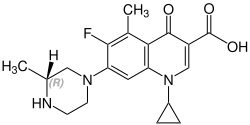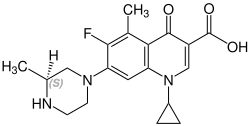 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Multum Consumer Information |
| ATC code | |
| Pharmacokinetic data | |
| Protein binding | 50% |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.159.692 |
| Chemical and physical data | |
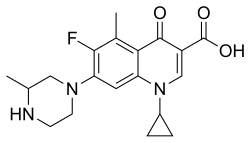
| Formula | C19H22FN3O3 |
| Molar mass | 359.401 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Grepafloxacin (trade name Raxar, Glaxo Wellcome) was an oral broad-spectrum fluoroquinolone antibacterial agent used to treat bacterial infections. Grepafloxacin was withdrawn worldwide from markets in 1999, [1] [2] due to the drug's potential to cause a potentially fatal cardiac arrhythmia. [3]