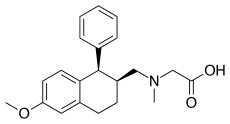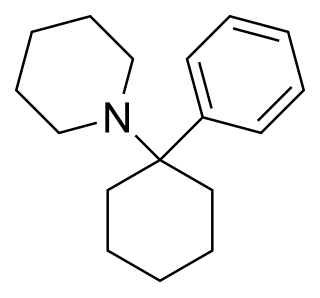
Phencyclidine or phenylcyclohexyl piperidine (PCP), also known as angel dust among other names, is a dissociative anesthetic mainly used recreationally for its significant mind-altering effects. PCP may cause hallucinations, distorted perceptions of sounds, and violent behavior. As a recreational drug, it is typically smoked, but may be taken by mouth, snorted, or injected. It may also be mixed with cannabis or tobacco.

Psychopharmacology is the scientific study of the effects drugs have on mood, sensation, thinking, behavior, judgment and evaluation, and memory. It is distinguished from neuropsychopharmacology, which emphasizes the correlation between drug-induced changes in the functioning of cells in the nervous system and changes in consciousness and behavior.

Dizocilpine (INN), also known as MK-801, is a pore blocker of the N-Methyl-D-aspartate (NMDA) receptor, a glutamate receptor, discovered by a team at Merck in 1982. Glutamate is the brain's primary excitatory neurotransmitter. The channel is normally blocked with a magnesium ion and requires depolarization of the neuron to remove the magnesium and allow the glutamate to open the channel, causing an influx of calcium, which then leads to subsequent depolarization. Dizocilpine binds inside the ion channel of the receptor at several of PCP's binding sites thus preventing the flow of ions, including calcium (Ca2+), through the channel. Dizocilpine blocks NMDA receptors in a use- and voltage-dependent manner, since the channel must open for the drug to bind inside it. The drug acts as a potent anti-convulsant and probably has dissociative anesthetic properties, but it is not used clinically for this purpose because of the discovery of brain lesions, called Olney's lesions (see below), in laboratory rats. Dizocilpine is also associated with a number of negative side effects, including cognitive disruption and psychotic-spectrum reactions. It inhibits the induction of long term potentiation and has been found to impair the acquisition of difficult, but not easy, learning tasks in rats and primates. Because of these effects of dizocilpine, the NMDA receptor pore blocker ketamine is used instead as a dissociative anesthetic in human medical procedures. While ketamine may also trigger temporary psychosis in certain individuals, its short half-life and lower potency make it a much safer clinical option. However, dizocilpine is the most frequently used uncompetitive NMDA receptor antagonist in animal models to mimic psychosis for experimental purposes.

AP-7 is a selective NMDA receptor (NMDAR) antagonist that competitively inhibits the glutamate binding site and thus activation of NMDAR. It has anticonvulsant effects.
Cross-tolerance is a phenomenon that occurs when tolerance to the effects of a certain drug produces tolerance to another drug. It often happens between two drugs with similar functions or effects—for example, acting on the same cell receptor or affecting the transmission of certain neurotransmitters. Cross-tolerance has been observed with pharmaceutical drugs such as anti-anxiety agents and illicit substances, and sometimes the two of them together. Often, a person who uses one drug can be tolerant to a drug that has a completely different function. This phenomenon allows one to become tolerant to a drug that they have never used before.

Acamprosate, sold under the brand name Campral, is a medication used along with counseling to treat alcohol use disorder.

NMDA receptor antagonists are a class of drugs that work to antagonize, or inhibit the action of, the N-Methyl-D-aspartate receptor (NMDAR). They are commonly used as anesthetics for animals and humans; the state of anesthesia they induce is referred to as dissociative anesthesia.
A serotonin–norepinephrine–dopamine reuptake inhibitor (SNDRI), also known as a triple reuptake inhibitor (TRI), is a type of drug that acts as a combined reuptake inhibitor of the monoamine neurotransmitters serotonin, norepinephrine, and dopamine. It does this by concomitantly inhibiting the serotonin transporter (SERT), norepinephrine transporter (NET), and dopamine transporter (DAT), respectively. Inhibition of the reuptake of these neurotransmitters increases their extracellular concentrations and, therefore, results in an increase in serotonergic, adrenergic, and dopaminergic neurotransmission. The naturally-occurring and potent SNDRI cocaine is widely used recreationally and often illegally for the euphoric effects it produces.

