Endorphins are peptides produced in the brain that block the perception of pain and increase feelings of wellbeing. They are produced and stored in the pituitary gland of the brain. Endorphins are endogenous painkillers often produced in the brain and adrenal medulla during physical exercise or orgasm and inhibit pain, muscle cramps, and relieve stress.

β-Endorphin (beta-endorphin) is an endogenous opioid neuropeptide and peptide hormone that is produced in certain neurons within the central nervous system and peripheral nervous system. It is one of three endorphins that are produced in humans, the others of which include α-endorphin and γ-endorphin.
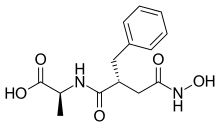
Opiorphin is an endogenous chemical compound first isolated from human saliva. Initial research with mice shows the compound has a painkilling effect greater than that of morphine. It works by stopping the normal breakup of enkephalins, natural pain-killing opioids in the spinal cord. It is a relatively simple molecule consisting of a five-amino acid polypeptide, Gln-Arg-Phe-Ser-Arg (QRFSR).

Met-enkephalin, also known as metenkefalin (INN), sometimes referred to as opioid growth factor (OGF), is a naturally occurring, endogenous opioid peptide that has opioid effects of a relatively short duration. It is one of the two forms of enkephalin, the other being leu-enkephalin. The enkephalins are considered to be the primary endogenous ligands of the δ-opioid receptor, due to their high potency and selectivity for the site over the other endogenous opioids.

Neprilysin is an enzyme that in humans is encoded by the MME gene. Neprilysin is a zinc-dependent metalloprotease that cleaves peptides at the amino side of hydrophobic residues and inactivates several peptide hormones including glucagon, enkephalins, substance P, neurotensin, oxytocin, and bradykinin. It also degrades the amyloid beta peptide whose abnormal folding and aggregation in neural tissue has been implicated as a cause of Alzheimer's disease. Synthesized as a membrane-bound protein, the neprilysin ectodomain is released into the extracellular domain after it has been transported from the Golgi apparatus to the cell surface.
Glutamyl aminopeptidase (EC 3.4.11.7, aminopeptidase A, aspartate aminopeptidase, angiotensinase A, glutamyl peptidase, Ca2+-activated glutamate aminopeptidase, membrane aminopeptidase II, antigen BP-1/6C3 of mouse B lymphocytes, L-aspartate aminopeptidase, angiotensinase A2) is an enzyme encoded by the ENPEP gene. Glutamyl aminopeptidase has also recently been designated CD249 (cluster of differentiation 249).

RB-101 is a drug that acts as an enkephalinase inhibitor, which is used in scientific research.

CI-988 (PD-134,308) is a drug which acts as a cholecystokinin antagonist, selective for the CCKB subtype. In animal studies it showed anxiolytic effects and potentiated the analgesic action of both morphine and endogenous opioid peptides, as well as preventing the development of tolerance to opioids and reducing symptoms of withdrawal. Consequently, it was hoped that it might have clinical applications for the treatment of pain and anxiety in humans, but trial results were disappointing with only minimal therapeutic effects observed even at high doses. The reason for the failure of CI-988 and other CCKB antagonists in humans despite their apparent promise in pre-clinical animal studies is unclear, although poor pharmacokinetic properties of the currently available drugs are a possible explanation, and CCKB antagonists are still being researched for possible uses as adjuvants to boost the activity of other drugs.
Enkephalinases are enzymes that degrade endogenous enkephalin opioid peptides. They include:

Ecadotril is a neutral endopeptidase inhibitor ((NEP) EC 3.4.24.11) and determined by the presence of peptidase family M13 as a neutral endopeptidase inhibited by phosphoramidon. Ecadotril is the (S)-enantiomer of racecadotril. NEP-like enzymes include the endothelin-converting enzymes. The peptidase M13 family believed to activate or inactivate oligopeptide (pro)-hormones such as opioid peptides, neprilysin is another member of this group, in the case of the metallopeptidases and aspartic, the nucleophiles clan or family for example MA, is an activated water molecule. The peptidase domain for members of this family also contains a bacterial member and resembles that of thermolysin the predicted active site residues for members of this family and thermolysin occur in the motif HEXXH. Thermolysin complexed with the inhibitor (S)-thiorphan are isomeric thiol-containing inhibitors of endopeptidase EC 24-11 (also called "enkephalinase").

