In nutrition, biology, and chemistry, fat usually means any ester of fatty acids, or a mixture of such compounds, most commonly those that occur in living beings or in food.

Margarine is a spread used for flavoring, baking, and cooking. It is most often used as a substitute for butter. Although originally made from animal fats, most margarine consumed today is made from vegetable oil. The spread was originally named oleomargarine from Latin for oleum and Greek margarite. The name was later shortened to margarine.

Coconut oil is an edible oil derived from the kernels, meat, and milk of the coconut palm fruit. Coconut oil is a white solid fat below around 25 °C (77 °F), and a clear thin liquid oil in warmer climates. Unrefined varieties have a distinct coconut aroma. Coconut oil is used as a food oil, and in industrial applications for cosmetics and detergent production. The oil is rich in medium-chain fatty acids.
A saturated fat is a type of fat in which the fatty acid chains have all single bonds. A fat known as a glyceride is made of two kinds of smaller molecules: a short glycerol backbone and fatty acids that each contain a long linear or branched chain of carbon (C) atoms. Along the chain, some carbon atoms are linked by single bonds (-C-C-) and others are linked by double bonds (-C=C-). A double bond along the carbon chain can react with a pair of hydrogen atoms to change into a single -C-C- bond, with each H atom now bonded to one of the two C atoms. Glyceride fats without any carbon chain double bonds are called saturated because they are "saturated with" hydrogen atoms, having no double bonds available to react with more hydrogen.

Arachidic acid, also known as icosanoic acid, is a saturated fatty acid with a 20-carbon chain. It is a minor constituent of cupuaçu butter (7%), perilla oil (0–1%), peanut oil (1.1–1.7%), corn oil (3%), and cocoa butter (1%). The salts and esters of arachidic acid are known as arachidates.

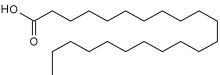
Oleic acid is a fatty acid that occurs naturally in various animal and vegetable fats and oils. It is an odorless, colorless oil, although commercial samples may be yellowish. In chemical terms, oleic acid is classified as a monounsaturated omega-9 fatty acid, abbreviated with a lipid number of 18:1 cis-9, and a main product of Δ9 desaturase. It has the formula CH3−(CH2)7−CH=CH−(CH2)7−COOH. The name derives from the Latin word oleum, which means oil. It is the most common fatty acid in nature. The salts and esters of oleic acid are called oleates. It is part of many oils and thus used in a lot of artificial food, as well as for soap.
Linoleic acid (LA) is an organic compound with the formula HOOC(CH
2)
7CH=CHCH
2CH=CH(CH
2)
4CH
3. Both alkene groups are cis. It is a fatty acid sometimes denoted 18:2 (n-6) or 18:2 cis-9,12. A linoleate is a salt or ester of this acid.
In biochemistry and nutrition, monounsaturated fats are fatty acids that have one double bond in the fatty acid chain with all of the remainder carbon atoms being single-bonded. By contrast, polyunsaturated fatty acids (PUFAs) have more than one double bond.

Peanut oil, also known as groundnut oil or arachis oil, is a vegetable oil derived from peanuts. The oil usually has a mild or neutral flavor but, if made with roasted peanuts, has a stronger peanut flavor and aroma. It is often used in American, Chinese, Indian, African and Southeast Asian cuisine, both for general cooking, and in the case of roasted oil, for added flavor. Peanut oil has a high smoke point relative to many other cooking oils, so it is commonly used for frying foods.
Lauric acid, systematically dodecanoic acid, is a saturated fatty acid with a 12-carbon atom chain, thus having many properties of medium-chain fatty acids. It is a bright white, powdery solid with a faint odor of bay oil or soap. The salts and esters of lauric acid are known as laurates.
Palm kernel oil is an edible plant oil derived from the kernel of the oil palm tree Elaeis guineensis. It is related to other two edible oils: palm oil, extracted from the fruit pulp of the oil palm, and coconut oil, extracted from the kernel of the coconut.
Fatty alcohols (or long-chain alcohols) are usually high-molecular-weight, straight-chain primary alcohols, but can also range from as few as 4–6 carbons to as many as 22–26, derived from natural fats and oils. The precise chain length varies with the source. Some commercially important fatty alcohols are lauryl, stearyl, and oleyl alcohols. They are colourless oily liquids (for smaller carbon numbers) or waxy solids, although impure samples may appear yellow. Fatty alcohols usually have an even number of carbon atoms and a single alcohol group (–OH) attached to the terminal carbon. Some are unsaturated and some are branched. They are widely used in industry. As with fatty acids, they are often referred to generically by the number of carbon atoms in the molecule, such as "a C12 alcohol", that is an alcohol having 12 carbons, for example dodecanol.

Monoglycerides are a class of glycerides which are composed of a molecule of glycerol linked to a fatty acid via an ester bond. As glycerol contains both primary and secondary alcohol groups two different types of monoglycerides may be formed; 1-monoacylglycerols where the fatty acid is attached to a primary alcohol, or a 2-monoacylglycerols where the fatty acid is attached to the secondary alcohol.

Ben oil is pressed from the seeds of the Moringa oleifera, known variously as the horseradish tree, ben oil tree, or drumstick tree. The oil is characterized by an unusually long shelf life and a mild, but pleasant taste. The name of the oil is derived from the presence of behenic acid. The oil's components are:

Pecan oil is an edible pressed oil extracted from the pecan nut. Pecan oil is neutral in flavor and takes on the flavor of whatever seasoning is being used with it. Pecan oil contains 9.5% saturated fat, which is less than in olive oil (13.5%), peanut oil (16.90%) or corn oil (12.70%). It is also used as a massage oil and in aromatherapy applications.
Margaric acid, or heptadecanoic acid, is a saturated fatty acid. Its molecular formula is CH3(CH2)15CO2H. Classified as an odd-chain fatty acid, it occurs as a trace component of the fat and milkfat of ruminants. Salts and esters of margaric acid are called heptadecanoates.

Cooking oil is a plant or animal liquid fat used in frying, baking, and other types of cooking. Oil allows higher cooking temperatures than water, making cooking faster and more flavorful, while likewise distributing heat, reducing burning and uneven cooking. It sometimes imparts its own flavor. Cooking oil is also used in food preparation and flavoring not involving heat, such as salad dressings and bread dips.

Hair oil is an oil-based cosmetic product intended to improve the condition of hair. Various types of oils may be included in hair oil products. These often purport to aid with hair growth, dryness, or damage.

Trans fat, also called trans-unsaturated fatty acids, or trans fatty acids, is a type of unsaturated fat that occurs in foods. Trace concentrations of trans fats occur naturally, but large amounts are found in some processed foods. Since consumption of trans fats is unhealthy, artificial trans fats are highly regulated or banned in many nations. However, they are still widely consumed in developing nations, resulting in hundreds of thousands of deaths each year. The World Health Organization (WHO) has set a goal to make the world free from industrially produced trans fat by the end of 2023.