
5-HT receptors, 5-hydroxytryptamine receptors, or serotonin receptors, are a group of G protein-coupled receptor and ligand-gated ion channels found in the central and peripheral nervous systems. They mediate both excitatory and inhibitory neurotransmission. The serotonin receptors are activated by the neurotransmitter serotonin, which acts as their natural ligand.
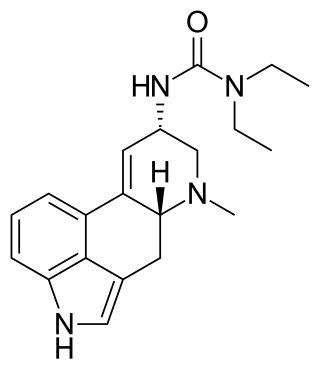
Lisuride, sold under the brand name Dopergin among others, is a monoaminergic medication of the ergoline class which is used in the treatment of Parkinson's disease, migraine, and high prolactin levels. It is taken by mouth.
The 5-HT2A receptor is a subtype of the 5-HT2 receptor that belongs to the serotonin receptor family and is a G protein-coupled receptor (GPCR). The 5-HT2A receptor is a cell surface receptor, but has several intracellular locations.

A serotonin receptor agonist is an agonist of one or more serotonin receptors. They activate serotonin receptors in a manner similar to that of serotonin, a neurotransmitter and hormone and the endogenous ligand of the serotonin receptors.

meta-Chlorophenylpiperazine (mCPP) is a psychoactive drug of the phenylpiperazine class. It was initially developed in the late-1970s and used in scientific research before being sold as a designer drug in the mid-2000s. It has been detected in pills touted as legal alternatives to illicit stimulants in New Zealand and pills sold as "ecstasy" in Europe and the United States.

5-Methoxytryptamine (5-MT), also known as mexamine, is a tryptamine derivative closely related to the neurotransmitters serotonin and melatonin. 5-MT has been shown to occur naturally in the body in low levels. It is formed via the deacetylation of melatonin in the pineal gland.

Naphthylaminopropane (PAL-287) is an experimental drug under investigation as of 2007 for the treatment of alcohol and stimulant addiction.
5-Hydroxytryptamine receptor 2B (5-HT2B) also known as serotonin receptor 2B is a protein that in humans is encoded by the HTR2B gene. 5-HT2B is a member of the 5-HT2 receptor family that binds the neurotransmitter serotonin (5-hydroxytryptamine, 5-HT). Like all 5-HT2 receptors, the 5-HT2B receptor is Gq/G11-protein coupled, leading to downstream activation of phospholipase C.
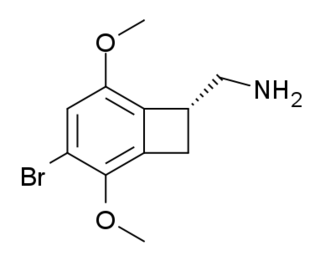
TCB-2 is a hallucinogen discovered in 2006 by Thomas McLean working in the lab of David Nichols at Purdue University. It is a conformationally-restricted derivative of the phenethylamine 2C-B, also a hallucinogen, and acts as a potent agonist for the 5-HT2A and 5-HT2C receptors with a Ki of 0.26 nM at the human 5-HT2A receptor. In drug-substitution experiments in rats, TCB-2 was found to be of similar potency to both LSD and Bromo-DragonFLY, ranking it among the most potent phenethylamine hallucinogens yet discovered. This high potency and selectivity has made TCB-2 useful for distinguishing 5-HT2A mediated responses from those produced by other similar receptors. TCB-2 has similar but not identical effects in animals to related phenethylamine hallucinogens such as DOI, and has been used for studying how the function of the 5-HT2A receptor differs from that of other serotonin receptors in a number of animal models, such as studies of cocaine addiction and neuropathic pain.

YM-348 is an indazole derivative drug which acts as a potent and selective 5-HT2C receptor agonist, with an EC50 of 1nM and 15x selectivity over 5-HT2A, although it only has moderate selectivity of 3x over the closely related 5-HT2B receptor. It has thermogenic and anorectic effects in animal studies, making it potentially useful for the treatment of obesity.

