| |
| Clinical data | |
|---|---|
| Trade names | Xtoro |
| Routes of administration | otic, oral, intavenous |
| ATC code |
|
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 10 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
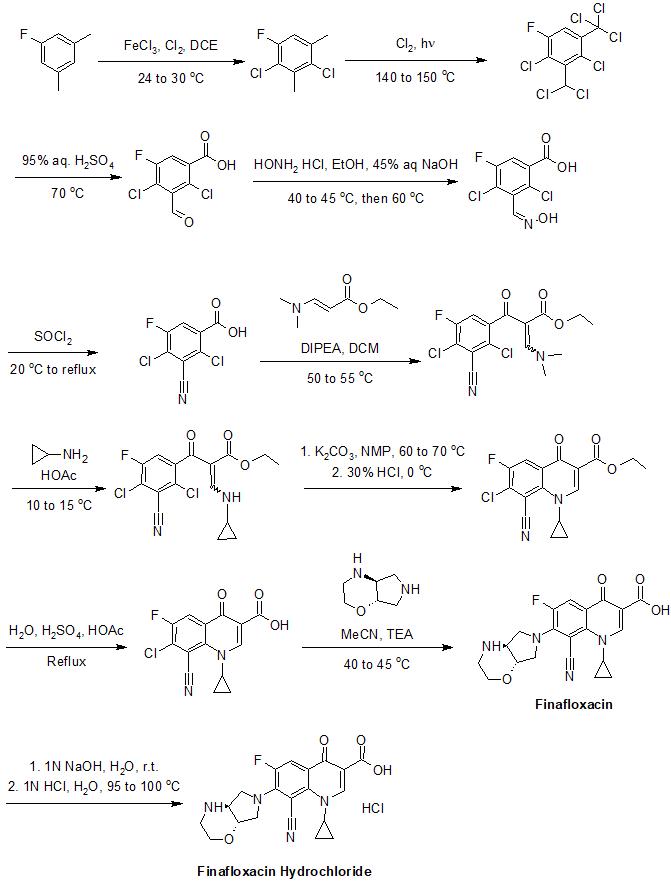
| Formula | C20H19FN4O4 |
| Molar mass | 398.394 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Finafloxacin (Xtoro) is a fluoroquinolone antibiotic. In the United States, it is approved by the Food and Drug Administration to treat acute otitis externa (swimmer's ear) caused by the bacteria Pseudomonas aeruginosa and Staphylococcus aureus . [2]