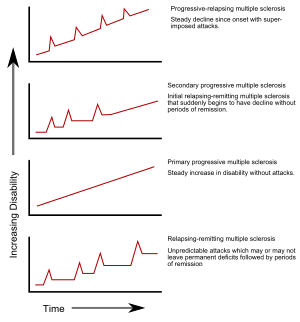
Multiple sclerosis (MS) is an autoimmune disease in which the insulating covers of nerve cells in the brain and spinal cord are damaged. This damage disrupts the ability of parts of the nervous system to transmit signals, resulting in a range of signs and symptoms, including physical, mental, and sometimes psychiatric problems. Specific symptoms can include double vision, vision loss, eye pain, muscle weakness, and loss of sensation or coordination. MS takes several forms, with new symptoms either occurring in isolated attacks or building up over time. In the relapsing forms of MS, between attacks, symptoms may disappear completely, although some permanent neurological problems often remain, especially as the disease advances. In the progressive forms of MS, bodily function slowly deteriorates and disability worsens once symptoms manifest and will steadily continue to do so if the disease is left untreated.

Oligoclonal bands (OCBs) are bands of immunoglobulins that are seen when a patient's blood serum, or cerebrospinal fluid (CSF) is analyzed. They are used in the diagnosis of various neurological and blood diseases. Oligoclonal bands are present in the CSF of more than 95% of patients with clinically definite multiple sclerosis.
Neuromyelitis optica spectrum disorders (NMOSD) are a spectrum of autoimmune diseases characterized by acute inflammation of the optic nerve and the spinal cord (myelitis). Episodes of ON and myelitis can be simultaneous or successive. A relapsing disease course is common, especially in untreated patients.
Interferon beta-1b is a cytokine in the interferon family used to treat the relapsing-remitting and secondary-progressive forms of multiple sclerosis (MS). It is approved for use after the first MS event. Closely related is interferon beta 1a, also indicated for MS, with a very similar drug profile.

The McDonald criteria are diagnostic criteria for multiple sclerosis (MS). These criteria are named after neurologist W. Ian McDonald who directed an international panel in association with the National Multiple Sclerosis Society (NMSS) of America and recommended revised diagnostic criteria for MS in April 2001. These new criteria intended to replace the Poser criteria and the older Schumacher criteria. They have undergone revisions in 2005, 2010 and 2017.
Experimental autoimmune encephalomyelitis, sometimes experimental allergic encephalomyelitis (EAE), is an animal model of brain inflammation. It is an inflammatory demyelinating disease of the central nervous system (CNS). It is mostly used with rodents and is widely studied as an animal model of the human CNS demyelinating diseases, including multiple sclerosis (MS) and acute disseminated encephalomyelitis (ADEM). EAE is also the prototype for T-cell-mediated autoimmune disease in general.

Multiple sclerosis is an inflammatory demyelinating disease of the CNS in which activated immune cells invade the central nervous system and cause inflammation, neurodegeneration, and tissue damage. The underlying cause is currently unknown. Current research in neuropathology, neuroimmunology, neurobiology, and neuroimaging, together with clinical neurology, provide support for the notion that MS is not a single disease but rather a spectrum.
Multiple sclerosis and other demyelinating diseases of the central nervous system (CNS) produce lesions and glial scars or scleroses. They present different shapes and histological findings according to the underlying condition that produces them.
Inflammatory demyelinating diseases (IDDs), sometimes called Idiopathic (IIDDs) due to the unknown etiology of some of them, are a heterogenous group of demyelinating diseases - conditions that cause damage to myelin, the protective sheath of nerve fibers - that occur against the background of an acute or chronic inflammatory process. IDDs share characteristics with and are often grouped together under Multiple Sclerosis. They are sometimes considered different diseases from Multiple Sclerosis, but considered by others to form a spectrum differing only in terms of chronicity, severity, and clinical course.

