
A phosphodiesterase inhibitor is a drug that blocks one or more of the five subtypes of the enzyme phosphodiesterase (PDE), thereby preventing the inactivation of the intracellular second messengers, cyclic adenosine monophosphate (cAMP) and cyclic guanosine monophosphate (cGMP) by the respective PDE subtype(s). The ubiquitous presence of this enzyme means that non-specific inhibitors have a wide range of actions, the actions in the heart, and lungs being some of the first to find a therapeutic use.
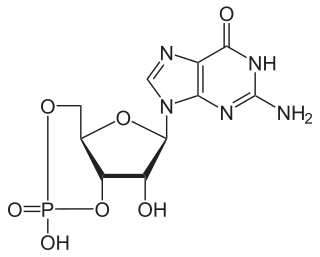
A phosphodiesterase (PDE) is an enzyme that breaks a phosphodiester bond. Usually, phosphodiesterase refers to cyclic nucleotide phosphodiesterases, which have great clinical significance and are described below. However, there are many other families of phosphodiesterases, including phospholipases C and D, autotaxin, sphingomyelin phosphodiesterase, DNases, RNases, and restriction endonucleases, as well as numerous less-well-characterized small-molecule phosphodiesterases.

Cyclic guanosine monophosphate-specific phosphodiesterase type 5 is an enzyme from the phosphodiesterase class. It is found in various tissues, most prominently the corpus cavernosum and the retina. It has also been recently discovered to play a vital role in the cardiovascular system.

Rolipram is a selective phosphodiesterase-4 inhibitor discovered and developed by Schering AG as a potential antidepressant drug in the early 1990s. It served as a prototype molecule for several companies' drug discovery and development efforts. Rolipram was discontinued after clinical trials showed that its therapeutic window was too narrow; it could not be dosed at high enough levels to be effective without causing significant gastrointestinal side effects.

PDE3 is a phosphodiesterase. The PDEs belong to at least eleven related gene families, which are different in their primary structure, substrate affinity, responses to effectors, and regulation mechanism. Most of the PDE families are composed of more than one gene. PDE3 is clinically significant because of its role in regulating heart muscle, vascular smooth muscle and platelet aggregation. PDE3 inhibitors have been developed as pharmaceuticals, but their use is limited by arrhythmic effects and they can increase mortality in some applications.
Phosphodiesterase 1, PDE1, EC 3.1.4.1, systematic name oligonucleotide 5′-nucleotidohydrolase) is a phosphodiesterase enzyme also known as calcium- and calmodulin-dependent phosphodiesterase. It is one of the 11 families of phosphodiesterase (PDE1-PDE11). Phosphodiesterase 1 has three subtypes, PDE1A, PDE1B and PDE1C which divide further into various isoforms. The various isoforms exhibit different affinities for cAMP and cGMP.

The PDE2 enzyme is one of 21 different phosphodiesterases (PDE) found in mammals. These different PDEs can be subdivided to 11 families. The different PDEs of the same family are functionally related despite the fact that their amino acid sequences show considerable divergence. The PDEs have different substrate specificities. Some are cAMP selective hydrolases, others are cGMP selective hydrolases and the rest can hydrolyse both cAMP and cGMP.
cAMP-specific 3',5'-cyclic phosphodiesterase 4D is an enzyme that in humans is encoded by the PDE4D gene.

Ibudilast is an anti-inflammatory drug used mainly in Japan, which acts as a phosphodiesterase inhibitor, inhibiting the PDE4 subtype to the greatest extent, but also showing significant inhibition of other PDE subtypes.

cAMP-specific 3',5'-cyclic phosphodiesterase 4A is an enzyme that in humans is encoded by the PDE4A gene.

cAMP-specific 3',5'-cyclic phosphodiesterase 4B is an enzyme that in humans is encoded by the PDE4B gene.

cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A is an enzyme that in humans is encoded by the PDE10A gene.
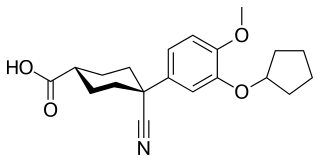
Cilomilast is a drug which was developed for the treatment of respiratory disorders such as asthma and chronic obstructive pulmonary disease (COPD). It is orally active and acts as a selective phosphodiesterase-4 inhibitor.

Roflumilast, sold under the trade name Daxas among others, is a drug that acts as a selective, long-acting inhibitor of the enzyme phosphodiesterase-4 (PDE-4). It has anti-inflammatory effects and is used as an orally administered drug for the treatment of inflammatory conditions of the lungs such as chronic obstructive pulmonary disease (COPD).

Piclamilast, is a selective PDE4 inhibitor. It is comparable to other PDE4 inhibitors for its anti-inflammatory effects. It has been investigated for its applications to the treatment of conditions such as chronic obstructive pulmonary disease, bronchopulmonary dysplasia and asthma. It is a second generation compound that exhibits structural functionalities of the PDE4 inhibitors cilomilast and roflumilast. The structure for piclamilast was first elucidated in a 1995 European patent application. The earliest mention of the name "piclamilast" was used in a 1997 publication.
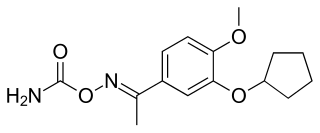
Filaminast was a drug candidate developed by Wyeth-Ayerst. It is a phosphodiesterase 4 inhibitor and an analog of rolipram, which served as a prototype molecule for several development efforts. It was discontinued after a Phase II trial showed that its therapeutic window was too narrow; it could not be dosed high enough without causing significant side effects, which was a problem with the rolipram class of molecules.
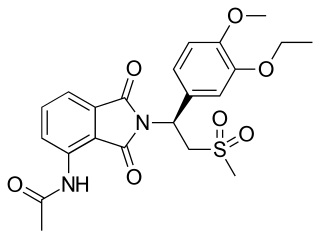
Apremilast, sold under the brand name Otezla among others, is a medication for the treatment of certain types of psoriasis and psoriatic arthritis. The drug acts as a selective inhibitor of the enzyme phosphodiesterase 4 (PDE4) and inhibits spontaneous production of TNF-alpha from human rheumatoid synovial cells. It is taken by mouth.
Phosphodiesterases (PDEs) are a superfamily of enzymes. This superfamily is further classified into 11 families, PDE1 - PDE11, on the basis of regulatory properties, amino acid sequences, substrate specificities, pharmacological properties and tissue distribution. Their function is to degrade intracellular second messengers such as cyclic adenine monophosphate (cAMP) and cyclic guanosine monophosphate (cGMP) which leads to several biological processes like effect on intracellular calcium level by the Ca2+ pathway.

Immunomodulatory imide drugs (IMiDs) are a class of immunomodulatory drugs containing an imide group. The IMiD class includes thalidomide and its analogues. These drugs may also be referred to as 'Cereblon modulators'. Cereblon (CRBN) is the protein targeted by this class of drugs.

Crisaborole, sold under the brand name Eucrisa among others, is a nonsteroidal topical medication used for the treatment of mild-to-moderate atopic dermatitis (eczema) in adults and children.

















