoulos, Panos; Dutta, Ranjan; Lincoln, Matthew R. (1 November 2022). "Toward Precision Phenotyping of Multiple Sclerosis". Neurology: Neuroimmunology & Neuroinflammation. 9 (6): e200025. doi: 10.1212/NXI.0000000000200025 . PMC 9427000 . PMID 36041861.
- 1 2 3 Dobson, R.; Giovannoni, G. (2019). "Multiple sclerosis - a review". European Journal of Neurology. 26 (1): 27–40. doi: 10.1111/ene.13819 . PMC 1589931 . PMID 30300457.
- ↑ Lublin, F. D.; Reingold, S. C.; Cohen, J. A.; Cutter, G. R.; Sorensen, P. S.; Thompson, A. J.; Wolinsky, J. S.; Balcer, L. J.; Banwell, B.; Barkhof, F.; Bebo, B.; Calabresi, P. A.; Clanet, M.; Comi, G.; Fox, R. J.; Freedman, M. S.; Goodman, A. D.; Inglese, M.; Kappos, L.; Kieseier, B. C.; Lincoln, J. A.; Lubetzki, C.; Miller, A. E.; Montalban, X.; O'Connor, P. W.; Petkau, J.; Pozzilli, C.; Rudick, R. A.; Sormani, M. P.; et al. (2014). "Defining the clinical course of multiple sclerosis: The 2013 revisions". Neurology. 83 (3): 278–286. doi:10.1212/WNL.0000000000000560. PMC 4117366 . PMID 24871874.
- ↑ Commissioner, Office of the (March 24, 2020). "FDA approves new oral treatment for multiple sclerosis". FDA.
- ↑ "Mavenclad | European Medicines Agency". 17 September 2018.
- ↑ Baecher-Allan, C.; Kaskow, B. J.; Weiner, H. L. (Feb 2018). "Multiple Sclerosis: Mechanisms and Immunotherapy". Neuron. 97 (4): 742–768. doi: 10.1016/j.neuron.2018.01.021 . PMID 29470968. S2CID 3499974.
- ↑ Díaz C, Zarco LA, Rivera DM, Highly active multiple sclerosis: An update, Mult Scler Relat Disord. 2019 May;30:215-224. doi: 10.1016/j.msard.2019.01.039. Epub 2019 Jan 24
- ↑ Michael P Pender; Scott R Burrows (31 October 2014). "Epstein–Barr virus and multiple sclerosis: potential opportunities for immunotherapy". Clin Transl Immunol. 3 (10): e27. doi:10.1038/cti.2014.25. PMC 4237030 . PMID 25505955.
- ↑ Bjornevik K, Cortese M, Healy BC, Kuhle J, Mina MJ, Leng Y, et al. (2022-01-21). "Longitudinal analysis reveals high prevalence of Epstein-Barr virus associated with multiple sclerosis". Science. 375 (6578): 296–301. Bibcode:2022Sci...375..296B. doi:10.1126/science.abj8222. PMID 35025605. S2CID 245983763.
- ↑ Kolata, Gina (13 January 2022). "Common Virus May Play Role in Debilitating Neurological Illness". The New York Times . Archived from the original on 14 January 2022.
- ↑ Robinson, WH; Steinman, L (13 January 2022). "Epstein-Barr virus and multiple sclerosis". Science. 375 (6578): 264–265. Bibcode:2022Sci...375..264R. doi: 10.1126/science.abm7930 . PMID 35025606. S2CID 245978874.
- ↑ Fleischer Vinzenz; et al. (2016). "Metabolic Patterns in Chronic Multiple Sclerosis Lesions and Normal-appearing White Matter: Intraindividual Comparison by Using 2D MR Spectroscopic Imaging". Radiology. 281 (2): 536–543. doi:10.1148/radiol.2016151654. PMID 27243371.
- ↑ Durhan, G.; Diker, S.; Has, A. C.; Karakaya, J.; Kurne, A. T.; Oguz, K. K. (2016). "Influence of cigarette smoking on white matter in patients with clinically isolated syndrome as detected by diffusion tensor imaging". Diagnostic and Interventional Radiology (Ankara, Turkey). 22 (3): 291–296. doi:10.5152/dir.2015.15415. PMC 4859748 . PMID 27015443.
- ↑ Mayer, Jonathan D. (March 1981). "Geographical Clues about Multiple Sclerosis". Annals of the Association of American Geographers. 71: 28–39. doi:10.1111/j.1467-8306.1981.tb01338.x.
- ↑ Ichiro Nakashima (December 2015). "Anti-myelin oligodendrocyte glycoprotein antibody in demyelinating diseases". Neuroimmunology. 6 (S1): 59–63. doi: 10.1111/cen3.12262 .
- 1 2 Misu, Tatsuro; Fujihara, Kazuo (2019). "Neuromyelitis optica spectrum and myelin oligodendrocyte glycoprotein antibody‐related disseminated encephalomyelitis". Clinical and Experimental Neuroimmunology. 10: 9–17. doi:10.1111/cen3.12491.
- ↑ "Link Between Gut Flora and Multiple Sclerosis Discovered". NeuroscienceNews. 2018-10-11.
