
Redox is a type of chemical reaction in which the oxidation states of substrate change.
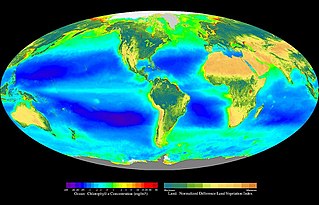
In ecology, primary production is the synthesis of organic compounds from atmospheric or aqueous carbon dioxide. It principally occurs through the process of photosynthesis, which uses light as its source of energy, but it also occurs through chemosynthesis, which uses the oxidation or reduction of inorganic chemical compounds as its source of energy. Almost all life on Earth relies directly or indirectly on primary production. The organisms responsible for primary production are known as primary producers or autotrophs, and form the base of the food chain. In terrestrial ecoregions, these are mainly plants, while in aquatic ecoregions algae predominate in this role. Ecologists distinguish primary production as either net or gross, the former accounting for losses to processes such as cellular respiration, the latter not.

The biological pump, also known as the marine carbon pump, is, in its simplest form, the ocean's biologically driven sequestration of carbon from the atmosphere and land runoff to the ocean interior and seafloor sediments. It is the part of the oceanic carbon cycle responsible for the cycling of organic matter formed mainly by phytoplankton during photosynthesis (soft-tissue pump), as well as the cycling of calcium carbonate (CaCO3) formed into shells by certain organisms such as plankton and mollusks (carbonate pump).

A biogeochemical cycle is the pathway by which a chemical substance cycles the biotic and the abiotic compartments of Earth. The biotic compartment is the biosphere and the abiotic compartments are the atmosphere, hydrosphere and lithosphere. There are biogeochemical cycles for chemical elements, such as for calcium, carbon, hydrogen, mercury, nitrogen, oxygen, phosphorus, selenium, iron and sulfur, as well as molecular cycles, such as for water and silica. There are also macroscopic cycles, such as the rock cycle, and human-induced cycles for synthetic compounds such as polychlorinated biphenyls (PCBs). In some cycles there are reservoirs where a substance can remain or be sequestered for a long period of time.
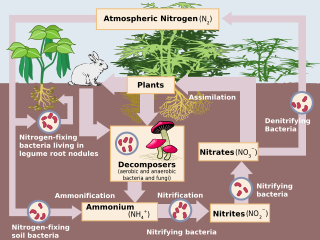
Denitrification is a microbially facilitated process where nitrate (NO3−) is reduced and ultimately produces molecular nitrogen (N2) through a series of intermediate gaseous nitrogen oxide products. Facultative anaerobic bacteria perform denitrification as a type of respiration that reduces oxidized forms of nitrogen in response to the oxidation of an electron donor such as organic matter. The preferred nitrogen electron acceptors in order of most to least thermodynamically favorable include nitrate (NO3−), nitrite (NO2−), nitric oxide (NO), nitrous oxide (N2O) finally resulting in the production of dinitrogen (N2) completing the nitrogen cycle. Denitrifying microbes require a very low oxygen concentration of less than 10%, as well as organic C for energy. Since denitrification can remove NO3−, reducing its leaching to groundwater, it can be strategically used to treat sewage or animal residues of high nitrogen content. Denitrification can leak N2O, which is an ozone-depleting substance and a greenhouse gas that can have a considerable influence on global warming.
The pedosphere is the outermost layer of the Earth that is composed of soil and subject to soil formation processes. It exists at the interface of the lithosphere, atmosphere, hydrosphere and biosphere. The pedosphere is the skin of the Earth and only develops when there is a dynamic interaction between the atmosphere, biosphere, lithosphere and the hydrosphere. The pedosphere is the foundation of terrestrial life on Earth.

Bioturbation is defined as the reworking of soils and sediments by animals or plants. It includes burrowing, ingestion, and defecation of sediment grains. Bioturbating activities have a profound effect on the environment and are thought to be a primary driver of biodiversity. The formal study of bioturbation began in the 1800s by Charles Darwin experimenting in his garden. The disruption of aquatic sediments and terrestrial soils through bioturbating activities provides significant ecosystem services. These include the alteration of nutrients in aquatic sediment and overlying water, shelter to other species in the form of burrows in terrestrial and water ecosystems, and soil production on land.

In oceanography and limnology, the sediment–water interface is the boundary between bed sediment and the overlying water column. The term usually refers to a thin layer of water at the very surface of sediments on the seafloor. In the ocean, estuaries, and lakes, this layer interacts with the water above it through physical flow and chemical reactions mediated by the micro-organisms, animals, and plants living at the bottom of the water body. The topography of this interface is often dynamic, as it is affected by physical processes and biological processes. Physical, biological, and chemical processes occur at the sediment-water interface as a result of a number of gradients such as chemical potential gradients, pore water gradients, and oxygen gradients.
Microbial metabolism is the means by which a microbe obtains the energy and nutrients it needs to live and reproduce. Microbes use many different types of metabolic strategies and species can often be differentiated from each other based on metabolic characteristics. The specific metabolic properties of a microbe are the major factors in determining that microbe's ecological niche, and often allow for that microbe to be useful in industrial processes or responsible for biogeochemical cycles.

