
A phosphodiesterase inhibitor is a drug that blocks one or more of the five subtypes of the enzyme phosphodiesterase (PDE), thereby preventing the inactivation of the intracellular second messengers, cyclic adenosine monophosphate (cAMP) and cyclic guanosine monophosphate (cGMP) by the respective PDE subtype(s). The ubiquitous presence of this enzyme means that non-specific inhibitors have a wide range of actions, the actions in the heart, and lungs being some of the first to find a therapeutic use.

Xanthine is a purine base found in most human body tissues and fluids, as well as in other organisms. Several stimulants are derived from xanthine, including caffeine, theophylline, and theobromine.

Adenosine (symbol A) is an organic compound that occurs widely in nature in the form of diverse derivatives. The molecule consists of an adenine attached to a ribose via a β-N9-glycosidic bond. Adenosine is one of the four nucleoside building blocks of RNA (and its derivative deoxyadenosine is a building block of DNA), which are essential for all life. Its derivatives include the energy carriers adenosine mono-, di-, and triphosphate, also known as AMP/ADP/ATP. Cyclic adenosine monophosphate (cAMP) is pervasive in signal transduction. Adenosine is used as an intravenous medication for some cardiac arrhythmias.

The adenosine receptors (or P1 receptors) are a class of purinergic G protein-coupled receptors with adenosine as the endogenous ligand. There are four known types of adenosine receptors in humans: A1, A2A, A2B and A3; each is encoded by a different gene.

Aminophylline is a compound of the bronchodilator theophylline with ethylenediamine in 2:1 ratio. The ethylenediamine improves solubility, and the aminophylline is usually found as a dihydrate.

Paraxanthine, or 1,7-dimethylxanthine, is a dimethyl derivative of xanthine, structurally related to caffeine.
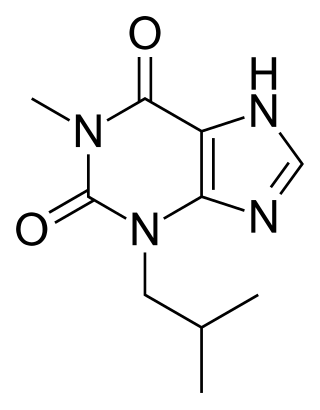
IBMX (3-isobutyl-1-methylxanthine), like other methylated xanthine derivatives, is both a:
- competitive non-selective phosphodiesterase inhibitor which raises intracellular cAMP, activates PKA, inhibits TNFα and leukotriene synthesis, and reduces inflammation and innate immunity, and
- nonselective adenosine receptor antagonist.

8-Cyclopentyl-1,3-dipropylxanthine (DPCPX, PD-116,948) is a drug which acts as a potent and selective antagonist for the adenosine A1 receptor. It has high selectivity for A1 over other adenosine receptor subtypes, but as with other xanthine derivatives DPCPX also acts as a phosphodiesterase inhibitor, and is almost as potent as rolipram at inhibiting PDE4. It has been used to study the function of the adenosine A1 receptor in animals, which has been found to be involved in several important functions such as regulation of breathing and activity in various regions of the brain, and DPCPX has also been shown to produce behavioural effects such as increasing the hallucinogen-appropriate responding produced by the 5-HT2A agonist DOI, and the dopamine release induced by MDMA, as well as having interactions with a range of anticonvulsant drugs.

SCH-58261 is a drug which acts as a potent and selective antagonist for the adenosine receptor A2A, with more than 50x selectivity for A2A over other adenosine receptors. It has been used to investigate the mechanism of action of caffeine, which is a mixed A1 / A2A antagonist, and has shown that the A2A receptor is primarily responsible for the stimulant and ergogenic effects of caffeine, but blockade of both A1 and A2A receptors is required to accurately replicate caffeine's effects in animals. SCH-58261 has also shown antidepressant, nootropic and neuroprotective effects in a variety of animal models, and has been investigated as a possible treatment for Parkinson's disease.

