In chemistry, an ester is a compound derived from an acid in which the hydrogen atom (H) of at least one acidic hydroxyl group of that acid is replaced by an organyl group. Analogues derived from oxygen replaced by other chalcogens belong to the ester category as well. According to some authors, organyl derivatives of acidic hydrogen of other acids are esters as well, but not according to the IUPAC.
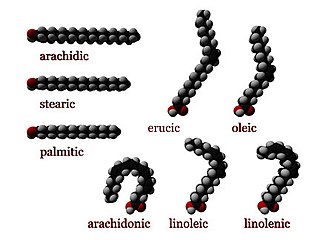
In chemistry, particularly in biochemistry, a fatty acid is a carboxylic acid with an aliphatic chain, which is either saturated or unsaturated. Most naturally occurring fatty acids have an unbranched chain of an even number of carbon atoms, from 4 to 28. Fatty acids are a major component of the lipids in some species such as microalgae but in some other organisms are not found in their standalone form, but instead exist as three main classes of esters: triglycerides, phospholipids, and cholesteryl esters. In any of these forms, fatty acids are both important dietary sources of fuel for animals and important structural components for cells.
Saponification is a process of converting esters into soaps and alcohols by the action of aqueous alkali. Soaps are salts of fatty acids, which in turn are carboxylic acids with long carbon chains. Sodium stearate is a typical soap.
Lactones are cyclic carboxylic esters, containing a 1-oxacycloalkan-2-one structure, or analogues having unsaturation or heteroatoms replacing one or more carbon atoms of the ring.

Propionic acid is a naturally occurring carboxylic acid with chemical formula CH
3CH
2CO
2H. It is a liquid with a pungent and unpleasant smell somewhat resembling body odor. The anion CH
3CH
2CO−
2 as well as the salts and esters of propionic acid are known as propionates or propanoates.

Stearic acid is a saturated fatty acid with an 18-carbon chain. The IUPAC name is octadecanoic acid. It is a soft waxy solid with the formula CH3(CH2)16CO2H. The triglyceride derived from three molecules of stearic acid is called stearin. Stearic acid is a prevalent FA in nature, found in many animal and vegetable fats, but is usually higher in animal fat than vegetable fat. It has a melting point of 69.4 °C and a pKa of 4.50.

Goat cheese, goat's cheese, or chèvre is cheese made from goat's milk. Goats were among the first animals to be domesticated for producing food. Goat cheese is made around the world with a variety of recipes, giving many different styles of cheeses, from fresh and soft to aged and hard.

Isobutyric acid, also known as 2-methylpropanoic acid or isobutanoic acid, is a carboxylic acid with structural formula (CH3)2CHCOOH. It is an isomer of n-butyric acid. It is classified as a short-chain fatty acid. Deprotonation or esterification gives derivatives called isobutyrates.

Valeric acid or pentanoic acid is a straight-chain alkyl carboxylic acid with the chemical formula CH3(CH2)3COOH. Like other low-molecular-weight carboxylic acids, it has an unpleasant odor. It is found in the perennial flowering plant Valeriana officinalis, from which it gets its name. Its primary use is in the synthesis of its esters. Salts and esters of valeric acid are known as valerates or pentanoates. Volatile esters of valeric acid tend to have pleasant odors and are used in perfumes and cosmetics. Several, including ethyl valerate and pentyl valerate are used as food additives because of their fruity flavors.
Myristic acid is a common saturated fatty acid with the molecular formula CH3(CH2)12COOH. Its salts and esters are commonly referred to as myristates or tetradecanoates. It is named after the binomial name for nutmeg, from which it was first isolated in 1841 by Lyon Playfair.
Capric acid, also known as decanoic acid or decylic acid, is a saturated fatty acid, medium-chain fatty acid (MCFA), and carboxylic acid. Its formula is CH3(CH2)8COOH. Salts and esters of decanoic acid are called caprates or decanoates. The term capric acid is derived from the Latin "caper / capra" (goat) because the sweaty, unpleasant smell of the compound is reminiscent of goats.

