Related Research Articles
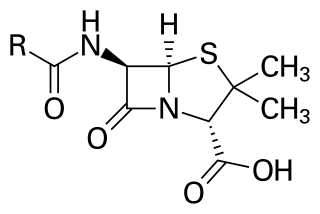
Penicillins are a group of β-lactam antibiotics originally obtained from Penicillium moulds, principally P. chrysogenum and P. rubens. Most penicillins in clinical use are synthesised by P. chrysogenum using deep tank fermentation and then purified. A number of natural penicillins have been discovered, but only two purified compounds are in clinical use: penicillin G and penicillin V. Penicillins were among the first medications to be effective against many bacterial infections caused by staphylococci and streptococci. They are still widely used today for different bacterial infections, though many types of bacteria have developed resistance following extensive use.
Peptidoglycan or murein is a unique large macromolecule, a polysaccharide, consisting of sugars and amino acids that forms a mesh-like peptidoglycan layer outside the plasma membrane, the rigid cell wall characteristic of most bacteria. The sugar component consists of alternating residues of β-(1,4) linked N-acetylglucosamine (NAG) and N-acetylmuramic acid (NAM). Attached to the N-acetylmuramic acid is an oligopeptide chain made of three to five amino acids. The peptide chain can be cross-linked to the peptide chain of another strand forming the 3D mesh-like layer. Peptidoglycan serves a structural role in the bacterial cell wall, giving structural strength, as well as counteracting the osmotic pressure of the cytoplasm. This repetitive linking results in a dense peptidoglycan layer which is critical for maintaining cell form and withstanding high osmotic pressures, and it is regularly replaced by peptidoglycan production. Peptidoglycan hydrolysis and synthesis are two processes that must occur in order for cells to grow and multiply, a technique carried out in three stages: clipping of current material, insertion of new material, and re-crosslinking of existing material to new material.
DD-transpeptidase is a bacterial enzyme that catalyzes the transfer of the R-L-αα-D-alanyl moiety of R-L-αα-D-alanyl-D-alanine carbonyl donors to the γ-OH of their active-site serine and from this to a final acceptor. It is involved in bacterial cell wall biosynthesis, namely, the transpeptidation that crosslinks the peptide side chains of peptidoglycan strands.
Penicillin-binding proteins (PBPs) are a group of proteins that are characterized by their affinity for and binding of penicillin. They are a normal constituent of many bacteria; the name just reflects the way by which the protein was discovered. All β-lactam antibiotics bind to PBPs, which are essential for bacterial cell wall synthesis. PBPs are members of a subgroup of enzymes called transpeptidases. Specifically, PBPs are DD-transpeptidases.
A carboxypeptidase is a protease enzyme that hydrolyzes (cleaves) a peptide bond at the carboxy-terminal (C-terminal) end of a protein or peptide. This is in contrast to an aminopeptidases, which cleave peptide bonds at the N-terminus of proteins. Humans, animals, bacteria and plants contain several types of carboxypeptidases that have diverse functions ranging from catabolism to protein maturation. At least two mechanisms have been discussed.
Lysyl oxidase (LOX), also known as protein-lysine 6-oxidase, is an enzyme that, in humans, is encoded by the LOX gene. It catalyzes the conversion of lysine residues into its aldehyde derivative allysine. Allysine form cross-links in extracellular matrix proteins. Inhibition of lysyl oxidase can cause osteolathyrism, but, at the same time, its upregulation by tumor cells may promote metastasis of the existing tumor, causing it to become malignant and cancerous.

