Related Research Articles
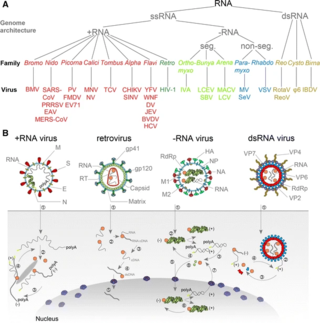
An RNA virus is a virus characterized by a ribonucleic acid (RNA) based genome. The genome can be single-stranded RNA (ssRNA) or double-stranded (dsRNA). Notable human diseases caused by RNA viruses include influenza, SARS, MERS, COVID-19, Dengue virus, hepatitis C, hepatitis E, West Nile fever, Ebola virus disease, rabies, polio, mumps, and measles.
Virology is the scientific study of biological viruses. It is a subfield of microbiology that focuses on their detection, structure, classification and evolution, their methods of infection and exploitation of host cells for reproduction, their interaction with host organism physiology and immunity, the diseases they cause, the techniques to isolate and culture them, and their use in research and therapy.

Poliovirus, the causative agent of polio, is a serotype of the species Enterovirus C, in the family of Picornaviridae. There are three poliovirus serotypes, numbered 1, 2, and 3.
Viral evolution is a subfield of evolutionary biology and virology that is specifically concerned with the evolution of viruses. Viruses have short generation times, and many—in particular RNA viruses—have relatively high mutation rates. Although most viral mutations confer no benefit and often even prove deleterious to viruses, the rapid rate of viral mutation combined with natural selection allows viruses to quickly adapt to changes in their host environment. In addition, because viruses typically produce many copies in an infected host, mutated genes can be passed on to many offspring quickly. Although the chance of mutations and evolution can change depending on the type of virus, viruses overall have high chances for mutations.

Defective interfering particles (DIPs), also known as defective interfering viruses, are spontaneously generated virus mutants in which a critical portion of the particle's genome has been lost due to defective replication or non-homologous recombination. The mechanism of their formation is presumed to be as a result of template-switching during replication of the viral genome, although non-replicative mechanisms involving direct ligation of genomic RNA fragments have also been proposed. DIPs are derived from and associated with their parent virus, and particles are classed as DIPs if they are rendered non-infectious due to at least one essential gene of the virus being lost or severely damaged as a result of the defection. A DIP can usually still penetrate host cells, but requires another fully functional virus particle to co-infect a cell with it, in order to provide the lost factors.

Picornaviruses are a group of related nonenveloped RNA viruses which infect vertebrates including fish, mammals, and birds. They are viruses that represent a large family of small, positive-sense, single-stranded RNA viruses with a 30 nm icosahedral capsid. The viruses in this family can cause a range of diseases including the common cold, poliomyelitis, meningitis, hepatitis, and paralysis.
Site-directed mutagenesis is a molecular biology method that is used to make specific and intentional mutating changes to the DNA sequence of a gene and any gene products. Also called site-specific mutagenesis or oligonucleotide-directed mutagenesis, it is used for investigating the structure and biological activity of DNA, RNA, and protein molecules, and for protein engineering.
Microviridae is a family of bacteriophages with a single-stranded DNA genome. The name of this family is derived from the ancient Greek word μικρός (mikrós), meaning "small". This refers to the size of their genomes, which are among the smallest of the DNA viruses. Enterobacteria, intracellular parasitic bacteria, and spiroplasma serve as natural hosts. There are 22 species in this family, divided among seven genera and two subfamilies.
VPg is a protein that is covalently attached to the 5′ end of positive strand viral RNA and acts as a primer during RNA synthesis in a variety of virus families including Picornaviridae, Potyviridae, Astroviridae and Caliciviridae. There are some studies showing that a possible VPg protein is also present in astroviridae, however, experimental evidence for this has not yet been provided and requires further study. The primer activity of VPg occurs when the protein becomes uridylated, providing a free hydroxyl that can be extended by the virally encoded RNA-dependent RNA polymerase. For some virus families, VPg also has a role in translation initiation by acting like a 5' mRNA cap.

The phi X 174 bacteriophage is a single-stranded DNA (ssDNA) virus that infects Escherichia coli. This virus was isolated in 1935 by Nicolas Bulgakov in Félix d'Hérelle's laboratory at the Pasteur Institute, from samples collected in Paris sewers. Its characterization and the study of its replication mechanism were carried out from the 1950s onwards. It was the first DNA-based genome to be sequenced. This work was completed by Fred Sanger and his team in 1977. In 1962, Walter Fiers and Robert Sinsheimer had already demonstrated the physical, covalently closed circularity of ΦX174 DNA. Nobel prize winner Arthur Kornberg used ΦX174 as a model to first prove that DNA synthesized in a test tube by purified enzymes could produce all the features of a natural virus, ushering in the age of synthetic biology. In 1972–1974, Jerard Hurwitz, Sue Wickner, and Reed Wickner with collaborators identified the genes required to produce the enzymes to catalyze conversion of the single stranded form of the virus to the double stranded replicative form. In 2003, it was reported by Craig Venter's group that the genome of ΦX174 was the first to be completely assembled in vitro from synthesized oligonucleotides. The ΦX174 virus particle has also been successfully assembled in vitro. In 2012, it was shown how its highly overlapping genome can be fully decompressed and still remain functional.
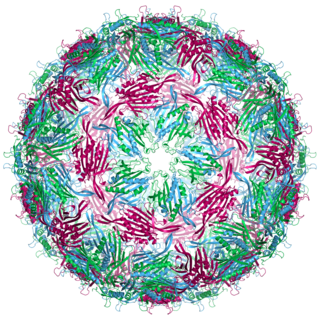
Bacteriophage MS2, commonly called MS2, is an icosahedral, positive-sense single-stranded RNA virus that infects the bacterium Escherichia coli and other members of the Enterobacteriaceae. MS2 is a member of a family of closely related bacterial viruses that includes bacteriophage f2, bacteriophage Qβ, R17, and GA.