BW-723C86 is a tryptamine derivative drug which acts as a 5-HT2B receptor agonist. It has anxiolytic effects in animal studies, and is also used for investigating the function of the 5-HT2B receptor in a range of other tissues.
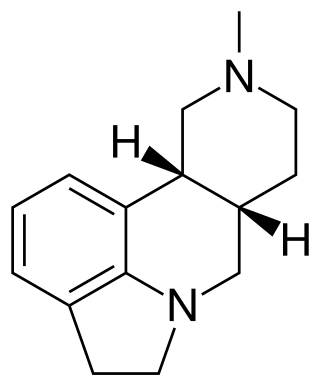
SDZ SER-082 is a drug which acts as a mixed antagonist for the 5-HT2B and 5-HT2C serotonin receptors, with good selectivity over other serotonin receptor subtypes and slight preference for 5-HT2C over 5-HT2B. It has been used in animal studies into the behavioural effects of the different 5-HT2 subtypes, and how they influence the effects of other drugs such as cocaine.
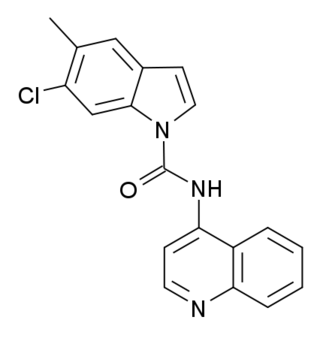
SB-215505 is a drug which acts as a potent and selective antagonist at the serotonin 5-HT2B receptor, with good selectivity over the related 5-HT2A and 5-HT2C receptors. It is used in scientific research into the function of the 5-HT2 family of receptors, especially to study the role of 5-HT2B receptors in the heart, and to distinguish 5-HT2B-mediated responses from those produced by 5-HT2A or 5-HT2C.
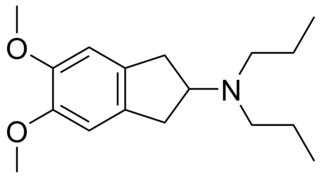
PNU-99,194(A) (or U-99,194(A)) is a drug which acts as a moderately selective D3 receptor antagonist with ~15-30-fold preference for D3 over the D2 subtype. Though it has substantially greater preference for D3 over D2, the latter receptor does still play some role in its effects, as evidenced by the fact that PNU-99,194 weakly stimulates both prolactin secretion and striatal dopamine synthesis, actions it does not share with the more selective (100-fold) D3 receptor antagonists S-14,297 and GR-103,691.

S-14671 is a naphthylpiperazine derivative which acts as a 5-HT1A receptor agonist (pKi = 9.3) with high efficacy and exceptional in vivo potency, and also as a 5-HT2A and 5-HT2C receptor antagonist (both are pKi = 7.8). It displays only low and non-significant affinity for 5-HT1B and 5-HT3 sites.

PRX-08066 is a drug discovered and developed by Predix Pharmaceuticals [Dale S. Dhanoa et al. Patent US 7,030,240 B2], which acts as a potent and selective antagonist at the serotonin 5-HT2B receptor, with a 5-HT2Bbinding affinity (Ki) of 3.4nM, and high selectivity over the closely related 5-HT2A and 5-HT2C receptors and other receptor targets. PRX-08066 and other selective 5-HT2B antagonists are being researched for the treatment of pulmonary arterial hypertension, following the discovery that the potent 5-HT2B agonist norfenfluramine produces pulmonary arterial hypertension and subsequent heart valve damage. In animal studies, PRX-08066 has been found to reduce several key indicators of pulmonary arterial hypertension and improved cardiac output, with similar efficacy to established drugs for this condition such as bosentan, sildenafil, beraprost and iloprost. It is also being researched for potential anti-cancer applications, due to its ability to inhibit fibroblast activation.

SB-206553 is a drug which acts as a mixed antagonist for the 5-HT2B and 5-HT2C serotonin receptors. It has anxiolytic properties in animal studies and interacts with a range of other drugs. It has also been shown to act as a positive allosteric modulator of α7 nicotinic acetylcholine receptors. Modified derivatives of SB-206553 have been used to probe the structure of the 5-HT2B receptor.
5-HT2C receptor agonists are a class of drugs that activate 5-HT2C receptors. They have been investigated for the treatment of a number of conditions including obesity, psychiatric disorders, sexual dysfunction and urinary incontinence.

25CN-NBOH is a compound indirectly derived from the phenethylamine series of hallucinogens, which was discovered in 2014 at the University of Copenhagen. This compound is notable as one of the most selective agonist ligands for the 5-HT2A receptor yet discovered, with a pKi of 8.88 at the human 5-HT2A receptor and with 100x selectivity for 5-HT2A over 5-HT2C, and 46x selectivity for 5-HT2A over 5-HT2B. A tritiated version of 25CN-NBOH has also been accessed and used for more detailed investigations of the binding to 5-HT2 receptors and autoradiography.

1-Methylpsilocin (developmental code names CMY, CMY-16) is a tryptamine derivative developed by Sandoz which acts as a selective agonist of the serotonin 5-HT2C receptor (IC50Tooltip half-maximal inhibitory concentration of 12 nM, vs. 633 nM at 5-HT2A), and an inverse agonist at 5-HT2B (Ki of 38 nM). While 1-methylpsilocin does have higher affinity for 5-HT2C than 5-HT2A, it does produce a head-twitch response in mice that is dependent on 5-HT2A, so it is not entirely free of effects on 5-HT2Ain vivo. In contrast to psilocin, 1-methylpsilocin did not activate 5-HT1A receptors in mice.



