Sodium- and chloride-dependent glycine transporter 1, also known as glycine transporter 1, is a protein that in humans is encoded by the SLC6A9 gene which is promising therapeutic target for treatment of diabetes and obesity.
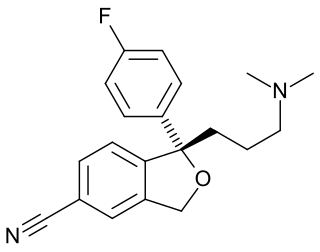
A reuptake inhibitor (RI) is a type of drug known as a reuptake modulator that inhibits the plasmalemmal transporter-mediated reuptake of a neurotransmitter from the synapse into the pre-synaptic neuron. This leads to an increase in extracellular concentrations of the neurotransmitter and an increase in neurotransmission. Various drugs exert their psychological and physiological effects through reuptake inhibition, including many antidepressants and psychostimulants.

Arylcyclohexylamines, also known as arylcyclohexamines or arylcyclohexanamines, are a chemical class of pharmaceutical, designer, and experimental drugs.

Metapramine is a tricyclic antidepressant (TCA) developed by Rhone Poulenc that was introduced for the treatment of depression in France in 1984. In addition to its efficacy against affective disorders, it also has analgesic properties, and may be useful in the treatment of pain.

Bitopertin is a glycine reuptake inhibitor which was under development by Roche as an adjunct to antipsychotics for the treatment of persistent negative symptoms or suboptimally controlled positive symptoms associated with schizophrenia. Research into this indication has been largely halted as a result of disappointing trial results.

7-Chlorokynurenic acid (7-CKA) is a tool compound that acts as a potent and selective competitive antagonist of the glycine site of the NMDA receptor. It produces ketamine-like rapid antidepressant effects in animal models of depression. However, 7-CKA is unable to cross the blood-brain-barrier, and for this reason, is unsuitable for clinical use. As a result, a centrally-penetrant prodrug of 7-CKA, 4-chlorokynurenine (AV-101), has been developed for use in humans, and is being studied in clinical trials as a potential treatment for major depressive disorder, and anti-nociception. In addition to antagonizing the NMDA receptor, 7-CKA also acts as a potent inhibitor of the reuptake of glutamate into synaptic vesicles, an action that it mediates via competitive blockade of vesicular glutamate transporters.
A glycine reuptake inhibitor (GRI) is a type of drug which inhibits the reuptake of the neurotransmitter glycine by blocking one or more of the glycine transporters (GlyTs). Examples of GRIs include bitopertin (RG1678), Org 24598, Org 25935, ALX-5407, and sarcosine, which are selective GlyT1 blockers, and Org 25543 and N-arachidonylglycine, which are selective GlyT2 blockers. Some weak and/or non-selective GlyT blockers include amoxapine and ethanol (alcohol).

Arketamine (developmental code names PCN-101, HR-071603), also known as (R)-ketamine or (R)-(−)-ketamine, is the (R)-(−) enantiomer of ketamine. Similarly to racemic ketamine and esketamine, the S(+) enantiomer of ketamine, arketamine is biologically active; however, it is less potent as an NMDA receptor antagonist and anesthetic and thus has never been approved or marketed for clinical use as an enantiopure drug. Arketamine is currently in clinical development as a novel antidepressant.

Neboglamine, formerly known as nebostinel, is a positive allosteric modulator of the glycine site of the NMDA receptor which is under investigation for Rottapharm for the treatment of schizophrenia and cocaine dependence. It shows cognition- and memory-enhancing effects in animal models. As of June 2015, it is in phase II clinical trials for both schizophrenia and cocaine abuse.

Pesampator is a positive allosteric modulator (PAM) of the AMPA receptor (AMPAR), an ionotropic glutamate receptor, which is under development by Pfizer for the treatment of cognitive symptoms in schizophrenia. It was also under development for the treatment of age-related sensorineural hearing loss, but development for this indication was terminated due to insufficient effectiveness. As of July 2018, pesampator is in phase II clinical trials for cognitive symptoms in schizophrenia.