
In organic chemistry a diene is a covalent compound that contains two double bonds, usually among carbon atoms. They thus contain two alkene units, with the standard prefix di of systematic nomenclature. As a subunit of more complex molecules, dienes occur in naturally occurring and synthetic chemicals and are used in organic synthesis. Conjugated dienes are widely used as monomers in the polymer industry. Polyunsaturated fats are of interest to nutrition.
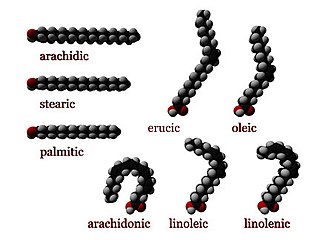
In chemistry, particularly in biochemistry, a fatty acid is a carboxylic acid with an aliphatic chain, which is either saturated or unsaturated. Most naturally occurring fatty acids have an unbranched chain of an even number of carbon atoms, from 4 to 28. Fatty acids are a major component of the lipids in some species such as microalgae but in some other organisms are not found in their standalone form, but instead exist as three main classes of esters: triglycerides, phospholipids, and cholesteryl esters. In any of these forms, fatty acids are both important dietary sources of fuel for animals and important structural components for cells.

In nutrition, biology, and chemistry, fat usually means any ester of fatty acids, or a mixture of such compounds, most commonly those that occur in living beings or in food.

A triglyceride is an ester derived from glycerol and three fatty acids. Triglycerides are the main constituents of body fat in humans and other vertebrates, as well as vegetable fat. They are also present in the blood to enable the bidirectional transference of adipose fat and blood glucose from the liver, and are a major component of human skin oils.
Essential fatty acids, or EFAs, are fatty acids that humans and other animals must ingest because the body requires them for good health but cannot synthesize them.

alpha-Linolenic acid (ALA), also known as α-Linolenic acid, is an n−3, or omega-3, essential fatty acid. ALA is found in many seeds and oils, including flaxseed, walnuts, chia, hemp, and many common vegetable oils.

Oleic acid is a fatty acid that occurs naturally in various animal and vegetable fats and oils. It is an odorless, colorless oil, although commercial samples may be yellowish. In chemical terms, oleic acid is classified as a monounsaturated omega-9 fatty acid, abbreviated with a lipid number of 18:1 cis-9. It has the formula CH3(CH2)7CH=CH(CH2)7COOH. The name derives from the Latin word oleum, which means oil. It is the most common fatty acid in nature. The salts and esters of oleic acid are called oleates.
Linoleic acid (LA) is an organic compound with the formula COOH(CH2)7CH=CHCH2CH=CH(CH2)4CH3. Both alkene groups are cis. It is a fatty acid sometimes denoted 18:2 (n-6) or 18:2 cis-9,12. A linoleate is a salt or ester of this acid.

Conjugated linoleic acids (CLA) are a family of at least 28 isomers of linoleic acid found mostly in the meat and dairy products derived from ruminants. Their two double bonds are conjugated. CLAs can be either cis-fats or trans-fats.

Falcarinol is a natural pesticide and fatty alcohol found in carrots, red ginseng and ivy. In carrots, it occurs in a concentration of approximately 2 mg/kg. As a toxin, it protects roots from fungal diseases, such as liquorice rot that causes black spots on the roots during storage. The compound requires the freezing condition to maintain well because it is sensitive to light and heat.

Trifluoroacetic acid (TFA) is an organofluorine compound with the chemical formula CF3CO2H. It is a structural analogue of acetic acid with all three of the acetyl group's hydrogen atoms replaced by fluorine atoms and is a colorless liquid with a vinegar-like odor.
Vaccenic acid is a naturally occurring trans fatty acid. It is the predominant kind of trans-fatty acid found in human milk, in the fat of ruminants, and in dairy products such as milk, butter, and yogurt. Trans fat in human milk may depend on trans fat content in food.

Rumenic acid, also known as bovinic acid, is a conjugated linoleic acid (CLA) found in the fat of ruminants and in dairy products. It is an omega-7 trans fat. Its lipid shorthand name is cis-9, trans-11 18:2 acid. The name was proposed by Kramer et al. in 1998. It is formed along with vaccenic acid by biohydrogenation of dietary polyunsaturated fatty acids in the rumen. It can be considered as the principal dietary form, accounting for as much as 85-90% of the total CLA content in dairy products.

α-Eleostearic acid or (9Z,11E,13E)-octadeca-9,11,13-trienoic acid, is an organic compound, a conjugated fatty acid and one of the isomers of octadecatrienoic acid. It is often called simply eleostearic acid although there is also a β-eleostearic acid. Its high degree of unsaturation gives tung oil its properties as a drying oil.

Conjugated fatty acids are polyunsaturated fatty acids in which at least one pair of double bonds is separated by only one single bond. The conjugation implies four consecutive unsaturated links (i.e., CH rather than CH2 groups), and results in a delocalization of electrons along the double-bonded carbons. An example of a conjugated fatty acid is the rumenic acid, found in the meat and milk of ruminants.

Punicic acid is a polyunsaturated fatty acid, 18:3 cis-9, trans-11, cis-13. It is named for the pomegranate,, and is obtained from pomegranate seed oil. It has also been found in the seed oils of snake gourd.
Elaidinization is any chemical reaction which convert a cis- olefin to a trans- olefin in unsaturated fatty acids. This is often performed on fats and oils to increase both the melting point and the shelf life without reducing the degree of unsaturation. The term originates from elaidic acid, the trans-isomer of oleic acid.

α-Parinaric acid is a conjugated polyunsaturated fatty acid. Discovered by Tsujimoto and Koyanagi in 1933, it contains 18 carbon atoms and 4 conjugated double bonds. The repeating single bond-double bond structure of α-parinaric acid distinguishes it structurally and chemically from the usual "methylene-interrupted" arrangement of polyunsaturated fatty acids that have double-bonds and single bonds separated by a methylene unit (−CH2−). Because of the fluorescent properties conferred by the alternating double bonds, α-parinaric acid is commonly used as a molecular probe in the study of biomembranes.

9-Hydroxyoctadecadienoic acid has been used in the literature to designate either or both of two stereoisomer metabolites of the essential fatty acid, linoleic acid: 9(S)-hydroxy-10(E),12(Z)-octadecadienoic acid and 9(R)-hydroxy-10(E),12(Z)-octadecadienoic acid ; these two metabolites differ in having their hydroxy residues in the S or R configurations, respectively. The accompanying figure gives the structure for 9(S)-HETE. Two other 9-hydroxy linoleic acid derivatives occur in nature, the 10E,12E isomers of 9(S)-HODE and 9(R)-HODE viz., 9(S)-hydroxy-10E,12E-octadecadienoic acid and 9(R)-hydroxy-10E,12E-octadecadienoic acid ; these two derivatives have their double bond at carbon 12 in the E or trans configuration as opposed to the Z or cis configuration. The four 9-HODE isomers, particularly under conditions of oxidative stress, may form together in cells and tissues; they have overlapping but not identical biological activities and significances. Because many studies have not distinguished between the S and R stereoisomers and, particularly in identifying tissue levels, the two EE isomers, 9-HODE is used here when the isomer studied is unclear.

Trans fat, also called trans-unsaturated fatty acids, or trans fatty acids, is a type of unsaturated fat that naturally occurs in small amounts in meat and milk fat. It became widely produced as an unintentional byproduct in the industrial processing of vegetable and fish oils in the early 20th century for use in margarine and later also in snack food, packaged baked goods, and for frying fast food.













