Related Research Articles
Endorphins are endogenous opioid neuropeptides and peptide hormones in humans and other animals. They are produced and stored in the pituitary gland. The classification of molecules as endorphins is based on their pharmacological activity, as opposed to a specific chemical formulation.

An enkephalin is a pentapeptide involved in regulating nociception in the body. The enkephalins are termed endogenous ligands, as they are internally derived and bind to the body's opioid receptors. Discovered in 1975, two forms of enkephalin have been found, one containing leucine ("leu"), and the other containing methionine ("met"). Both are products of the proenkephalin gene.

Beta-Endorphin or β-Endorphin is an endogenous opioid neuropeptide and peptide hormone that is produced in certain neurons within the central nervous system and peripheral nervous system. It is one of three endorphins that are produced in humans, the others of which include α-endorphin and γ-endorphin.

Opioid peptides are peptides that bind to opioid receptors in the brain; opiates and opioids mimic the effect of these peptides. Such peptides may be produced by the body itself, for example endorphins. The effects of these peptides vary, but they all resemble those of opiates. Brain opioid peptide systems are known to play an important role in motivation, emotion, attachment behaviour, the response to stress and pain, and the control of food intake.
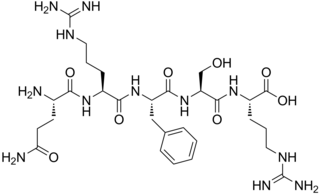
Opiorphin is an endogenous chemical compound first isolated from human saliva. Initial research with mice shows the compound has a painkilling effect greater than that of morphine. It works by stopping the normal breakup of enkephalins, natural pain-killing opioids in the spinal cord. It is a relatively simple molecule consisting of a five-amino acid polypeptide, Gln-Arg-Phe-Ser-Arg (QRFSR).

Met-enkephalin, also known as metenkefalin (INN), sometimes referred to as opioid growth factor (OGF), is a naturally occurring, endogenous opioid peptide that has opioid effects of a relatively short duration. It is one of the two forms of enkephalin, the other being leu-enkephalin. The enkephalins are considered to be the primary endogenous ligands of the δ-opioid receptor, due to their high potency and selectivity for the site over the other endogenous opioids.
Leu-enkephalin is an endogenous opioid peptide neurotransmitter with the amino acid sequence Tyr-Gly-Gly-Phe-Leu that is found naturally in the brains of many animals, including humans. It is one of the two forms of enkephalin; the other is met-enkephalin. The tyrosine residue at position 1 is thought to be analogous to the 3-hydroxyl group on morphine. Leu-enkephalin has agonistic actions at both the μ- and δ-opioid receptors, with significantly greater preference for the latter. It has little to no effect on the κ-opioid receptor.
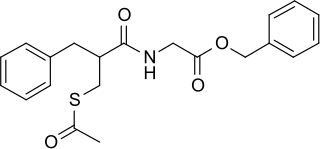
Racecadotril, also known as acetorphan, is an antidiarrheal medication which acts as a peripheral enkephalinase inhibitor. Unlike other opioid medications used to treat diarrhea, which reduce intestinal motility, racecadotril has an antisecretory effect — it reduces the secretion of water and electrolytes into the intestine. It is available in France and other European countries as well as most of South America and some South East Asian countries, but not in the United States. It is sold under the tradename Hidrasec, among others. Thiorphan is the active metabolite of racecadotril, which exerts the bulk of its inhibitory actions on enkephalinases.

RB-101 is a drug that acts as an enkephalinase inhibitor, which is used in scientific research.

Thiorphan is the active metabolite of the antidiarrheal racecadotril (acetorphan). It prevents the degradation of endogenous enkephalins by acting as an enkephalinase inhibitor.
Enkephalinases are enzymes that degrade endogenous enkephalin opioid peptides. They include:

Ecadotril is a neutral endopeptidase inhibitor and determined by the presence of peptidase family M13 as a neutral endopeptidase inhibited by phosphoramidon. Ecadotril is the (S)-enantiomer of racecadotril. NEP-like enzymes include the endothelin-converting enzymes. The peptidase M13 family believed to activate or inactivate oligopeptide (pro)-hormones such as opioid peptides, neprilysin is another member of this group, in the case of the metallopeptidases and aspartic, the nucleophiles clan or family for example MA, is an activated water molecule. The peptidase domain for members of this family also contains a bacterial member and resembles that of thermolysin the predicted active site residues for members of this family and thermolysin occur in the motif HEXXH. Thermolysin complexed with the inhibitor (S)-thiorphan are isomeric thiol-containing inhibitors of endopeptidase EC 24-11.