- ↑ Planas, Raquel; Santos, Radleigh; Tomas-Ojer, Paula; Cruciani, Carolina; Lutterotti, Andreas; Faigle, Wolfgang; Schaeren-Wiemers, Nicole; Espejo, Carmen; Eixarch, Herena; Pinilla, Clemencia; Martin, Roland; Sospedra, Mireia (2018). "GDP-l-fucose synthase is a CD4+ T cell–specific autoantigen in DRB3*02:02 patients with multiple sclerosis" (PDF). Science Translational Medicine. 10 (462): eaat4301. doi: 10.1126/scitranslmed.aat4301 . PMID 30305453. S2CID 52959112.
- ↑ Hendrickson, Megan. "Myelocortical multiple sclerosis: a subgroup of multiple sclerosis patients with spinal cord and cortical demyelination". Onlinelibrary.ectrims-congress.eu.
- ↑ Trapp, Bruce D.; Vignos, Megan; Dudman, Jessica; Chang, Ansi; Fisher, Elizabeth; Staugaitis, Susan M.; Battapady, Harsha; Mork, Sverre; Ontaneda, Daniel; Jones, Stephen E.; Fox, Robert J.; Chen, Jacqueline; Nakamura, Kunio; Rudick, Richard A. (2018). "Cortical neuronal densities and cerebral white matter demyelination in multiple sclerosis: A retrospective study". The Lancet Neurology. 17 (10): 870–884. doi:10.1016/S1474-4422(18)30245-X. PMC 6197820 . PMID 30143361.
- ↑ Popescu, Bogdan F. Gh.; Pirko, Istvan; Lucchinetti, Claudia F. (2013). "Pathology of Multiple Sclerosis". Continuum: Lifelong Learning in Neurology. 19 (4 Multiple Sclerosis): 901–921. doi:10.1212/01.CON.0000433291.23091.65. PMC 3915566 . PMID 23917093.
- ↑ Wekerle, Hartmut (15 February 2022). "Epstein–Barr virus sparks brain autoimmunity in multiple sclerosis". Nature. 603 (7900): 230–232. Bibcode:2022Natur.603..230W. doi:10.1038/d41586-022-00382-2. PMID 35169323. S2CID 246866517.
- ↑ Kremer, David; Gruchot, Joel; Weyers, Vivien; Oldemeier, Lisa; Göttle, Peter; Healy, Luke; Ho Jang, Jeong; Kang t. Xu, Yu; Volsko, Christina; Dutta, Ranjan; Trapp, Bruce D.; Perron, Hervé; Hartung, Hans-Peter; Küry, Patrick (2019). "PHERV-W envelope protein fuels microglial cell-dependent damage of myelinated axons in multiple sclerosis". Proceedings of the National Academy of Sciences. 116 (30): 15216–15225. Bibcode:2019PNAS..11615216K. doi: 10.1073/pnas.1901283116 . PMC 6660731 . PMID 31213545.
- ↑ Lisak, Robert P. (2019). "Human retrovirus pHEV-W envelope protein and the pathogenesis of multiple sclerosis". Proceedings of the National Academy of Sciences. 116 (30): 14791–14793. Bibcode:2019PNAS..11614791L. doi: 10.1073/pnas.1909786116 . PMC 6660775 . PMID 31289223.
- ↑ Hans-Peter Hartung et al, Efficacy and Safety of Temelimab, an Antibody Antagonist of the Human Endogenous Retrovirus Type-W env Protein, in Participants with Relapsing Remitting Multiple Sclerosis: A Double-Blind, Randomised, Placebo-Controlled Phase 2b Clinical Trial, The Lancet 17 May 2019 SSRN 3388820
- 1 2 3 Baranzini SE (June 2011). "Revealing the genetic basis of multiple sclerosis: are we there yet?". Current Opinion in Genetics & Development. 21 (3): 317–24. doi:10.1016/j.gde.2010.12.006. PMC 3105160 . PMID 21247752.
- ↑ Sawcer S.; Hellenthal G.; Pirinen M.; Spencer C.C.A.; Patsopoulos N. A.; Moutsianas L.; et al. (2011). "Genetic risk and a primary role for cell-mediated immune mechanisms in multiple sclerosis". Nature. 476 (7359): 214–219. Bibcode:2011Natur.476..214T. doi:10.1038/nature10251. PMC 3182531 . PMID 21833088.
- ↑ "Mutation Identified as Genetic Marker for Multiple Sclerosis". 2015-03-16.
- ↑ Frischer J.M.; Bramow S.; Dal-Bianco A.; Lucchinetti C.F.; Rauschka H.; et al. (2009). "The relation between inflammation and neurodegeneration in multiple sclerosis brains". Brain. 132 (5): 1175–89. doi:10.1093/brain/awp070. PMC 2677799 . PMID 19339255.
- ↑ Zurawski Jonathan; Lassmann Hans; Bakshi Rohit (2016). "Use of Magnetic Resonance Imaging to Visualize Leptomeningeal Inflammation in Patients With Multiple Sclerosis: A Review". JAMA Neurol. 74 (1): 100–109. doi:10.1001/jamaneurol.2016.4237. PMID 27893883. S2CID 11268426.