| |
 | |
| Names | |
|---|---|
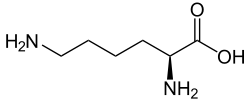
| Preferred IUPAC name (2S)-2-Amino-6-oxohexanoic acid | |
| Other names 2-aminoadipate semialdehyde, 2-amino-5-formylvaleric acid, norvaline, 6-oxo-DL-norleucine | |
| Identifiers | |
3D model (JSmol) | |
| ChEBI | |
| ChemSpider | |
| KEGG | |
| MeSH | allysine |
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| C6H11NO3 | |
| Molar mass | 145.158 g·mol−1 |
| Appearance | unstable |
| Density | 1.74g/cm3 |
| Boiling point | 295.2 °C (563.4 °F; 568.3 K) |
| Hazards | |
| Flash point | 132.3 °C (270.1 °F; 405.4 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
Allysine is a derivative of lysine that features a formyl group in place of the terminal amine. The free amino acid does not exist, but the allysine residue does. It is produced by aerobic oxidation of lysine residues by the enzyme lysyl oxidase. The transformation is an example of a post-translational modification. The semialdehyde form exists in equilibrium with a cyclic derivative. [1]
Contents