Kelatorphan is a drug which acts as a powerful and complete inhibitor of nearly all of the enzymes responsible for catabolism of the endogenous enkephalins, including neutral endopeptidase (NEP), dipeptidyl peptidase III (DPP3), aminopeptidase N (APN), and angiotensin-converting enzyme (ACE). In mice, with the intracerebroventricular co-administration of a 50 µg dose of kelatorphan (this route is necessary because kelatorphan is incapable of crossing the blood-brain-barrier) hence alongside exogenous [Met]enkephalin (ED50 approximately 10 ng), it potentiated the analgesic effects of the latter by 50,000 times. Kelatorphan also displays potent antinociceptive effects alone, and does not depress respiration, although at high doses it actually increases it.
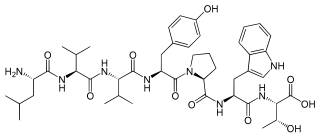
Spinorphin is an endogenous, non-classical opioid peptide of the hemorphin family first isolated from the bovine spinal cord (hence the prefix spin-) and acts as a regulator of the enkephalinases, a class of enzymes that break down endogenous the enkephalin peptides. It does so by inhibiting the enzymes aminopeptidase N (APN), dipeptidyl peptidase III (DPP3), angiotensin-converting enzyme (ACE), and neutral endopeptidase (NEP). Spinorphin is a heptapeptide and has the amino acid sequence Leu-Val-Val-Tyr-Pro-Trp-Thr (LVVYPWT). It has been observed to possess antinociceptive, antiallodynic, and anti-inflammatory properties. The mechanism of action of spinorphin has not been fully elucidated (i.e., how it acts to inhibit the enkephalinases), but it has been found to act as an antagonist of the P2X3 receptor, and as a weak partial agonist/antagonist of the FP1 receptor.

Tynorphin is a synthetic opioid peptide which is a potent and competitive inhibitor of the enkephalinase class of enzymes which break down the endogenous enkephalin peptides. It specifically inactivates dipeptidyl aminopeptidase III (DPP3) with very high efficacy, but also inhibits neutral endopeptidase (NEP), aminopeptidase N (APN), and angiotensin-converting enzyme (ACE) to a lesser extent. It has a pentapeptide structure with the amino acid sequence Val-Val-Tyr-Pro-Trp (VVYPW).

Hemorphin-4 is an endogenous opioid peptide of the hemorphin family which possesses antinociceptive properties and is derived from the β-chain of hemoglobin in the bloodstream. It is a tetrapeptide with the amino acid sequence Tyr-Pro-Trp-Thr. Hemorphin-4 has affinities for the μ-, δ-, and κ-opioid receptors that are in the same range as the structurally related β-casomorphins, although affinity to the κ-opioid receptor is markedly higher in comparison. It acts as an agonist at these sites. Hemorphin-4 also has inhibitory effects on angiotensin-converting enzyme (ACE), and as a result, may play a role in the regulation of blood pressure. Notably, inhibition of ACE also reduces enkephalin catabolism.
Hemorphins are a class of naturally occurring, endogenous opioid peptides which are found in the bloodstream, and are derived from the β-chain of hemoglobin. They have antinociceptive effects via activation of the opioid receptors, and some may also play a role in blood pressure through inhibition of the angiotensin-converting enzyme (ACE), as well as cause an elevation of endogenous enkephalin levels. Some examples of hemorphins include hemorphin-4, spinorphin, and valorphin.

RB-120 is an orally active analog of the drug RB-101. It acts as an enkephalinase inhibitor, which is used in scientific research. Via intravenous administration, it is approximately three times as potent as RB-101 or twice as potent as the isolated (S,S) isomer of RB101. However, via i.p. administration it is approximately twice as potent as racemic RB-101 and about as potent as the isolated (S,S) isomer of RB101. During i.v. administration RB120 is approximately twice as weak as morphine in terms of analgesia, however it is 16x weaker during i.p. and p.o. administration.

RB-3007 is an orally active analogue of RB-101. It acts as an enkephalinase inhibitor, which is used in scientific research.
References
- 1 2 Thanawala V, Kadam VJ, Ghosh R (October 2008). "Enkephalinase inhibitors: potential agents for the management of pain". Current Drug Targets. 9 (10): 887–94. doi:10.2174/138945008785909356. PMID 18855623. Archived from the original on 2012-03-18.