Doxofylline is a phosphodiesterase inhibiting bronchodilator used in the treatment of chronic respiratory diseases such as asthma and COPD. Like theophylline, it is a xanthine derivative.
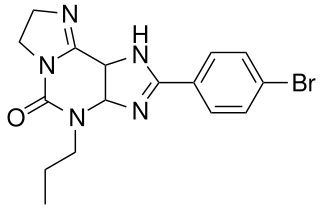
KF-26777 is a drug which acts as a potent and selective antagonist for the adenosine A3 receptor, with sub-nanomolar affinity (A3 Ki=0.2nM) and high selectivity over the other three adenosine receptor subtypes. Simple xanthine derivatives such as caffeine and DPCPX have generally low affinity for the A3 subtype and must be extended by expanding the ring system and adding an aromatic group to give high A3 affinity and selectivity.

PSB-10 is a drug which acts as a selective antagonist for the adenosine A3 receptor (ki value at human A3 receptor is 0.44 nM), with high selectivity over the other three adenosine receptor subtypes (ki values at human A1, A2A and A2B receptors are 4.1, 3.3 and 30 μM). Further pharmacological experiments in a [35S]GTPγS binding assay using hA3-CHO-cells indicated that PSB-10 acts as an inverse agonist (IC50 = 4 nM). It has been shown to produce antiinflammatory effects in animal studies. Simple xanthine derivatives such as caffeine and DPCPX have generally low affinity for the A3 subtype and must be extended by expanding the ring system and adding an aromatic group to give high A3 affinity and selectivity. The affinity towards adenosine A3 subtype was measured against the radioligand PSB-11.

Bamifylline is a drug of the xanthine chemical class which acts as a selective adenosine A1 receptor antagonist.

Diprophylline (INN) or dyphylline (USAN), is a xanthine derivative with bronchodilator and vasodilator effects. It is used in the treatment of respiratory disorders like asthma, cardiac dyspnea, and bronchitis. It acts as an adenosine receptor antagonist and phosphodiesterase inhibitor.

Cartazolate (SQ-65,396) is a drug of the pyrazolopyridine class. It acts as a GABAA receptor positive allosteric modulator at the barbiturate binding site of the complex and has anxiolytic effects in animals. It is also known to act as an adenosine antagonist at the A1 and A2 subtypes and as a phosphodiesterase inhibitor. Cartazolate was tested in human clinical trials and was found to be efficacious for anxiety but was never marketed. It was developed by a team at E.R. Squibb and Sons in the 1970s.

CGS-15943 is a drug which acts as a potent and reasonably selective antagonist for the adenosine receptors A1 and A2A, having a Ki of 3.3nM at A2A and 21nM at A1. It was one of the first adenosine receptor antagonists discovered that is not a xanthine derivative, instead being a triazoloquinazoline. Consequently, CGS-15943 has the advantage over most xanthine derivatives that it is not a phosphodiesterase inhibitor, and so has more a specific pharmacological effects profile. It produces similar effects to caffeine in animal studies, though with higher potency.

8-Cyclopentyl-1,3-dimethylxanthine (8-Cyclopentyltheophylline, 8-CPT, CPX) is a drug which acts as a potent and selective antagonist for the adenosine receptors, with some selectivity for the A1 receptor subtype, as well as a non-selective phosphodiesterase inhibitor. It has stimulant effects in animals with slightly higher potency than caffeine.
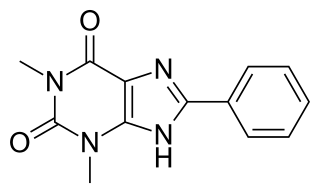
8-Phenyltheophylline (8-phenyl-1,3-dimethylxanthine, 8-PT) is a drug derived from the xanthine family which acts as a potent and selective antagonist for the adenosine receptors A1 and A2A, but unlike other xanthine derivatives has virtually no activity as a phosphodiesterase inhibitor. It has stimulant effects in animals with similar potency to caffeine. Coincidentally 8-phenyltheophylline has also been found to be a potent and selective inhibitor of the liver enzyme CYP1A2 which makes it likely to cause interactions with other drugs which are normally metabolised by CYP1A2.
An adenosine receptor antagonist is a drug which acts as an antagonist of one or more of the adenosine receptors. The best known are xanthines and their derivatives, but there are also non-xanthine representatives
Adenosine A2A receptor antagonists are a class of drugs that blocks adenosine at the adenosine A2A receptor. Notable adenosine A2A receptor antagonists include caffeine, theophylline and istradefylline.



