The phosphorus cycle is the biogeochemical cycle that describes the movement of phosphorus through the lithosphere, hydrosphere, and biosphere. Unlike many other biogeochemical cycles, the atmosphere does not play a significant role in the movement of phosphorus, because phosphorus and phosphorus-based compounds are usually solids at the typical ranges of temperature and pressure found on Earth. The production of phosphine gas occurs in only specialized, local conditions. Therefore, the phosphorus cycle should be viewed from whole Earth system and then specifically focused on the cycle in terrestrial and aquatic systems.

In the deep ocean, marine snow is a continuous shower of mostly organic detritus falling from the upper layers of the water column. It is a significant means of exporting energy from the light-rich photic zone to the aphotic zone below, which is referred to as the biological pump. Export production is the amount of organic matter produced in the ocean by primary production that is not recycled (remineralised) before it sinks into the aphotic zone. Because of the role of export production in the ocean's biological pump, it is typically measured in units of carbon. The term was first coined by the explorer William Beebe as he observed it from his bathysphere. As the origin of marine snow lies in activities within the productive photic zone, the prevalence of marine snow changes with seasonal fluctuations in photosynthetic activity and ocean currents. Marine snow can be an important food source for organisms living in the aphotic zone, particularly for organisms which live very deep in the water column.

Ecosystem respiration is the sum of all respiration occurring by the living organisms in a specific ecosystem. The two main processes that contribute to ecosystem respiration are photosynthesis and cellular respiration. Photosynthesis uses carbon-dioxide and water, in the presence of sunlight to produce glucose and oxygen whereas cellular respiration uses glucose and oxygen to produce carbon-dioxide, water, and energy. The coordination of inputs and outputs of these two processes creates a completely interconnected system, constituting the underlying functioning of the ecosystems overall respiration.
The Southern Pacific Gyre is part of the Earth's system of rotating ocean currents, bounded by the Equator to the north, Australia to the west, the Antarctic Circumpolar Current to the south, and South America to the east. The center of the South Pacific Gyre is the oceanic pole of inaccessibility, the site on Earth farthest from any continents and productive ocean regions and is regarded as Earth's largest oceanic desert. The gyre, as with Earth's other four gyres, contains an area with elevated concentrations of pelagic plastics, chemical sludge, and other debris known as the South Pacific garbage patch.
Aerobic denitrification or co-respiration the simultaneous use of both oxygen (O2) and nitrate (NO3−) as oxidizing agents, performed by various genera of microorganisms. This process differs from anaerobic denitrification not only in its insensitivity to the presence of oxygen, but also in that it has a higher potential to create the harmful byproduct nitrous oxide.

A redox gradient is a series of reduction-oxidation (redox) reactions sorted according to redox potential. The redox ladder displays the order in which redox reactions occur based on the free energy gained from redox pairs. These redox gradients form both spatially and temporally as a result of differences in microbial processes, chemical composition of the environment, and oxidative potential. Common environments where redox gradients exist are coastal marshes, lakes, contaminant plumes, and soils.
Dissimilatory nitrate reduction to ammonium (DNRA), also known as nitrate/nitrite ammonification, is the result of anaerobic respiration by chemoorganoheterotrophic microbes using nitrate (NO3−) as an electron acceptor for respiration. In anaerobic conditions microbes which undertake DNRA oxidise organic matter and use nitrate (rather than oxygen) as an electron acceptor, reducing it to nitrite, then ammonium (NO3−→NO2−→NH4+).
The sulfate-methane transition zone (SMTZ) is a zone in oceans, lakes, and rivers found below the sediment surface in which sulfate and methane coexist. The formation of a SMTZ is driven by the diffusion of sulfate down the sediment column and the diffusion of methane up the sediments. At the SMTZ, their diffusion profiles meet and sulfate and methane react with one another, which allows the SMTZ to harbor a unique microbial community whose main form of metabolism is anaerobic oxidation of methane (AOM). The presence of AOM marks the transition from dissimilatory sulfate reduction to methanogenesis as the main metabolism utilized by organisms.
An oxygen minimum zone (OMZ) is characterized as an oxygen-deficient layer in the world oceans. Typically found between 200m to 1500m deep below regions of high productivity, such as the western coasts of continents. OMZs can be seasonal following the spring-summer upwelling season. Upwelling of nutrient-rich water leads to high productivity and labile organic matter, that is respired by heterotrophs as it sinks down the water column. High respiration rates deplete the oxygen in the water column to concentrations of 2 mg/L or less forming the OMZ. OMZs are expanding, with increasing ocean deoxygenation. Under these oxygen-starved conditions, energy is diverted from higher trophic levels to microbial communities that have evolved to use other biogeochemical species instead of oxygen, these species include Nitrate, Nitrite, Sulphate etc. Several Bacteria and Archea have adapted to live in these environments by using these alternate chemical species and thrive. The most abundant phyla in OMZs are Pseudomonadota, Bacteroidota, Actinomycetota, and Planctomycetota.
The deep biosphere is the part of the biosphere that resides below the first few meters of the surface. It extends down at least 5 kilometers below the continental surface and 10.5 kilometers below the sea surface, at temperatures that may reach beyond 120 °C, which is comparable to the maximum temperature where a metabolically active organism has been found. It includes all three domains of life and the genetic diversity rivals that on the surface.

Benthic-pelagic coupling are processes that connect the benthic zone and the pelagic zone through the exchange of energy, mass, or nutrients. These processes play a prominent role in both freshwater and marine ecosystems and are influenced by a number of chemical, biological, and physical forces that are crucial to functions from nutrient cycling to energy transfer in food webs.






















