
Glucagon is a peptide hormone, produced by alpha cells of the pancreas. It raises the concentration of glucose and fatty acids in the bloodstream and is considered to be the main catabolic hormone of the body. It is also used as a medication to treat a number of health conditions. Its effect is opposite to that of insulin, which lowers extracellular glucose. It is produced from proglucagon, encoded by the GCG gene.

Incretins are a group of metabolic hormones that stimulate a decrease in blood glucose levels. Incretins are released after eating and augment the secretion of insulin released from pancreatic beta cells of the islets of Langerhans by a blood-glucose–dependent mechanism.

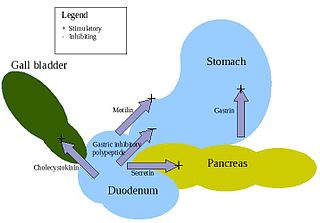
Glucose-dependent insulinotropic polypeptide, abbreviated as GIP, is an inhibiting hormone of the secretin family of hormones. While it is a weak inhibitor of gastric acid secretion, its main role, being an incretin, is to stimulate insulin secretion.
Glucagon-like peptide-2 (GLP-2) is a 33 amino acid peptide with the sequence HADGSFSDEMNTILDNLAARDFINWLIQTKITD in humans. GLP-2 is created by specific post-translational proteolytic cleavage of proglucagon in a process that also liberates the related glucagon-like peptide-1 (GLP-1). GLP-2 is produced by the intestinal endocrine L cell and by various neurons in the central nervous system. Intestinal GLP-2 is co-secreted along with GLP-1 upon nutrient ingestion.
Enteroglucagon is a peptide hormone derived from preproglucagon. It is a gastrointestinal hormone, secreted from mucosal cells primarily of the colon and terminal ileum. It consists of 37 amino acids. Enteroglucagon is released when fats and glucose are present in the small intestine; which decrease the motility to allow sufficient time for these nutrients to be absorbed.

Proglucagon is a protein that is cleaved from preproglucagon. Preproglucagon in humans is encoded by the GCG gene.

Glucagon-like peptide-1 (GLP-1) is a 30- or 31-amino-acid-long peptide hormone deriving from the tissue-specific posttranslational processing of the proglucagon peptide. It is produced and secreted by intestinal enteroendocrine L-cells and certain neurons within the nucleus of the solitary tract in the brainstem upon food consumption. The initial product GLP-1 (1–37) is susceptible to amidation and proteolytic cleavage, which gives rise to the two truncated and equipotent biologically active forms, GLP-1 (7–36) amide and GLP-1 (7–37). Active GLP-1 protein secondary structure includes two α-helices from amino acid position 13–20 and 24–35 separated by a linker region.

The glucagon receptor is a 62 kDa protein that is activated by glucagon and is a member of the class B G-protein coupled family of receptors, coupled to G alpha i, Gs and to a lesser extent G alpha q. Stimulation of the receptor results in the activation of adenylate cyclase and phospholipase C and in increased levels of the secondary messengers intracellular cAMP and calcium. In humans, the glucagon receptor is encoded by the GCGR gene.
Enteroendocrine cells are specialized cells of the gastrointestinal tract and pancreas with endocrine function. They produce gastrointestinal hormones or peptides in response to various stimuli and release them into the bloodstream for systemic effect, diffuse them as local messengers, or transmit them to the enteric nervous system to activate nervous responses. Enteroendocrine cells of the intestine are the most numerous endocrine cells of the body. They constitute an enteric endocrine system as a subset of the endocrine system just as the enteric nervous system is a subset of the nervous system. In a sense they are known to act as chemoreceptors, initiating digestive actions and detecting harmful substances and initiating protective responses. Enteroendocrine cells are located in the stomach, in the intestine and in the pancreas. Microbiota play key roles in the intestinal immune and metabolic responses in these enteroendocrine cells via their fermentation product, acetate.

The glucagon-like peptide-1 receptor (GLP1R) is a G protein-coupled receptor (GPCR) found on beta cells of the pancreas and on neurons of the brain. It is involved in the control of blood sugar level by enhancing insulin secretion. In humans it is synthesised by the gene GLP1R, which is present on chromosome 6. It is a member of the glucagon receptor family of GPCRs. GLP1R is composed of two domains, one extracellular (ECD) that binds the C-terminal helix of GLP-1, and one transmembrane (TMD) domain that binds the N-terminal region of GLP-1. In the TMD domain there is a fulcrum of polar residues that regulates the biased signaling of the receptor while the transmembrane helical boundaries and extracellular surface are a trigger for biased agonism.

Structures of GIPR human variant Sun, B., Kobilka, B.K., Sloop, K.W., Feng, D., Kobilka, T.S.
The glucagon receptor family is a group of closely related G-protein coupled receptors which include:

Free fatty acid receptor 2 (FFAR2), also termed G-protein coupled receptor 43 (GPR43), is a rhodopsin-like G-protein coupled receptor. It is coded by the FFAR2 gene. In humans, the FFAR2 gene is located on the long arm of chromosome 19 at position 13.12. Like other GPCRs, FFAR2s reside on the surface membrane of cells and when bond to one of their activating ligands regulate the function of their parent cells. FFAR2 is a member of a small family of structurally and functionally related GPRs termed free fatty acid receptors (FFARs). This family includes three other receptors which, like FFAR2, are activated by certain fatty acids: FFAR1, FFAR3 (GPR41), and FFAR4 (GPR120). FFAR2 and FFAR3 are activated by short-chain fatty acids whereas FFAR1 and FFAR4 are activated by long-chain fatty acids.

Vasoactive intestinal peptide receptor 2 also known as VPAC2, is a G-protein coupled receptor that in humans is encoded by the VIPR2 gene.

G protein-coupled receptor 119 also known as GPR119 is a G protein-coupled receptor that in humans is encoded by the GPR119 gene.
Vasoactive intestinal polypeptide receptor 1 also known as VPAC1, is a protein, that in humans is encoded by the VIPR1 gene. VPAC1 is expressed in the brain (cerebral cortex, hippocampus, amygdala), lung, prostate, peripheral blood leukocytes, liver, small intestine, heart, spleen, placenta, kidney, thymus and testis.
Secretin receptor family consists of secretin receptors regulated by peptide hormones from the glucagon hormone family. The family is different from adhesion G protein-coupled receptors.

Guanine nucleotide-binding protein G(t) subunit alpha-3, also known as gustducin alpha-3 chain, is a protein subunit that in humans is encoded by the GNAT3 gene.

Daniel Joshua Drucker is a Canadian endocrinologist. A Fellow of the Royal Society, he is a professor of medicine at the Lunenfeld-Tanenbaum Research Institute, Mount Sinai Hospital, Toronto. He is known for his research into intestinal hormones and their use in the treatment of diabetes, obesity, and other metabolic diseases, as well as intestinal failure.
GLP1 poly-agonist peptides are a class of drugs that activate multiple peptide hormone receptors including the glucagon-like peptide-1 (GLP-1) receptor. These drugs are developed for the same indications as GLP-1 receptor agonists—especially obesity, type 2 diabetes, and non-alcoholic fatty liver disease. They are expected to provide superior efficacy with fewer adverse effects compared to GLP-1 mono-agonists, which are dose-limited by gastrointestinal disturbances. The effectiveness of multi-receptor agonists could possibly equal or exceed that of bariatric surgery. The first such drug to receive approval is tirzepatide, a dual agonist of GLP-1 and GIP receptors.

















