Related Research Articles
Reverse-transcriptase inhibitors (RTIs) are a class of antiretroviral drugs used to treat HIV infection or AIDS, and in some cases hepatitis B. RTIs inhibit activity of reverse transcriptase, a viral DNA polymerase that is required for replication of HIV and other retroviruses.

Emtricitabine, with trade name Emtriva, is a nucleoside reverse-transcriptase inhibitor (NRTI) for the prevention and treatment of HIV infection in adults and children. In 2019, it was the 494th most commonly prescribed medication in the United States, with more than 3 thousand prescriptions.
ATC code J05Antivirals for systemic use is a therapeutic subgroup of the Anatomical Therapeutic Chemical Classification System, a system of alphanumeric codes developed by the World Health Organization (WHO) for the classification of drugs and other medical products. Subgroup J05 is part of the anatomical group J Antiinfectives for systemic use.
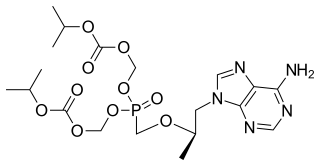
Tenofovir disoproxil, sold under the trade name Viread among others, is a medication used to treat chronic hepatitis B and to prevent and treat HIV/AIDS. It is generally recommended for use with other antiretrovirals. It may be used for prevention of HIV/AIDS among those at high risk before exposure, and after a needlestick injury or other potential exposure. It is sold both by itself and together in combinations such as emtricitabine/tenofovir, efavirenz/emtricitabine/tenofovir, and elvitegravir/cobicistat/emtricitabine/tenofovir. It does not cure HIV/AIDS or hepatitis B. It is available by mouth as a tablet or powder.

Emtricitabine/tenofovir, sold under the brand name Truvada among others, is a fixed-dose combination antiretroviral medication used to treat and prevent HIV/AIDS. It contains the antiretroviral medications emtricitabine and tenofovir disoproxil. For treatment, it must be used in combination with other antiretroviral medications. For prevention before exposure, in those who are at high risk, it is recommended along with safer sex practices. It does not cure HIV/AIDS. Emtricitabine/tenofovir is taken by mouth.

Efavirenz/emtricitabine/tenofovir, sold under the brand name Atripla among others, is a fixed-dose combination antiretroviral medication used to treat HIV/AIDS. It contains efavirenz, emtricitabine, and tenofovir disoproxil. It can be used by itself or together with other antiretroviral medications. It is taken by mouth.

Raltegravir, sold under the brand name Isentress, is an antiretroviral medication used, together with other medication, to treat HIV/AIDS. It may also be used, as part of post exposure prophylaxis, to prevent HIV infection following potential exposure. It is taken by mouth.

Elvitegravir (EVG) is an integrase inhibitor used to treat HIV infection. It was developed by the pharmaceutical company Gilead Sciences, which licensed EVG from Japan Tobacco in March 2008. The drug gained approval by the U.S. Food and Drug Administration on August 27, 2012, for use in adult patients starting HIV treatment for the first time as part of the fixed dose combination known as Stribild. On September 24, 2014, the FDA approved Elvitegravir as a single pill formulation under the trade name Vitekta. On November 5, 2015, the FDA approved the drug for use in patients affected with HIV-1 as a part of a second fixed dose combination pill known as Genvoya.

Rilpivirine, sold under the brand names Edurant and Rekambys, is a medication, developed by Tibotec, used for the treatment of HIV/AIDS. It is a second-generation non-nucleoside reverse transcriptase inhibitor (NNRTI) with higher potency, longer half-life and reduced side-effect profile compared with older NNRTIs such as efavirenz.

Cobicistat, sold under the brand name Tybost, is a medication for use in the treatment of human immunodeficiency virus infection (HIV/AIDS). Its major mechanism of action is through the inhibition of human CYP3A proteins.
Emtricitabine/rilpivirine/tenofovir is a fixed-dose combination of antiretroviral drugs for the treatment of HIV/AIDS. The drug was co-developed by Gilead Sciences and Johnson & Johnson's Tibotec division and was approved by the Food and Drug Administration in August 2011, and by the European Medicines Agency in November 2011, for patients who have not previously been treated for HIV. It is available as a once-a-day single tablet.
The first human immunodeficiency virus (HIV) case was reported in the United States in the early 1980s. Many drugs have been discovered to treat the disease but mutations in the virus and resistance to the drugs make development difficult. Integrase is a viral enzyme that integrates retroviral DNA into the host cell genome. Integrase inhibitors are a new class of drugs used in the treatment of HIV. The first integrase inhibitor, raltegravir, was approved in 2007 and other drugs were in clinical trials in 2011.
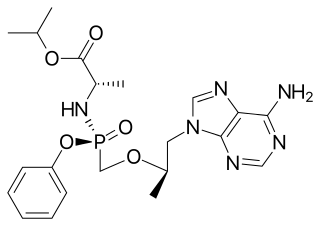
Tenofovir alafenamide, sold under the brand name Vemlidy, is an antiviral medication used against hepatitis B and HIV. It is used for the treatment of chronic hepatitis B virus (HBV) infection in adults with compensated liver disease and is given in combination with other medications for the prevention and treatment of HIV. It is taken by mouth.
Elvitegravir/cobicistat/emtricitabine/tenofovir, sold under the brand name Stribild, also known as the Quadpill, is a fixed-dose combination antiretroviral medication for the treatment of HIV/AIDS. Elvitegravir, emtricitabine and tenofovir disoproxil directly suppress viral reproduction. Cobicistat increases the effectiveness of the combination by inhibiting the liver and gut wall enzymes that metabolize elvitegravir. It is taken by mouth.