Caprylic acid, also known under the systematic name octanoic acid or C8 Acid, is a saturated fatty acid, medium-chain fatty acid (MCFA). It has the structural formula H3C−(CH2)6−COOH, and is a colorless oily liquid that is minimally soluble in water with a slightly unpleasant rancid-like smell and taste. Salts and esters of octanoic acid are known as octanoates or caprylates. It is a common industrial chemical, which is produced by oxidation of the C8 aldehyde. Its compounds are found naturally in the milk of various mammals and as a minor constituent of coconut oil and palm kernel oil.
Enanthic acid, also called heptanoic acid, is an organic compound composed of a seven-carbon chain terminating in a carboxylic acid functional group. It is a colorless oily liquid with an unpleasant, rancid odor. It contributes to the odor of some rancid oils. It is slightly soluble in water, but very soluble in ethanol and ether. Salts and esters of enanthic acid are called enanthates or heptanoates.

3-Methylbutanoic acid, also known as β-methylbutyric acid or more commonly isovaleric acid, is a branched-chain alkyl carboxylic acid with the chemical formula (CH3)2CHCH2CO2H. It is classified as a short-chain fatty acid. Like other low-molecular-weight carboxylic acids, it has an unpleasant odor. The compound occurs naturally and can be found in many foods, such as cheese, soy milk, and apple juice.
The Polenske value is a value determined when examining fat. It is an indicator of how much volatile fatty acid can be extracted from fat through saponification. It is equal to the number of milliliters of 0.1 normal alkali solution necessary for the neutralization of the water-insoluble volatile fatty acids distilled and filtered from 5 grams of a given saponified fat. It is measure of the steam volatile and water insoluble fatty acids, chiefly caprylic, capric and lauric acids, present in oil and fat. The value is named for the chemist who developed it, Eduard Polenske.
Oleochemistry is the study of vegetable oils and animal oils and fats, and oleochemicals derived from these fats and oils. The resulting product can be called oleochemicals (from Latin: oleum "olive oil"). The major product of this industry is soap, approximately 8.9×106 tons of which were produced in 1990. Other major oleochemicals include fatty acids, fatty acid methyl esters, fatty alcohols and fatty amines. Glycerol is a side product of all of these processes. Intermediate chemical substances produced from these basic oleochemical substances include alcohol ethoxylates, alcohol sulfates, alcohol ether sulfates, quaternary ammonium salts, monoacylglycerols (MAG), diacylglycerols (DAG), structured triacylglycerols (TAG), sugar esters, and other oleochemical products.
Ethyl octanoate, also known as ethyl caprylate, is a fatty acid ester formed from caprylic acid and ethanol. A colorless liquid at room temperature, it has the semi-developed formula of CH3(CH2)6COOCH2CH3, and is used in food industries as a flavoring and in the perfume industry as a scent additive. It is present in many fruits and alcoholic beverages, and has a strong odor of fruit and flowers. It is used in the creation of synthetic fruity scents.
Viscoleo is a thin or low-viscosity vegetable oil. It is specifically a proprietary form of fractionated coconut oil and a medium-chain triglyceride (MCT) oil. It is prepared from the dried, solid endosperm of the fruit Cocos nucifera via hydrolysis, fractionation, and purification. Viscoleo is composed of the medium-chain fatty acids caprylic acid (C8) (55–60%), capric acid (C10) (40%), lauric acid (C12) (1–5%), and caproic acid (C6) (0.5%). It is used as an oil vehicle for several depot antipsychotics including clopentixol decanoate, flupentixol decanoate, pipotiazine palmitate, zuclopentixol acetate, and zuclopentixol decanoate. Injectable antipsychotics using Viscoleo as a carrier may be absorbed more rapidly and have shorter durations than preparations using sesame oil.
Methyl hexanoate is the fatty acid methyl ester of hexanoic acid, a colourless liquid organic compound with the chemical formula CH3−(CH2)4−COO−CH3. It is found naturally in many foods and has a role as a plant metabolite. It can also be found in the cytoplasm of cells.