Carboxypeptidase E (CPE), also known as carboxypeptidase H (CPH) and enkephalin convertase, is an enzyme that in humans is encoded by the CPE gene. This enzyme catalyzes the release of C-terminal arginine or lysine residues from polypeptides.
D-amino-acid dehydrogenase is a bacterial enzyme that catalyses the oxidation of D-amino acids into their corresponding oxoacids. It contains both flavin and nonheme iron as cofactors. The enzyme has a very broad specificity and can act on most D-amino acids.
In enzymology, a lysine—tRNA ligase is an enzyme that catalyzes the chemical reaction
In enzymology, an UDP-N-acetylmuramoylpentapeptide-lysine N6-alanyltransferase (EC 2.3.2.10) is an enzyme that catalyzes the chemical reaction

In enzymology, 4-aminobutyrate transaminase, also called GABA transaminase or 4-aminobutyrate aminotransferase, or GABA-T, is an enzyme that catalyzes the chemical reaction:

Carboxypeptidase Z is an enzyme that in humans is encoded by the CPZ gene.
Jack Leonard Strominger is the Higgins Professor of Biochemistry at Harvard University, specializing in the structure and function of human histocompatibility proteins and their role in disease. He won the Albert Lasker Award for Basic Medical Research in 1995.
N-acetylmuramoyl-L-alanyl-D-glutamyl-L-lysyl-(N6-glycyl)-D-alanyl-D-alanine-diphosphoundecaprenyl-N-acetylglucosamine:glycine glycyltransferase (EC 2.3.2.17, femA (gene)) is an enzyme with systematic name N-acetylmuramoyl-L-alanyl-D-glutamyl-L-lysyl-(N6-glycyl)-D-alanyl-D-alanine-ditrans,octacis-diphosphoundecaprenyl-N-acetylglucosamine:glycine glycyltransferase. This enzyme catalyses the following chemical reaction
N-acetylmuramoyl-L-alanyl-D-glutamyl-L-lysyl-(N6-triglycine)-D-alanyl-D-alanine-diphosphoundecaprenyl-N-acetylglucosamine:glycine glycyltransferase (EC 2.3.2.18, femB (gene)) is an enzyme with systematic name N-acetylmuramoyl-L-alanyl-D-glutamyl-L-lysyl-(N6-triglycine)-D-alanyl-D-alanine-ditrans,octacis-diphosphoundecaprenyl-N-acetylglucosamine:glycine glycyltransferase. This enzyme catalyses the following chemical reaction
Alanine carboxypeptidase is an enzyme. This enzyme catalyses the following chemical reaction
Muramoylpentapeptide carboxypeptidase is an enzyme. This enzyme catalyses the following chemical reaction.
Zinc D-Ala-D-Ala carboxypeptidase (EC 3.4.17.14, Zn2+ G peptidase, D-alanyl-D-alanine hydrolase, D-alanyl-D-alanine-cleaving carboxypeptidase, DD-carboxypeptidase, G enzyme, DD-carboxypeptidase-transpeptidase) is an enzyme. This enzyme catalyses the following chemical reaction
Metallocarboxypeptidase D is an enzyme. This enzyme catalyses the following chemical reaction
Carboxypeptidase D can refer to one of several enzymes. A family of serine carboxypeptidases includes is an enzyme. This enzyme has an optimal pH of 4.5-6.0, is inhibited by diisopropyl fluorophosphate, and catalyses the following chemical reaction
References
- ↑ DasGupta H, Fan DP (July 1979). "Purification and characterization of a carboxypeptidase-transpeptidase of Bacillus megaterium acting on the tetrapeptide moiety of the peptidoglycan". The Journal of Biological Chemistry. 254 (13): 5672–83. PMID 109439.
- ↑ Rousset A, Nguyen-Distèche M, Minck R, Ghuysen JM (December 1982). "Penicillin-binding proteins and carboxypeptidase/transpeptidase activities in Proteus vulgaris P18 and its penicillin-induced stable L-forms". Journal of Bacteriology. 152 (3): 1042–8. PMC 221607 . PMID 6754695.
- ↑ Metz R, Henning S, Hammes WP (March 1986). "LD-carboxypeptidase activity in Escherichia coli. II. Isolation, purification and characterization of the enzyme from E. coli K 12". Archives of Microbiology. 144 (2): 181–6. doi:10.1007/bf00414732. PMID 3521530.