Cabotegravir, sold under the brand name Vocabria among others, is a antiretroviral medication used for the treatment of HIV/AIDS. It is available in the form of tablets and as an intramuscular injection, as well as in an injectable combination with rilpivirine under the brand name Cabenuva.
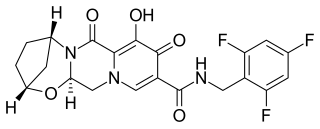
Bictegravir is a second-generation integrase inhibitor (INSTI) class that was structurally derived from an earlier compound dolutegravir by scientists at Gilead Sciences. In vitro and clinical results were presented by Gilead in the summer of 2016. In 2016, bictegravir was in a Phase 3 trial as part of a single tablet regimen in combination with tenofovir alafenamide (TAF) and emtricitabine (FTC) for the treatment of HIV-1 infection and the combination drug bictegravir/emtricitabine/tenofovir alafenamide (Biktarvy) was approved for use in the United States in 2018.

Bictegravir/emtricitabine/tenofovir alafenamide, sold under the brand name Biktarvy, is a fixed-dose combination antiretroviral medication for the treatment of HIV/AIDS. One tablet, taken orally once daily, contains 50 mg bictegravir, 200 mg emtricitabine, and 25 mg tenofovir alafenamide. It was approved for use in the United States in February 2018, and for use in the European Union in June 2018.
Dolutegravir/lamivudine, sold under the brand name Dovato, is a fixed-dose combination antiretroviral medication for the treatment of HIV/AIDS. It contains dolutegravir, as the salt, an integrase strand transfer inhibitor (INSTI), and lamivudine, a nucleoside analogue reverse transcriptase inhibitor (NRTI). It is taken by mouth.

Chloe Meave Orkin is a British physician and Professor of HIV/AIDS medicine at Queen Mary University of London. She works as a consultant at the Royal London Hospital, Barts Health NHS Trust. She is an internationally renowned expert in HIV therapeutics and led the first phase III clinical trial of injectable anti-retrovirals. She is immediate past Chair of the British HIV Association, where she championed the Undetectable=Untransmittable (U=U) campaign within the United Kingdom. She is president elect of the Medical Women's Federation. Orkin is gay and was on the Top 100 Lesbian influencer lists in both the UK and in the US in 2020. She considers herself a medical activist and much of her work focuses on inequalities in healthcare and in Medicine.

Cabotegravir/rilpivirine, sold under the brand name Cabenuva, is a co-packaged antiretroviral medication for the treatment of HIV/AIDS. It contains cabotegravir and rilpivirine in a package with two separate injection vials.
References
- ↑ "FDA approval of Isentress (raltegravir)". U.S. Food and Drug Administration. October 12, 2007. Archived from the original on January 12, 2017. Retrieved October 8, 2019.
- ↑ Steigbigel RT, Cooper DA, Kumar PN, et al. (July 2008). "Raltegravir with optimized background therapy for resistant HIV-1 infection". N. Engl. J. Med. 359 (4): 339–54. doi: 10.1056/NEJMoa0708975 . PMID 18650512. S2CID 17257548.
- ↑ USA (2014). "Antiretroviral Therapy: Current Drugs". Infectious Disease Clinics of North America. Ncbi.nlm.nih.gov. 28 (3): 371–402. doi:10.1016/j.idc.2014.06.001. PMC 4143801 . PMID 25151562.
- ↑ "Elvitegravir". AIDSinfo. National Institutes of Health. Archived from the original on 2011-09-27. Retrieved 2007-10-13.
- ↑ "U.S. Food and Drug Administration Approves Gilead's Biktarvy (Bictegravir, Emtricitabine, Tenofovir Alafenamide) for Treatment of HIV-1 Infection" (Press release). Gilead. February 7, 2018.