An Oligopeptidase is an enzyme that cleaves peptides but not proteins. This property is due to its structure: the active site of this enzyme is located at the end of a narrow cavity which can only be reached by peptides.
An enkephalinase inhibitor is a type of enzyme inhibitor which inhibits one or more members of the enkephalinase class of enzymes that break down the endogenous enkephalin opioid peptides. Examples include racecadotril, ubenimex (bestatin), RB-101, and D-phenylalanine, as well as the endogenous opioid peptides opiorphin and spinorphin. It also includes RB-3007, Semax and Selank. Analgesic, anticraving, antidepressant, anxiolytic, and antidiarrheal effects are common properties of enkephalinase inhibitors.
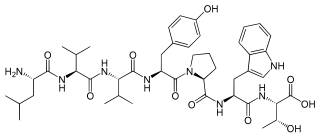
Spinorphin is an endogenous, non-classical opioid peptide of the hemorphin family first isolated from the bovine spinal cord (hence the prefix spin-) and acts as a regulator of the enkephalinases, a class of enzymes that break down endogenous the enkephalin peptides. It does so by inhibiting the enzymes aminopeptidase N (APN), dipeptidyl peptidase III (DPP3), angiotensin-converting enzyme (ACE), and neutral endopeptidase (NEP). Spinorphin is a heptapeptide and has the amino acid sequence Leu-Val-Val-Tyr-Pro-Trp-Thr (LVVYPWT). It has been observed to possess antinociceptive, antiallodynic, and anti-inflammatory properties. The mechanism of action of spinorphin has not been fully elucidated (i.e., how it acts to inhibit the enkephalinases), but it has been found to act as an antagonist of the P2X3 receptor, and as a weak partial agonist/antagonist of the FP1 receptor.

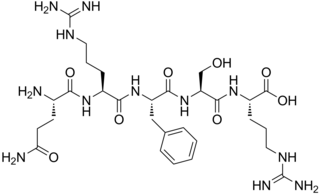
Tynorphin is a synthetic opioid peptide which is a potent and competitive inhibitor of the enkephalinase class of enzymes which break down the endogenous enkephalin peptides. It specifically inactivates dipeptidyl aminopeptidase III (DPP3) with very high efficacy, but also inhibits neutral endopeptidase (NEP), aminopeptidase N (APN), and angiotensin-converting enzyme (ACE) to a lesser extent. It has a pentapeptide structure with the amino acid sequence Val-Val-Tyr-Pro-Trp (VVYPW).

Hemorphin-4 is an endogenous opioid peptide of the hemorphin family which possesses antinociceptive properties and is derived from the β-chain of hemoglobin in the bloodstream. It is a tetrapeptide with the amino acid sequence Tyr-Pro-Trp-Thr. Hemorphin-4 has affinities for the μ-, δ-, and κ-opioid receptors that are in the same range as the structurally related β-casomorphins, although affinity to the κ-opioid receptor is markedly higher in comparison. It acts as an agonist at these sites. Hemorphin-4 also has inhibitory effects on angiotensin-converting enzyme (ACE), and as a result, may play a role in the regulation of blood pressure. Notably, inhibition of ACE also reduces enkephalin catabolism.
Hemorphins are a class of naturally occurring, endogenous opioid peptides which are found in the bloodstream, and are derived from the β-chain of hemoglobin. They have antinociceptive effects via activation of the opioid receptors, and some may also play a role in blood pressure through inhibition of the angiotensin-converting enzyme (ACE), as well as cause an elevation of endogenous enkephalin levels. Some examples of hemorphins include hemorphin-4, spinorphin, and valorphin.

LY-2183240 is a drug which acts both as a potent inhibitor of the reuptake of the endocannabinoid anandamide and as an inhibitor of fatty acid amide hydrolase (FAAH), the primary enzyme responsible for degrading anandamide. This leads to markedly elevated anandamide levels in the brain, and LY-2183240 has been shown to produce both analgesic and anxiolytic effects in animal models. While LY-2183240 is a potent inhibitor of FAAH, it has relatively poor selectivity and also inhibits several other enzyme side targets. Consequently, it was never developed for clinical use, though it remains widely used in research, and has also been sold as a designer drug.
Oxytocinase is a type of enzyme that metabolizes the endogenous neuropeptide, oxytocin. The most well-characterized oxytocinase is leucyl/cystinyl aminopeptidase, which is also an enkephalinase. Other oxytocinases are also known. During pregnancy, oxytocinase plays a role in balancing concentration of oxytocin by degrading the oxytocin produced by the fetus, as production of oxytocin increases with growth of fetus. One study found that concentration level of oxytocinase increased progressively with gestational age until labor, which indicates that pregnancy development can be statistically evaluated by comparing oxytocinase levels.

RB-120 is an orally active analog of the drug RB-101. It acts as an enkephalinase inhibitor, which is used in scientific research. Via intravenous administration, it is approximately three times as potent as RB-101 or twice as potent as the isolated (S,S) isomer of RB101. However, via i.p. administration it is approximately twice as potent as racemic RB-101 and about as potent as the isolated (S,S) isomer of RB101. During i.v. administration RB120 is approximately twice as weak as morphine in terms of analgesia; however, it is 16x weaker during i.p. and p.o. administration.

RB-3007 is an orally active analogue of RB-101. It acts as an enkephalinase inhibitor, which is used in scientific research.