
Hepatitis C is an infectious disease caused by the hepatitis C virus (HCV) that primarily affects the liver; it is a type of viral hepatitis. During the initial infection period, people often have mild or no symptoms. Early symptoms can include fever, dark urine, abdominal pain, and yellow tinged skin. The virus persists in the liver, becoming chronic, in about 70% of those initially infected. Early on, chronic infection typically has no symptoms. Over many years however, it often leads to liver disease and occasionally cirrhosis. In some cases, those with cirrhosis will develop serious complications such as liver failure, liver cancer, or dilated blood vessels in the esophagus and stomach.
Protease inhibitors (PIs) are medications that act by interfering with enzymes that cleave proteins. Some of the most well known are antiviral drugs widely used to treat HIV/AIDS, hepatitis C and COVID-19. These protease inhibitors prevent viral replication by selectively binding to viral proteases and blocking proteolytic cleavage of protein precursors that are necessary for the production of infectious viral particles.

The hepatitis C virus (HCV) is a small, enveloped, positive-sense single-stranded RNA virus of the family Flaviviridae. The hepatitis C virus is the cause of hepatitis C and some cancers such as liver cancer and lymphomas in humans.

Ritonavir, sold under the brand name Norvir, is an antiretroviral medication used along with other medications to treat HIV/AIDS. This combination treatment is known as highly active antiretroviral therapy (HAART). Ritonavir is a protease inhibitor, though it now mainly serves to boost the potency of other protease inhibitors. It may also be used in combination with other medications to treat hepatitis C and COVID-19. It is taken by mouth.

Boceprevir is a protease inhibitor used to treat hepatitis caused by hepatitis C virus (HCV) genotype 1. It binds to the HCV nonstructural protein 3 active site.

Telaprevir (VX-950), marketed under the brand names Incivek and Incivo, is a pharmaceutical drug for the treatment of hepatitis C co-developed by Vertex Pharmaceuticals and Johnson & Johnson. It is a member of a class of antiviral drugs known as protease inhibitors. Specifically, telaprevir inhibits the hepatitis C viral enzyme NS3/4A serine protease. Telaprevir is only indicated for use against hepatitis C genotype 1 viral infections and has not been proven to be safe or effective when used for other genotypes of the virus. The standard therapy of pegylated interferon and ribavirin is less effective than telaprevir in those with genotype 1.

Sofosbuvir, sold under the brand name Sovaldi among others, is a medication used to treat hepatitis C. It is taken by mouth.

Asunaprevir is an experimental drug candidate for the treatment of hepatitis C. It was undergoing development by Bristol-Myers Squibb and has completed Phase III clinical trials in 2013.

Simeprevir, sold under the brand name Olysio among others, is a medication used in combination with other medications for the treatment of hepatitis C. It is specifically used for hepatitis C genotype 1 and 4. Medications it is used with include sofosbuvir or ribavirin and peginterferon-alfa. Cure rates are in 80s to 90s percent. It may be used in those who also have HIV/AIDS. It is taken by mouth once daily for typically 12 weeks.
Ombitasvir is an antiviral drug for the treatment of hepatitis C virus (HCV) infection by AbbVie. In the United States, it is approved by the Food and Drug Administration for use in combination with paritaprevir, ritonavir and dasabuvir in the product Viekira Pak for the treatment of HCV genotype 1, and with paritaprevir and ritonavir in the product Technivie for the treatment of HCV genotype 4.

Dasabuvir, sold under the brand name Exviera, is an antiviral medication for the treatment of hepatitis C. It is often used together with the combination medication ombitasvir/paritaprevir/ritonavir specifically for hepatitis C virus (HCV) type 1. Ribavirin may also additionally be used. These combinations result in a cure in more than 90% of people. It is taken by mouth.
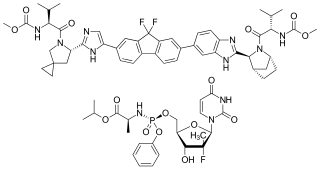
Ledipasvir/sofosbuvir, sold under the trade name Harvoni among others, is a medication used to treat hepatitis C. It is a fixed-dose combination of ledipasvir and sofosbuvir. Cure rates are 94% to 99% in people infected with hepatitis C virus (HCV) genotype 1. Some evidence also supports use in HCV genotype 3 and 4. It is taken daily by mouth for 8–24 weeks.
Ombitasvir/paritaprevir/ritonavir, sold under the brand name Technivie among others, is a medication used to treat hepatitis C. It is a fixed-dose combination of ombitasvir, paritaprevir, and ritonavir. Specifically it is used together with dasabuvir or ribavirin for cases caused by hepatitis C virus genotype 1 or 4. Cure rates are around 95%. It is taken by mouth.
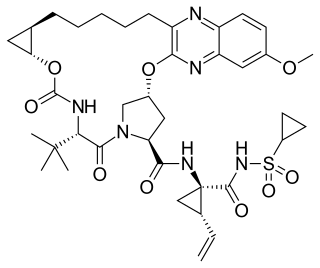
Grazoprevir is a drug approved for the treatment of hepatitis C. It was developed by Merck and completed Phase III trials, used in combination with the NS5A replication complex inhibitor elbasvir under the trade name Zepatier, either with or without ribavirin.

Elbasvir is a drug approved by the FDA in January 2016 for the treatment of hepatitis C. It was developed by Merck and completed Phase III trials, used in combination with the NS3/4A protease inhibitor grazoprevir under the trade name Zepatier, either with or without ribavirin.
Elbasvir/grazoprevir, sold under the brand name Zepatier, is a fixed-dose combination for the treatment of hepatitis C, containing elbasvir and grazoprevir. It is used to treat chronic hepatitis C virus (HCV) genotypes 1 or 4 infection in both treatment-naïve and treatment-experienced patients.
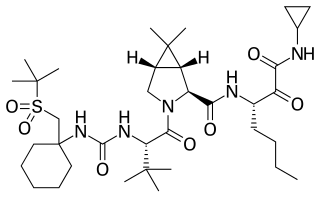
Narlaprevir, is an inhibitor of NS3/4A serine protease, intended for the treatment of chronic hepatitis C caused by genotype 1 virus in combination with other antiviral drugs.

Danoprevir (INN) is an orally available 15-membered macrocyclic peptidomimetic inhibitor of NS3/4A HCV protease. It contains acylsulfonamide, fluoroisoindole and tert-butyl carbamate moieties. Danoprevir is a clinical candidate based on its favorable potency profile against multiple HCV genotypes 1–6 and key mutants (GT1b, IC50 = 0.2–0.4 nM; replicon GT1b, EC50 = 1.6 nM). Danoprevir has been evaluated in an open-label, single arm clinical trial in combination with ritonavir for treating COVID-19 and favourably compared to lopinavir/ritonavir in a second trial.

Glecaprevir/pibrentasvir (G/P), sold under the brand names Mavyret and Maviret, is a fixed-dose combination medication used to treat hepatitis C. It contains glecaprevir and pibrentasvir. It works against all six types of hepatitis C. At twelve weeks following treatment between 81% and 100% of people have no evidence of hepatitis C. It is taken once a day by mouth with food.
Sofosbuvir/velpatasvir/voxilaprevir, sold under the brand name Vosevi, is a fixed-dose combination medication for the treatment of hepatitis C. It contains sofosbuvir, a hepatitis C virus (HCV) nucleotide analog NS5B polymerase inhibitor; velpatasvir, an HCV NS5A inhibitor; and voxilaprevir an HCV NS3/4A protease inhibitor.














