Related Research Articles
An exopeptidase is any peptidase that catalyzes the cleavage of the terminal peptide bond; the process releases a single amino acid, dipeptide or a tripeptide from the peptide chain. Depending on whether the amino acid is released from the amino or the carboxy terminal, an exopeptidase is further classified as an aminopeptidase or a carboxypeptidase, respectively. Thus, an aminopeptidase, an enzyme in the brush border of the small intestine, will cleave a single amino acid from the amino terminal, whereas carboxypeptidase, which is a digestive enzyme present in pancreatic juice, will cleave a single amino acid from the carboxylic end of the peptide.
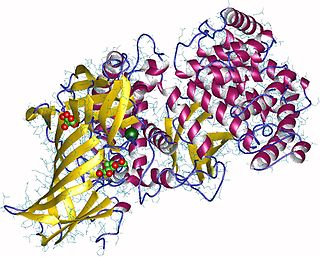
Aminopeptidases are enzymes that catalyze the cleavage of amino acids from the amino terminus (N-terminus) of proteins or peptides (exopeptidases). They are widely distributed throughout the animal and plant kingdoms and are found in many subcellular organelles, in cytosol, and as membrane components. Aminopeptidases are used in essential cellular functions. Many, but not all, of these peptidases are zinc metalloenzymes.
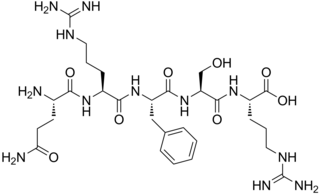
Opiorphin is an endogenous chemical compound first isolated from human saliva. Initial research with mice shows the compound has a painkilling effect greater than that of morphine. It works by stopping the normal breakup of enkephalins, natural pain-killing opioids in the spinal cord. It is a relatively simple molecule consisting of a five-amino acid polypeptide, Gln-Arg-Phe-Ser-Arg (QRFSR).

Met-enkephalin, also known as metenkefalin (INN), sometimes referred to as opioid growth factor (OGF), is a naturally occurring, endogenous opioid peptide that has opioid effects of a relatively short duration. It is one of the two forms of enkephalin, the other being leu-enkephalin. The enkephalins are considered to be the primary endogenous ligands of the δ-opioid receptor, due to their high potency and selectivity for the site over the other endogenous opioids.

Carboxypeptidase E (CPE), also known as carboxypeptidase H (CPH) and enkephalin convertase, is an enzyme that in humans is encoded by the CPE gene. This enzyme catalyzes the release of C-terminal arginine or lysine residues from polypeptides.
Glutamyl aminopeptidase (EC 3.4.11.7, aminopeptidase A, aspartate aminopeptidase, angiotensinase A, glutamyl peptidase, Ca2+-activated glutamate aminopeptidase, membrane aminopeptidase II, antigen BP-1/6C3 of mouse B lymphocytes, L-aspartate aminopeptidase, angiotensinase A2) is an enzyme encoded by the ENPEP gene. Glutamyl aminopeptidase has also recently been designated CD249 (cluster of differentiation 249).

Leucyl aminopeptidases are enzymes that preferentially catalyze the hydrolysis of leucine residues at the N-terminus of peptides and proteins. Other N-terminal residues can also be cleaved, however. LAPs have been found across superkingdoms. Identified LAPs include human LAP, bovine lens LAP, porcine LAP, Escherichia coli LAP, and the solanaceous-specific acidic LAP (LAP-A) in tomato.
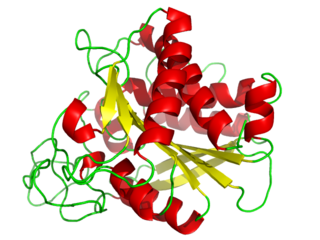
Carboxypeptidase A usually refers to the pancreatic exopeptidase that hydrolyzes peptide bonds of C-terminal residues with aromatic or aliphatic side-chains. Most scientists in the field now refer to this enzyme as CPA1, and to a related pancreatic carboxypeptidase as CPA2.

Puromycin-sensitive amino peptidase also known as cytosol alanyl aminopeptidase or alanine aminopeptidase (AAP) is an enzyme that in humans is encoded by the NPEPPS gene. It is used as a biomarker to detect damage to the kidneys, and that may be used to help diagnose certain kidney disorders. It is found at high levels in the urine when there are kidney problems.
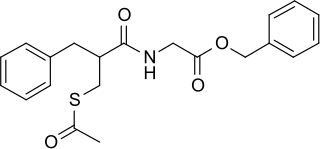
Racecadotril, also known as acetorphan, is an antidiarrheal medication which acts as a peripheral enkephalinase inhibitor. Unlike other opioid medications used to treat diarrhea, which reduce intestinal motility, racecadotril has an antisecretory effect — it reduces the secretion of water and electrolytes into the intestine. It is available in France and other European countries as well as most of South America and some South East Asian countries, but not in the United States. It is sold under the tradename Hidrasec, among others. Thiorphan is the active metabolite of racecadotril, which exerts the bulk of its inhibitory actions on enkephalinases.

RB-101 is a drug that acts as an enkephalinase inhibitor, which is used in scientific research.

Ubenimex (INN), also known more commonly as bestatin, is a competitive, reversible protease inhibitor. It is an inhibitor of arginyl aminopeptidase (aminopeptidase B), leukotriene A4 hydrolase (a zinc metalloprotease that displays both epoxide hydrolase and aminopeptidase activities), alanyl aminopeptidase (aminopeptidase M/N), leucyl/cystinyl aminopeptidase (oxytocinase/vasopressinase), and membrane dipeptidase (leukotriene D4 hydrolase). It is being studied for use in the treatment of acute myelocytic leukemia and lymphedema. It is derived from Streptomyces olivoreticuli. Ubenimex has been found to inhibit the enzymatic degradation of oxytocin, vasopressin, enkephalins, and various other peptides and compounds.

