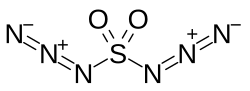
 | |
| Names | |
|---|---|
| IUPAC name Sulfuryl diazide | |
| Other names Sulfuryl azide; Sulfonyl diazide | |
| Identifiers | |
3D model (JSmol) | |
| ChemSpider | |
PubChem CID | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| SO2(N3)2 | |
| Molar mass | 148.10 g·mol−1 |
| Melting point | −15 °C (5 °F; 258 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Sulfuryl diazide or sulfuryl azide is a chemical compound with the molecular formula SO2(N3)2. It was first described in the 1920s when its reactions with benzene and p-xylene were studied by Theodor Curtius and Karl Friedrich Schmidt. [1] [2] [3] The compound is reported as having "exceedingly explosive, unpredictable properties" and "in many cases very violent explosions occurred without any apparent reason". [1]