Vincent R. Racaniello is a Higgins Professor in the Department of Microbiology and Immunology at Columbia University's College of Physicians and Surgeons. He is a co-author of a textbook on virology, Principles of Virology.
The history of virology – the scientific study of viruses and the infections they cause – began in the closing years of the 19th century. Although Edward Jenner and Louis Pasteur developed the first vaccines to protect against viral infections, they did not know that viruses existed. The first evidence of the existence of viruses came from experiments with filters that had pores small enough to retain bacteria. In 1892, Dmitri Ivanovsky used one of these filters to show that sap from a diseased tobacco plant remained infectious to healthy tobacco plants despite having been filtered. Martinus Beijerinck called the filtered, infectious substance a "virus" and this discovery is considered to be the beginning of virology.

A virus is a submicroscopic infectious agent that replicates only inside the living cells of an organism. Viruses infect all life forms, from animals and plants to microorganisms, including bacteria and archaea. Viruses are found in almost every ecosystem on Earth and are the most numerous type of biological entity. Since Dmitri Ivanovsky's 1892 article describing a non-bacterial pathogen infecting tobacco plants and the discovery of the tobacco mosaic virus by Martinus Beijerinck in 1898, more than 11,000 of the millions of virus species have been described in detail. The study of viruses is known as virology, a subspeciality of microbiology.
Eckard Wimmer is a German American virologist, organic chemist and distinguished professor of molecular genetics and microbiology at Stony Brook University. He is best known for his seminal work on the molecular biology of poliovirus and the first chemical synthesis of a viral genome capable of infection and subsequent production of live viruses.

Cis-acting replication elements (cre) bring together the 5′ and 3′ ends during replication of positive-sense single-stranded RNA viruses and double-stranded RNA viruses.
Clyde A. Hutchison III is an American biochemist and microbiologist notable for his research on site-directed mutagenesis and synthetic biology. He is Professor Emeritus of Microbiology and Immunology at the University of North Carolina at Chapel Hill, distinguished professor at the J Craig Venter Institute, a member of the National Academy of Sciences, and a fellow of the American Academy of Arts and Sciences.

Reverse genetics is a method in molecular genetics that is used to help understand the function(s) of a gene by analysing the phenotypic effects caused by genetically engineering specific nucleic acid sequences within the gene. The process proceeds in the opposite direction to forward genetic screens of classical genetics. While forward genetics seeks to find the genetic basis of a phenotype or trait, reverse genetics seeks to find what phenotypes are controlled by particular genetic sequences.

Viral metagenomics uses metagenomic technologies to detect viral genomic material from diverse environmental and clinical samples. Viruses are the most abundant biological entity and are extremely diverse; however, only a small fraction of viruses have been sequenced and only an even smaller fraction have been isolated and cultured. Sequencing viruses can be challenging because viruses lack a universally conserved marker gene so gene-based approaches are limited. Metagenomics can be used to study and analyze unculturable viruses and has been an important tool in understanding viral diversity and abundance and in the discovery of novel viruses. For example, metagenomics methods have been used to describe viruses associated with cancerous tumors and in terrestrial ecosystems.
Nicola Jane Stonehouse is a British virologist who is a professor in molecular virology at the University of Leeds. Her research investigates viral diseases and the use of RNA aptamers to study viral proteins. She has worked on the development of a novel poliovirus vaccine that makes use of virus-like particles.
References
- ↑ Cello, Jeronimo; Paul, Aniko V.; Wimmer, Eckard (2002-08-09). "Chemical Synthesis of Poliovirus cDNA: Generation of Infectious Virus in the Absence of Natural Template". Science. 297 (5583): 1016–1018. Bibcode:2002Sci...297.1016C. doi: 10.1126/science.1072266 . ISSN 0036-8075. PMID 12114528. S2CID 5810309.
- ↑ Heather, James M.; Chain, Benjamin (January 2016). "The sequence of sequencers: The history of sequencing DNA". Genomics. 107 (1): 1–8. doi:10.1016/j.ygeno.2015.11.003. ISSN 0888-7543. PMC 4727787 . PMID 26554401.
- ↑ Stauft, Charles B; Wimmer, Eckard (2018-02-16), "Synthetic Viruses", eLS, John Wiley & Sons, Ltd, pp. 1–7, doi:10.1002/9780470015902.a0027771, ISBN 9780470015902
- ↑ "The Little Phage That Could". California Institute of Technology. 2023-07-13. Retrieved 2025-01-22.
- ↑ Wimmer, Eckard; Mueller, Steffen; Tumpey, Terrence M; Taubenberger, Jeffery K (December 2009). "Synthetic viruses: a new opportunity to understand and prevent viral disease". Nature Biotechnology. 27 (12): 1163–72. doi:10.1038/nbt.1593. ISSN 1087-0156. PMC 2819212 . PMID 20010599.
- ↑ Guenther, Caitlin M.; Kuypers, Brianna E.; Lam, Michael T.; Robinson, Tawana M.; Zhao, Julia; Suh, Junghae (November 2014). "Synthetic Virology: Engineering Viruses for Gene Delivery". Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology. 6 (6): 548–558. doi:10.1002/wnan.1287. ISSN 1939-5116. PMC 4227300 . PMID 25195922.