
Flavonoids are a class of polyphenolic secondary metabolites found in plants, and thus commonly consumed in the diets of humans.

Polyphenols are a large family of naturally occurring phenols. They are abundant in plants and structurally diverse. Polyphenols include phenolic acids, flavonoids, tannic acid, and ellagitannin, some of which have been used historically as dyes and for tanning garments.

Catechin is a flavan-3-ol, a type of secondary metabolite providing antioxidant roles in plants. It belongs to the subgroup of polyphenols called flavonoids.

Citrus depressa (Citrus × depressa, formerly C. pectinifera, Okinawan: シークヮーサー/シークァーサー, romanized: shiikwaasa, Japanese: ヒラミレモン, romanized: hirami remon or シークヮーサー, shiikwāsā, in English sometimes called shiikuwasha, shequasar, Taiwan tangerine, Okinawa lime, flat lemon, hirami lemon, or thin-skinned flat lemon, is a small citrus fruit often harvested and used when green, rich in flavonoids and native to East Asia.
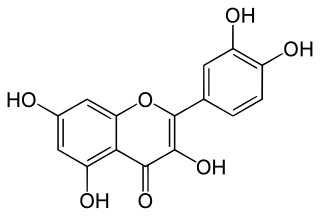
Quercetin is a plant flavonol from the flavonoid group of polyphenols. It is found in many fruits, vegetables, leaves, seeds, and grains; capers, red onions, and kale are common foods containing appreciable amounts of it. It has a bitter flavor and is used as an ingredient in dietary supplements, beverages, and foods.

Cranberry juice is the liquid juice of the cranberry – a fruit recognized for its bright red color, tart taste, and versatility for product manufacturing. Major cranberry products include cranberry juice, dried cranberry, cranberry sauce, frozen cranberry, cranberry powder, and dietary supplements containing cranberry extracts.
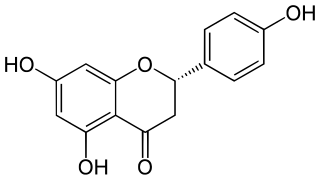
Naringenin is a flavanone from the flavonoid group of polyphenols. It is commonly found in citrus fruits, especially as the predominant flavonone in grapefruit.

Rutin is the glycoside combining the flavonol quercetin and the disaccharide rutinose. It is a flavonoid glycoside found in a wide variety of plants, including citrus.

Kaempferol (3,4′,5,7-tetrahydroxyflavone) is a natural flavonol, a type of flavonoid, found in a variety of plants and plant-derived foods including kale, beans, tea, spinach, and broccoli. Kaempferol is a yellow crystalline solid with a melting point of 276–278 °C (529–532 °F). It is slightly soluble in water and highly soluble in hot ethanol, ethers, and DMSO. Kaempferol is named for 17th-century German naturalist Engelbert Kaempfer.

A polyphenol antioxidant is a hypothetized type of antioxidant, in which each instance would contain a polyphenolic substructure; such instances which have been studied in vitro. Numbering over 4,000 distinct chemical structures, such polyphenols may have antioxidant activity {{{1}}} in vitro (although they are unlikely to be antioxidants in vivo). Hypothetically, they may affect cell-to-cell signaling, receptor sensitivity, inflammatory enzyme activity or gene regulation, although high-quality clinical research has not confirmed any of these possible effects in humans as of 2020.

Flavones are a class of flavonoids based on the backbone of 2-phenylchromen-4-one (2-phenyl-1-benzopyran-4-one).

Anthocyanins, also called anthocyans, are water-soluble vacuolar pigments that, depending on their pH, may appear red, purple, blue, or black. In 1835, the German pharmacist Ludwig Clamor Marquart named a chemical compound that gives flowers a blue color, Anthokyan, in his treatise "Die Farben der Blüthen". Food plants rich in anthocyanins include the blueberry, raspberry, black rice, and black soybean, among many others that are red, blue, purple, or black. Some of the colors of autumn leaves are derived from anthocyanins.

Phenolic compounds—natural phenol and polyphenols—occur naturally in wine. These include a large group of several hundred chemical compounds that affect the taste, color and mouthfeel of wine. These compounds include phenolic acids, stilbenoids, flavonols, dihydroflavonols, anthocyanins, flavanol monomers (catechins) and flavanol polymers (proanthocyanidins). This large group of natural phenols can be broadly separated into two categories, flavonoids and non-flavonoids. Flavonoids include the anthocyanins and tannins which contribute to the color and mouthfeel of the wine. The non-flavonoids include the stilbenoids such as resveratrol and phenolic acids such as benzoic, caffeic and cinnamic acids.

Fisetin (7,3′,4′-flavon-3-ol) is a plant flavonol from the flavonoid group of polyphenols. It can be found in many plants, where it serves as a yellow/ochre colouring agent. It is also found in many fruits and vegetables, such as strawberries, apples, persimmons, onions and cucumbers. Its chemical formula was first described by Austrian chemist Josef Herzig in 1891.

3-Hydroxyflavone is a chemical compound. It is the backbone of all flavonols, a type of flavonoid. It is a synthetic compound, which is not found naturally in plants. It serves as a model molecule as it possesses an excited-state intramolecular proton transfer (ESIPT) effect to serve as a fluorescent probe to study membranes for example or intermembrane proteins. The green tautomer emission and blue-violet normal emission originate from two different ground state populations of 3HF molecules. The phenomenon also exists in natural flavonols. Although 3-hydroxyflavone is almost insoluble in water, its aqueous solubility can be increased by encapsulation in cyclodextrin cavities.

Syringetin is an O-methylated flavonol, a type of flavonoid. It is found in red grape, in Lysimachia congestiflora and in Vaccinium uliginosum. It is one of the phenolic compounds present in wine.

In biochemistry, naturally occurring phenols are natural products containing at least one phenol functional group. Phenolic compounds are produced by plants and microorganisms. Organisms sometimes synthesize phenolic compounds in response to ecological pressures such as pathogen and insect attack, UV radiation and wounding. As they are present in food consumed in human diets and in plants used in traditional medicine of several cultures, their role in human health and disease is a subject of research. Some phenols are germicidal and are used in formulating disinfectants.

Miquelianin is a flavonol glucuronide, a type of phenolic compound present in wine, in species of St John's wort, like Hypericum hirsutum, in Nelumbo nucifera or in green beans.

Sanguiin H-6 is an ellagitannin.



