Baló's concentric sclerosis is a disease in which the white matter of the brain appears damaged in concentric layers, leaving the axis cylinder intact. It was described by József Mátyás Baló who initially named it "leuko-encephalitis periaxialis concentrica" from the previous definition, and it is currently considered one of the borderline forms of multiple sclerosis.
Research in multiple sclerosis may find new pathways to interact with the disease, improve function, curtail attacks, or limit the progression of the underlying disease. Many treatments already in clinical trials involve drugs that are used in other diseases or medications that have not been designed specifically for multiple sclerosis. There are also trials involving the combination of drugs that are already in use for multiple sclerosis. Finally, there are also many basic investigations that try to understand better the disease and in the future may help to find new treatments.
A clinically isolated syndrome (CIS) is a clinical situation of an individual's first neurological episode, caused by inflammation or demyelination of nerve tissue. An episode may be monofocal, in which symptoms present at a single site in the central nervous system, or multifocal, in which multiple sites exhibit symptoms. CIS with enough paraclinical evidence can be considered as a clinical stage of multiple sclerosis (MS). It can also be retrospectively diagnosed as a kind of MS when more evidence is available.

Tumefactive multiple sclerosis is a condition in which the central nervous system of a person has multiple demyelinating lesions with atypical characteristics for those of standard multiple sclerosis (MS). It is called tumefactive as the lesions are "tumor-like" and they mimic tumors clinically, radiologically and sometimes pathologically.
Diffuse myelinoclastic sclerosis, sometimes referred to as Schilder's disease, is a very infrequent neurodegenerative disease that presents clinically as pseudotumoural demyelinating lesions, making its diagnosis difficult. It usually begins in childhood, affecting children between 5 and 14 years old, but cases in adults are also possible.
Poser criteria are diagnostic criteria for multiple sclerosis (MS). They replaced the older Schumacher criteria, and are now considered obsolete as McDonald criteria have superseded them. Nevertheless, some of the concepts introduced have remained in MS research, like CDMS, and newer criteria are often calibrated against them. The criteria were unveiled in the Annals of Neurology in 1983 by a team led by Dr. Charles M. Poser.
Schumacher criteria are diagnostic criteria that were previously used for identifying multiple sclerosis (MS). Multiple sclerosis, understood as a central nervous system (CNS) condition, can be difficult to diagnose since its signs and symptoms may be similar to other medical problems. Medical organizations have created diagnostic criteria to ease and standardize the diagnostic process especially in the first stages of the disease. Schumacher criteria were the first internationally recognized criteria for diagnosis, and introduced concepts still in use, as CDMS.

Multiple sclerosis (MS) can be pathologically defined as the presence of distributed glial scars (scleroses) in the central nervous system that must show dissemination in time (DIT) and in space (DIS) to be considered MS lesions.
MOG antibody disease (MOGAD) or MOG antibody-associated encephalomyelitis (MOG-EM) is an inflammatory demyelinating disease of the central nervous system. Serum anti-myelin oligodendrocyte glycoprotein antibodies are present in up to half of patients with an acquired demyelinating syndrome and have been described in association with a range of phenotypic presentations, including acute disseminated encephalomyelitis, optic neuritis, transverse myelitis, and neuromyelitis optica.
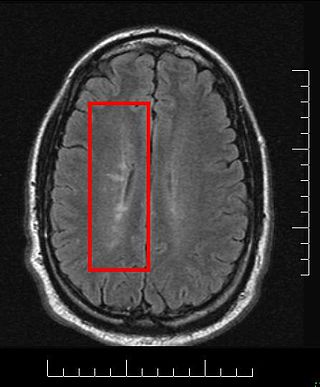
Radiologically isolated syndrome (RIS) is a clinical situation in which a person has white matter lesions suggestive of multiple sclerosis (MS), as shown on an MRI scan that was done for reasons unrelated to MS symptoms. The nerve lesions in these people show dissemination in space with an otherwise normal neurological examination and without historical accounts of typical MS symptoms.
Several biomarkers for diagnosis of multiple sclerosis, disease evolution and response to medication are under research. While most of them are still under research, there are some of them already well stablished: