| |
| Clinical data | |
|---|---|
| Trade names | Agrylin |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601020 |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Liver, partially through CYP1A2 |
| Elimination half-life | 1.3 hours |
| Excretion | Urine (<1%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.317.113 |
| Chemical and physical data | |
| Formula | C10H7Cl2N3O |
| Molar mass | 256.09 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
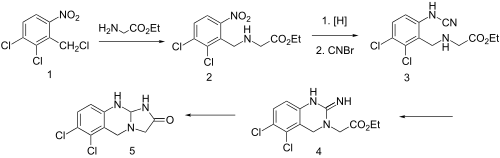
Anagrelide (Agrylin/Xagrid, Shire and Thromboreductin, AOP Orphan Pharmaceuticals AG) is a drug used for the treatment of essential thrombocytosis (also known as essential thrombocythemia), or overproduction of blood platelets. It has also been used in the treatment of chronic myeloid leukemia. [2]
Contents
Anagrelide controlled release (GALE-401) is in phase III clinical trials by Galena Biopharma for the treatment of essential thrombocytosis. [3]