
In neuroscience, repolarization refers to the change in membrane potential that returns it to a negative value just after the depolarization phase of an action potential which has changed the membrane potential to a positive value. The repolarization phase usually returns the membrane potential back to the resting membrane potential. The efflux of potassium (K+) ions results in the falling phase of an action potential. The ions pass through the selectivity filter of the K+ channel pore.
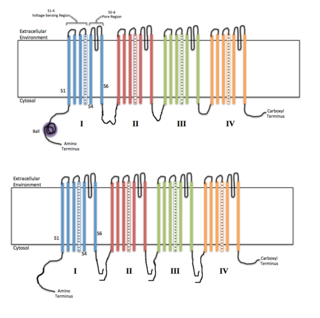
Voltage-gated ion channels are a class of transmembrane proteins that form ion channels that are activated by changes in a cell's electrical membrane potential near the channel. The membrane potential alters the conformation of the channel proteins, regulating their opening and closing. Cell membranes are generally impermeable to ions, thus they must diffuse through the membrane through transmembrane protein channels.
The shaker (Sh) gene, when mutated, causes a variety of atypical behaviors in the fruit fly, Drosophila melanogaster. Under ether anesthesia, the fly’s legs will shake ; even when the fly is unanaesthetized, it will exhibit aberrant movements. Sh-mutant flies have a shorter lifespan than regular flies; in their larvae, the repetitive firing of action potentials as well as prolonged exposure to neurotransmitters at neuromuscular junctions occurs.
Ciguatoxins are a class of toxic polycyclic polyethers found in fish that cause ciguatera.

Maurotoxin is a peptide toxin from the venom of the Tunisian chactoid scorpion Scorpio maurus palmatus, from which it was first isolated and from which the chemical gets its name. It acts by blocking several types of voltage-gated potassium channel.

Kappa- Hefutoxin 1 and 2 are toxins from the venom of the Asian forest black scorpion Heterometrus fulvipes with a unique structure. It blocks the potassium channels Kv1.2 and Kv1.3 and slows the activation of Kv1.3.

Margatoxin (MgTX) is a peptide that selectively inhibits Kv1.3 voltage-dependent potassium channels. It is found in the venom of Centruroides margaritatus, also known as the Central American Bark Scorpion. Margatoxin was first discovered in 1993. It was purified from scorpion venom and its amino acid sequence was determined.

Pumiliotoxin 251D is a toxic organic compound. It is found in the skin of poison frogs from the genera Dendrobates, Epipedobates, Minyobates, and Phyllobates and toads from the genus Melanophryniscus. Its name comes from the pumiliotoxin family (PTXs) and its molecular mass of 251 daltons. When the toxin enters the bloodstream through cuts in the skin or by ingestion, it can cause hyperactivity, convulsions, cardiac arrest and ultimately death. It is especially toxic to arthropods, even at low concentrations.

Potassium voltage-gated channel, shaker-related subfamily, member 3, also known as KCNA3 or Kv1.3, is a protein that in humans is encoded by the KCNA3 gene.

Potassium voltage-gated channel, Shab-related subfamily, member 1, also known as KCNB1 or Kv2.1, is a protein that, in humans, is encoded by the KCNB1 gene.

Guangxitoxin, also known as GxTX, is a peptide toxin found in the venom of the tarantula Plesiophrictus guangxiensis. It primarily inhibits outward voltage-gated Kv2.1 potassium channel currents, which are prominently expressed in pancreatic β-cells, thus increasing insulin secretion.

Pi3 toxin is a purified peptide derivative of the Pandinus imperator scorpion venom. It is a potent blocker of voltage-gated potassium channel, Kv1.3 and is closely related to another peptide found in the venom, Pi2.

Antillatoxin (ATX) is a potent lipopeptide neurotoxin produced by the marine cyanobacterium Lyngbya majuscula. ATX activates voltage-gated sodium channels, which can cause cell depolarisation, NMDA-receptor overactivity, excess calcium influx and neuronal necrosis.
The ion channel hypothesis of Alzheimer's disease (AD), also known as the channel hypothesis or the amyloid beta ion channel hypothesis, is a more recent variant of the amyloid hypothesis of AD, which identifies amyloid beta (Aβ) as the underlying cause of neurotoxicity seen in AD. While the traditional formulation of the amyloid hypothesis pinpoints insoluble, fibrillar aggregates of Aβ as the basis of disruption of calcium ion homeostasis and subsequent apoptosis in AD, the ion channel hypothesis in 1993 introduced the possibility of an ion-channel-forming oligomer of soluble, non-fibrillar Aβ as the cytotoxic species allowing unregulated calcium influx into neurons in AD.
Pi4 is a short toxin from the scorpion Pandinus imperator that blocks specific potassium channels.
BmP02, also known as α-KTx 9.1 or Bmkk(6), is a toxin from the Buthus Martensi Karsch (BmK) scorpion. The toxin acts on potassium channels, blocking Kv1.3 and slowing the deactivation of Kv4.2. BmP02 is not toxic to humans or mice.

BrMT (6-bromo-2-mercaptotryptamine) is a neurotoxin found in the hypobranchial gland of the marine snail species Calliostoma canaliculatum. The disulfide-linked dimer of BrMT possesses inhibitory effects on the Kv1 and Kv4 families of voltage-gated potassium channels.

Scaritoxin, a potent toxic substance, is a ciguatoxin with molecular formula C60H84O16. Scaritoxin is also referred to as ciguaotoxin 4A, CTX4A. Like other ciguatoxins, CTX4A is produced by dinoflagellate Gambierdiscus toxicus and isolated from poisonous fish.

Ciguatoxin 1 or CTX-1 is a toxic chemical compound, the most common and potent type in the group of ciguatoxins. It is a large molecule consisting of polycyclic polyethers that can be found in certain types of fish in the Pacific Ocean. The compound is produced by Dinoflagellates Gambierdiscus toxicus and is passed on through the food chain by fish. The compound has no effect in fish but is toxic to humans.
ImKTX58 is a peptide toxin from the venom of the scorpion species Isometrus maculatus. It is known for its selective inhibition of Kv1.3 channels, on which it acts as a pore-blocker.
















