Related Research Articles

Apoptosis is a form of programmed cell death that occurs in multicellular organisms and in some eukaryotic, single-celled microorganisms such as yeast. Biochemical events lead to characteristic cell changes (morphology) and death. These changes include blebbing, cell shrinkage, nuclear fragmentation, chromatin condensation, DNA fragmentation, and mRNA decay. The average adult human loses 50 to 70 billion cells each day due to apoptosis. For the average human child between 8 and 14 years old, each day the approximate loss is 20 to 30 billion cells.

Caspases are a family of protease enzymes playing essential roles in programmed cell death. They are named caspases due to their specific cysteine protease activity – a cysteine in its active site nucleophilically attacks and cleaves a target protein only after an aspartic acid residue. As of 2009, there are 12 confirmed caspases in humans and 10 in mice, carrying out a variety of cellular functions.
Programmed cell death is the death of a cell as a result of events inside of a cell, such as apoptosis or autophagy. PCD is carried out in a biological process, which usually confers advantage during an organism's lifecycle. For example, the differentiation of fingers and toes in a developing human embryo occurs because cells between the fingers apoptose; the result is that the digits are separate. PCD serves fundamental functions during both plant and animal tissue development.

Caspase-9 is an enzyme that in humans is encoded by the CASP9 gene. It is an initiator caspase, critical to the apoptotic pathway found in many tissues. Caspase-9 homologs have been identified in all mammals for which they are known to exist, such as Mus musculus and Pan troglodytes.

Caspase-8 is a caspase protein, encoded by the CASP8 gene. It most likely acts upon caspase-3. CASP8 orthologs have been identified in numerous mammals for which complete genome data are available. These unique orthologs are also present in birds.
Inhibitors of apoptosis are a group of proteins that mainly act on the intrinsic pathway that block programmed cell death, which can frequently lead to cancer or other effects for the cell if mutated or improperly regulated. Many of these inhibitors act to block caspases, a family of cysteine proteases that play an integral role in apoptosis. Some of these inhibitors include the Bcl-2 family, viral inhibitor crmA, and IAP's.
Caspase 2 also known as CASP2 is an enzyme that, in humans, is encoded by the CASP2 gene. CASP2 orthologs have been identified in nearly all mammals for which complete genome data are available. Unique orthologs are also present in birds, lizards, lissamphibians, and teleosts.

X-linked inhibitor of apoptosis protein (XIAP), also known as inhibitor of apoptosis protein 3 (IAP3) and baculoviral IAP repeat-containing protein 4 (BIRC4), is a protein that stops apoptotic cell death. In humans, this protein (XIAP) is produced by a gene named XIAP gene located on the X chromosome.

Caspase-3 is a caspase protein that interacts with caspase-8 and caspase-9. It is encoded by the CASP3 gene. CASP3 orthologs have been identified in numerous mammals for which complete genome data are available. Unique orthologs are also present in birds, lizards, lissamphibians, and teleosts.

Caspase-7, apoptosis-related cysteine peptidase, also known as CASP7, is a human protein encoded by the CASP7 gene. CASP7 orthologs have been identified in nearly all mammals for which complete genome data are available. Unique orthologs are also present in birds, lizards, lissamphibians, and teleosts.

Caspase-6 is an enzyme that in humans is encoded by the CASP6 gene. CASP6 orthologs have been identified in numerous mammals for which complete genome data are available. Unique orthologs are also present in birds, lizards, lissamphibians, and teleosts. Caspase-6 has known functions in apoptosis, early immune response and neurodegeneration in Huntington's and Alzheimer's disease.

Caspase-10 is an enzyme that, in humans, is encoded by the CASP10 gene.

Apoptotic protease activating factor 1, also known as APAF1, is a human homolog of C. elegans CED-4 gene.

Serine protease HTRA2, mitochondrial is an enzyme that in humans is encoded by the HTRA2 gene. This protein is involved in caspase-dependent apoptosis and in Parkinson's disease.
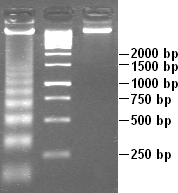
Apoptotic DNA fragmentation is a key feature of apoptosis, a type of programmed cell death. Apoptosis is characterized by the activation of endogenous endonucleases, particularly the caspase-3 activated DNase (CAD), with subsequent cleavage of nuclear DNA into internucleosomal fragments of roughly 180 base pairs (bp) and multiples thereof (360, 540 etc.). The apoptotic DNA fragmentation is being used as a marker of apoptosis and for identification of apoptotic cells either via the DNA laddering assay, the TUNEL assay, or the by detection of cells with fractional DNA content ("sub G1 cells") on DNA content frequency histograms e.g. as in the Nicoletti assay.

Caspase-activated DNase (CAD) or DNA fragmentation factor subunit beta is a protein that in humans is encoded by the DFFB gene. It breaks up the DNA during apoptosis and promotes cell differentiation. It is usually an inactive monomer inhibited by ICAD. This is cleaved before dimerization.
Caspase-2 is an enzyme. This enzyme catalyses the following chemical reaction
Murine caspase-11, and its human homologs caspase-4 and caspase-5, are mammalian intracellular receptor proteases activated by TLR4 and TLR3 signaling during the innate immune response. Caspase-11, also termed the non-canonical inflammasome, is activated by TLR3/TLR4-TRIF signaling and directly binds cytosolic lipopolysaccharide (LPS), a major structural element of Gram-negative bacterial cell walls. Activation of caspase-11 by LPS is known to cause the activation of other caspase proteins, leading to septic shock, pyroptosis, and often organismal death.

The Early 35 kDa protein, or P35 in short, is a baculoviral protein that inhibits apoptosis in the cells infected by the virus. Although baculoviruses infect only invertebrates in nature, ectopic expression of P35 in vertebrate animals and cells also results in inhibition of apoptosis, thus indicating a universal mechanism. P35 has been shown to be a caspase inhibitor with a very wide spectrum of activity both in regard to inhibited caspase types and to species in which the mechanism is conserved.

Death regulator Nedd2-like caspase was firstly identified and characterised in Drosophila in 1999 as a cysteine protease containing an amino-terminal caspase recruitment domain. At first, it was thought of as an effector caspase involved in apoptosis, but subsequent findings have proved that it is, in fact, an initiator caspase with a crucial role in said type of programmed cell death.
References
- ↑ Chang HY, Yang X (December 2000). "Proteases for cell suicide: functions and regulation of caspases". Microbiology and Molecular Biology Reviews. 64 (4): 821–46. doi:10.1128/mmbr.64.4.821-846.2000. PMC 99015 . PMID 11104820.
- ↑ Fischer U, Stroh C, Schulze-Osthoff K (January 2006). "Unique and overlapping substrate specificities of caspase-8 and caspase-10". Oncogene. 25 (1): 152–9. doi: 10.1038/sj.onc.1209015 . PMID 16186808.
- ↑ Shikama Y, Yamada M, Miyashita T (July 2003). "Caspase-8 and caspase-10 activate NF-kappaB through RIP, NIK and IKKalpha kinases". European Journal of Immunology. 33 (7): 1998–2006. doi: 10.1002/eji.200324013 . PMID 12884866.
- ↑ Boatright KM, Deis C, Denault JB, Sutherlin DP, Salvesen GS (September 2004). "Activation of caspases-8 and -10 by FLIP(L)". The Biochemical Journal. 382 (Pt 2): 651–7. doi:10.1042/bj20040809. PMC 1133822 . PMID 15209560.