Carboxypeptidase A6 (CPA6) is a metallocarboxypeptidase enzyme that in humans is encoded by the CPA6 gene. It is highly expressed in the adult mouse olfactory bulb and is broadly expressed in the embryonic brain and other tissues.

Kelatorphan is a drug which acts as a powerful and complete inhibitor of nearly all of the enzymes responsible for catabolism of the endogenous enkephalins, including neutral endopeptidase (NEP), dipeptidyl peptidase III (DPP3), aminopeptidase N (APN), and angiotensin-converting enzyme (ACE). In mice, with the intracerebroventricular co-administration of a 50 μg dose of kelatorphan (this route is necessary because kelatorphan is incapable of crossing the blood-brain-barrier) hence alongside exogenous [Met]enkephalin (ED50 approximately 10 ng), it potentiated the analgesic effects of the latter by 50,000 times. Kelatorphan also displays potent antinociceptive effects alone, and does not depress respiration, although at high doses it actually increases it.
An enkephalinase inhibitor is a type of enzyme inhibitor which inhibits one or more members of the enkephalinase class of enzymes that break down the endogenous enkephalin opioid peptides. Examples include racecadotril, ubenimex (bestatin), RB-101, and D-phenylalanine, as well as the endogenous opioid peptides opiorphin and spinorphin. It also includes RB-3007, Semax and Selank. Analgesic, anticraving, antidepressant, anxiolytic, and antidiarrheal effects are common properties of enkephalinase inhibitors.
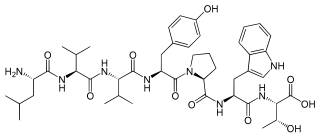
Spinorphin is an endogenous, non-classical opioid peptide of the hemorphin family first isolated from the bovine spinal cord (hence the prefix spin-) and acts as a regulator of the enkephalinases, a class of enzymes that break down endogenous the enkephalin peptides. It does so by inhibiting the enzymes aminopeptidase N (APN), dipeptidyl peptidase III (DPP3), angiotensin-converting enzyme (ACE), and neutral endopeptidase (NEP). Spinorphin is a heptapeptide and has the amino acid sequence Leu-Val-Val-Tyr-Pro-Trp-Thr (LVVYPWT). It has been observed to possess antinociceptive, antiallodynic, and anti-inflammatory properties. The mechanism of action of spinorphin has not been fully elucidated (i.e., how it acts to inhibit the enkephalinases), but it has been found to act as an antagonist of the P2X3 receptor, and as a weak partial agonist/antagonist of the FP1 receptor.

Tynorphin is a synthetic opioid peptide which is a potent and competitive inhibitor of the enkephalinase class of enzymes which break down the endogenous enkephalin peptides. It specifically inactivates dipeptidyl aminopeptidase III (DPP3) with very high efficacy, but also inhibits neutral endopeptidase (NEP), aminopeptidase N (APN), and angiotensin-converting enzyme (ACE) to a lesser extent. It has a pentapeptide structure with the amino acid sequence Val-Val-Tyr-Pro-Trp (VVYPW).
Oxytocinase is a type of enzyme that metabolizes the endogenous neuropeptide, oxytocin. The most well-characterized oxytocinase is leucyl/cystinyl aminopeptidase, which is also an enkephalinase. Other oxytocinases are also known. During pregnancy, oxytocinase plays a role in balancing concentration of oxytocin by degrading the oxytocin produced by the fetus, as production of oxytocin increases with growth of fetus. One study found that concentration level of oxytocinase increased progressively with gestational age until labor, which indicates that pregnancy development can be statistically evaluated by comparing oxytocinase levels.

Amastatin, also known as 3-amino-2-hydroxy-5-methylhexanoyl-L-valyl-L-valyl-L-aspartic acid, is a naturally occurring, competitive and reversible aminopeptidase inhibitor that was isolated from Streptomyces sp. ME 98-M3. It specifically inhibits leucyl aminopeptidase, alanyl aminopeptidase, bacterial leucyl aminopeptidase, leucyl/cystinyl aminopeptidase (oxytocinase/vasopressinase), and, to a lesser extent, glutamyl aminopeptidase, as well as other aminopeptidases. It does not inhibit arginyl aminopeptidase. Amastatin has been found to potentiate the central nervous system effects of oxytocin and vasopressin in vivo. It also inhibits the degradation of met-enkephalin, dynorphin A, and other endogenous peptides.

RB-3007 is an orally active analogue of RB-101. It acts as an enkephalinase inhibitor, which is used in scientific research.
References
- 1 2 3 Thanawala V, Kadam VJ, Ghosh R (October 2008). "Enkephalinase inhibitors: potential agents for the management of pain". Current Drug Targets. 9 (10): 887–94. doi:10.2174/138945008785909356. PMID 18855623. Archived from the original on 2013-04-14.
{{cite journal}}: CS1 maint: unfit URL (link) - ↑ Lyons PJ, Callaway MB, Fricker LD (March 2008). "Characterization of carboxypeptidase A6, an extracellular matrix peptidase". The Journal of Biological Chemistry. 283 (11): 7054–63. doi: 10.1074/jbc.M707680200 . PMID 18178555.
- ↑ Benuck M, Berg MJ, Marks N (1982). "Separate metabolic pathways for Leu-enkephalin and Met-enkephalin-Arg(6)-Phe(7) degradation by rat striatal synaptosomal membranes". Neurochemistry International. 4 (5): 389–96. doi:10.1016/0197-0186(82)90081-X. PMID 20487892. S2CID 23138078.